DNA/RNA
Note
FPR1 is a G protein-coupled receptor (GPCR), originally identified in phagocytic leukocytes, which mediates cell chemotaxis and activation in response to the bacterial chemotactic peptide N-formyl-methionyl-leucyl-phenylalanine (fMLF).
A number of host-derived chemotactic agonists of FPR1 have been identified, including formyl peptides potentially released by mitochondria of ruptured cells, Annexin I produced by activated epithelia, and a neutrophil granule protein, cathepsin G. In addition, functional FPR1 has been detected in cells of nonhematopoietic origin, such as lung epithelial cells and hepatocytes. These findings suggest that FPR1 is involved in a broader spectrum of pathophysiologic processes.
A number of host-derived chemotactic agonists of FPR1 have been identified, including formyl peptides potentially released by mitochondria of ruptured cells, Annexin I produced by activated epithelia, and a neutrophil granule protein, cathepsin G. In addition, functional FPR1 has been detected in cells of nonhematopoietic origin, such as lung epithelial cells and hepatocytes. These findings suggest that FPR1 is involved in a broader spectrum of pathophysiologic processes.
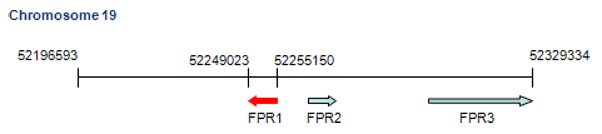
This gene is located in formylpeptide receptor gene cluster region including FPR1, FPR2 and FPR3 on chromosome 19p.
Description
Size: 6127 bases.
Transcription
All three genes, FPR1, FPR2 and FPR3, are clustered on chromosome 19q13.3. FPR1 is encoded by a 6 kb single copy gene. The open reading frame is intronless but the 5 untranslated region resides in three exons. The start sites for transcription and translation are separated by approximately 5 kb. The FPR1 gene contains three Alu repeats, one in each intron and a third in the 3 flanking region. The proposed promoter contains a nonconsensus TATA box and an inverted CCAAT element.
Pseudogene
No known pseudogenes.
Proteins
Note
FPR1 gene encodes a putative 350 aminoacid protein with seven transmembrane segments, three extra- and two intra-cellular loops.
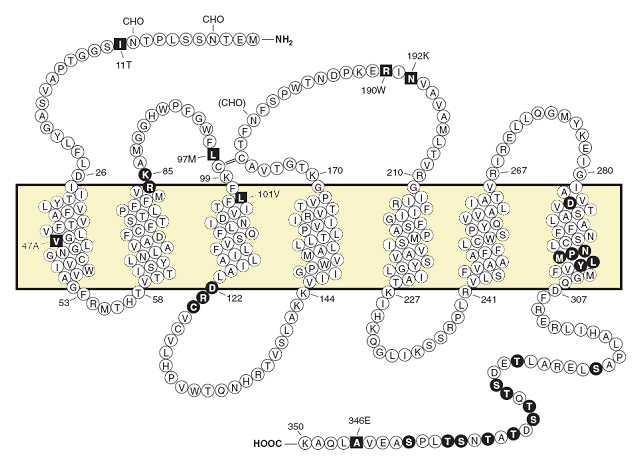
Predicted transmembrane disposition of the human FPR1.
Description
The protein sequence of the FPR-98 isoform (Leu110, Ala346) is shown (Boulay et al.,1990; Ye et al., 2009). The transmembrane domains (TMs) are predicted based on hydrophobicity of the amino acid sequence and on similarities to the rhodopsin structure. The amino acids that form the boundaries of the transmembrane domains are numbered. One-letter amino acid code is used. The square blocks in reverce color represent positions at which amino acid substitutions result from polymorphisms, including amino acids 11 (Ile/Thr), 47 (Val/Ala), 101 (Leu/Val), 190 (Arg/Trp), 192 (Asn/Lys) and 346 (Ala/Glu). The circle blocks in reverse color indicate amino acids with known functions as follows. Arg84, Lys85, and Asp284 are critical for high-affinity binding of fMLF (Mills et al., 1998; Quehenberger et al., 1997). Asp122, Arg123, and Cys124 are the signature sequence for G protein interaction (DRY in many GPCRs). NPMLY in the TM7 are known signature sequence (NPXXY) for receptor internalization (Gripentrog et al., 2000; He et al., 2001). The 11 Ser and Thr residues in the cytoplasmic tail are potential phosphorylation sites for GRK2 and GRK3 (Prossnitz et al., 1995). CHO, carbohydrate, marks the identified and potential (in parenthesis) sites for N-glycosylation. The predicted disulfide bond between Cys98 and Cys176 is marked with double-line (=).
Expression
FPR1 has been detected in phagocytic leukocytes, hepatocytes, dendritic cells, astrocytes, microglia cells, and the tunica media of coronary arteries. Becker et al. showed that FPR1 or an antigenically similar receptor is located in a number of human tissues and organs, including secretory cells in the thyroid, adrenals and other glands, the liver, the central nervous system, and neurons in the autonomic nervous system. FPR1 is also expressed in neutrophils of non-human primates and rodents.
Localisation
Cell membrane.
Function
Agonist binding to FPR1 elicits a cascade of signal transduction pathways that involve phosphatidylinositol 3-kinase (PI3K), mitogen-activated protein kinases (MAPK), and the transcription factors nuclear factor-κB and hypoxic inducible factor-1α (HIF-1α). Because of its expression in cells of the immune system and its interaction with bacterial chemotactic peptides, this receptor was thought to participate in host defense against microbial infection. In addition, FPR1 expressed in highly malignant human glioblastoma promotes tumor progression.
Homology
In primates, the sequence of FPR1 is highly conserved. Rabbit and mouse FPR1 share 78 and 76% identity with human FPR1 respectively.
Mutations
Note
Two loss of funtion mutations (F110S and C126W) that correlate with localized juvenile periodontitis. The F110S mutation resides in the third transmembrane domain, whereas the C126W mutation resides in the second intracellular loop.
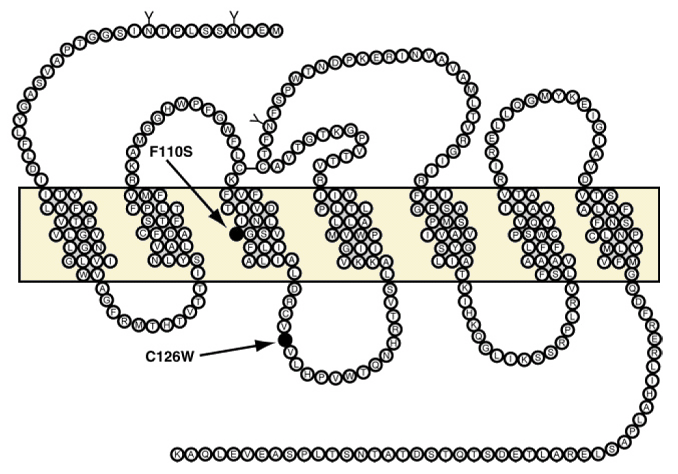
Amino acid sequence of FPR-WT and localization of the F110S and C126W mutations (Seifert et al., 2001). Shown is the two-dimensional structure of FPR-WT (isoform 26) (27). Amino acids are given in one-letter code. The FPR N terminus (top) faces the extracellular space; the FPR C terminus (bottom) faces the cytosol. The transmembrane domains are included in the boxed area. Extracellular consensus sites for N-glycosylation are shown (Y). The positions of the F110S and C126W mutations are indicated (•). There is a disulfide bridge between the first and second extracellular loops. Note that the consensus sites for N-glycosylation are not altered in FPR-F110S and FPR-C126W.
Implicated in
Entity name
Glioblastoma
Note
Promoting glioblastoma progression.
Prognosis
FPR1 protein staining was detected in 11 of 14 grade III anaplastic astrocytoma specimens and six of six grade IV glioblastoma multiforme specimens. Microvessels and necrotic tumor cells were readily visible among FPR1-positive intact tumor cells. In contrast, only two of 13 less aggressive grade II astrocytoma specimens showed positive FPR staining. Thus, FPR expression appears to be associated with a majority of poorly differentiated primary human gliomas of grades III and IV.
Cytogenetics
Highly malignant human glioblastoma and anaplastic astrocytoma specimens were stained positively for FPR1. FPR1 was expressed selectively in glioma cell lines with a more highly malignant phenotype. FPR expressed in glioblastoma cell lines mediates cell chemotaxis, proliferation and production of an angiogenic factor, vascular endothelial growth factor (VEGF), in response to agonists released by necrotic tumor cells. Furthermore, FPR in glioblastoma cells activates the receptor for epidermal growth factor (EGFR) by increasing the phosphorylation of a selected tyrosine residue in the intracellular tail of EGFR. Thus, FPR hijacked by human glioblastoma cells exploits the function of EGFR to promote rapid tumor progression.
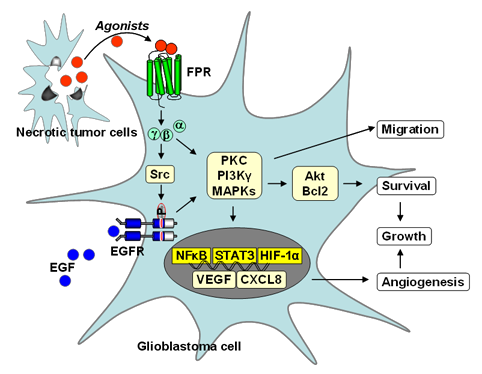
The role of FPR in glioblastoma progression. FPR on glioblastoma cells is activated by agonists released by necrotic tumor cells. The signaling cascade coupled to FPR in tumor cells activates PI3 kinase, MAPKs, PLC, PLD, Akt/Bcl2 and transcription factors such as NFκB, STAT3 and HIF-1α, to enhance cell chemotaxis, growth and release of angiogenic factors. The FPR function in glioblastoma cells is partially mediated by EGFR through a Src-kinase dependent transactivation pathway (Huang et al., 2008).
Entity name
Inflammation
Note
Mediating neutrophil accumulation at the sites of injury.
Entity name
Antibacteria host defense
Note
Mediating host resistance against Listeria infection.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 9506920 | 1998 | Broad immunocytochemical localization of the formylpeptide receptor in human organs, tissues, and cells. | Becker EL et al |
| 2161213 | 1990 | Synthesis and use of a novel N-formyl peptide derivative to isolate a human N-formyl peptide receptor cDNA. | Boulay F et al |
| 9989980 | 1999 | Impaired antibacterial host defense in mice lacking the N-formylpeptide receptor. | Gao JL et al |
| 11085933 | 2000 | A single amino acid substitution (N297A) in the conserved NPXXY sequence of the human N-formyl peptide receptor results in inhibition of desensitization and endocytosis, and a dose-dependent shift in p42/44 mitogen-activated protein kinase activation and chemotaxis. | Gripentrog JM et al |
| 11238659 | 2001 | Differential roles of the NPXXY motif in formyl peptide receptor signaling. | He R et al |
| 18433988 | 2008 | Receptor "hijacking" by malignant glioma cells: a tactic for tumor progression. | Huang J et al |
| 17575160 | 2007 | Transactivation of the epidermal growth factor receptor by formylpeptide receptor exacerbates the malignant behavior of human glioblastoma cells. | Huang J et al |
| 12401407 | 2002 | Formyl-peptide receptors revisited. | Le Y et al |
| 9553101 | 1998 | Identification of a ligand binding site in the human neutrophil formyl peptide receptor using a site-specific fluorescent photoaffinity label and mass spectrometry. | Mills JS et al |
| 7836371 | 1995 | Phosphorylation of the N-formyl peptide receptor carboxyl terminus by the G protein-coupled receptor kinase, GRK2. | Prossnitz ER et al |
| 9299516 | 1997 | Identification of an N-formyl peptide receptor ligand binding domain by a gain-of-function approach. | Quehenberger O et al |
| 11559706 | 2001 | Defective Gi protein coupling in two formyl peptide receptor mutants associated with localized juvenile periodontitis. | Seifert R et al |
| 19498085 | 2009 | International Union of Basic and Clinical Pharmacology. LXXIII. Nomenclature for the formyl peptide receptor (FPR) family. | Ye RD et al |
| 15928303 | 2005 | Formylpeptide receptor FPR and the rapid growth of malignant human gliomas. | Zhou Y et al |
Other Information
Locus ID:
NCBI: 2357
MIM: 136537
HGNC: 3826
Ensembl: ENSG00000171051
Variants:
dbSNP: 2357
ClinVar: 2357
TCGA: ENSG00000171051
COSMIC: FPR1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000171051 | ENST00000304748 | P21462 |
| ENSG00000171051 | ENST00000594900 | M0QZT0 |
| ENSG00000171051 | ENST00000595042 | P21462 |
| ENSG00000171051 | ENST00000600815 | M0R315 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38438091 | 2024 | FPR1: A critical gatekeeper of the heart and brain. | 0 |
| 38438091 | 2024 | FPR1: A critical gatekeeper of the heart and brain. | 0 |
| 36750693 | 2023 | Formyl peptide receptor 2 is an emerging modulator of inflammation in the liver. | 6 |
| 36970071 | 2023 | Rs867228 in FPR1 accelerates the manifestation of luminal B breast cancer. | 3 |
| 36750693 | 2023 | Formyl peptide receptor 2 is an emerging modulator of inflammation in the liver. | 6 |
| 36970071 | 2023 | Rs867228 in FPR1 accelerates the manifestation of luminal B breast cancer. | 3 |
| 35217703 | 2022 | Molecular recognition of formylpeptides and diverse agonists by the formylpeptide receptors FPR1 and FPR2. | 23 |
| 35382852 | 2022 | Breast cancer metastasis to brain results in recruitment and activation of microglia through annexin-A1/formyl peptide receptor signaling. | 0 |
| 35878025 | 2022 | Agonist concentration-dependent changes in FPR1 conformation lead to biased signaling for selective activation of phagocyte functions. | 2 |
| 36309087 | 2022 | Amyloid beta and its naturally occurring N-terminal variants are potent activators of human and mouse formyl peptide receptor 1. | 3 |
| 35217703 | 2022 | Molecular recognition of formylpeptides and diverse agonists by the formylpeptide receptors FPR1 and FPR2. | 23 |
| 35382852 | 2022 | Breast cancer metastasis to brain results in recruitment and activation of microglia through annexin-A1/formyl peptide receptor signaling. | 0 |
| 35878025 | 2022 | Agonist concentration-dependent changes in FPR1 conformation lead to biased signaling for selective activation of phagocyte functions. | 2 |
| 36309087 | 2022 | Amyloid beta and its naturally occurring N-terminal variants are potent activators of human and mouse formyl peptide receptor 1. | 3 |
| 33489470 | 2021 | A major genetic accelerator of cancer diagnosis: rs867228 in FPR1. | 4 |
Citation
Jian Huang ; Ji Ming Wang
FPR1 (formyl peptide receptor 1)
Atlas Genet Cytogenet Oncol Haematol. 2012-06-01
Online version: http://atlasgeneticsoncology.org/gene/44328/fpr1
