LOXL2 (lysyl oxidase-like 2)
2009-02-01 Sheri FT Fong , Keith SK Fong , Katalin Csiszar AffiliationJohn A. Burns School of Medicine, University of Hawaii, 1960 East West Road, Biomed T415, Honolulu, HI 96822, USA
Identity
HGNC
LOCATION
8p21.3
LOCUSID
ALIAS
LOR,LOR2,WS9-14
FUSION GENES
DNA/RNA
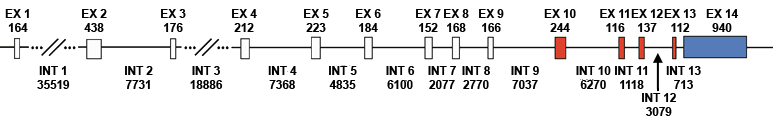
Figure 1. Lysyl oxidase-like 2 gene structure. Exons are depicted as boxes separated by intron sequences (solid lines). The size of each exon and intron is shown in base pairs, above and below the gene, respectively. The exons shaded in red encode amino acids sequences that are conserved in all lysyl oxidase family members. The exon shaded in blue contains the 3 UTR sequence.
Description
The LOXL2 gene is located on chromosome 8p21.2-p21.3 (Jourdan-Le Saux et al., 1998). It is composed of fourteen exons and thirteen introns, distributed through approximately 107 kb of genomic DNA (Fong et al., 2007). Two transcripts of sizes 3.6 kb and 4.9 kb have been reported, with the smaller transcript much more abundant and resulting from three possible termination sites located 690 bp, 740 bp and 900 bp 3 of the termination codon (Jourdan-Le Saux et al., 1999). The termination site used for the larger 4.9 kb transcript has not been described. Intron 4 contains a polymorphic CA-repeat microsatellite (Fong et al., 2007) and there are at least 17 SNPs within the LOXL2 gene (Akagawa et al., 2007). The LOXL2 gene also contains a CpG island of approximately 1150 bp, starting 176 bases upstream of the transcriptional start and extending into intron 1 (Fong et al., 2007).
Transcription
The LOXL2 promoter region contains numerous putative binding sites for transcription factors, including WT1, SP1, NF-kB and AP-1 (Fong et al., 2007).
LOXL2 gene expression is induced by TGF-b1 or indomethacin, and inhibited by retinoic acid or phorbol ester (Saito et al., 1997). It has also been described as a type II TGF-b receptor-dependent gene in lung adenocarcinoma subtypes (Borczuk et al., 2005). LOXL2 expression could be induced in MCF-7 breast cancer cells cultured on a collagen I matrix conditioned by fibroblasts (Kirschmann et al., 2002), in H292 human airway epithelial cells exposed to mite allergen (Vroling et al., 2007), and in MC3T3-E1 osteoblastic cells exposed to active vitamin D3 in culture (Nagaoka et al., 2008). LOXL2 is also induced by hypoxia. Hypoxia-inducible factor-1 alpha (HIF-1a) stimulates LOXL2 mRNA transcription in fibroblasts and renal tubular epithelial cells (Higgins et al., 2007; Salnikow et al., 2008). This upregulation was inhibited in fibroblasts by siRNA targeting the ETS1 transcription factor (Salnikow et al., 2008).
Transcription of the LOXL2 gene may also be affected by the methylation status of its CpG island (Fong et al., 2007).
LOXL2 gene expression is induced by TGF-b1 or indomethacin, and inhibited by retinoic acid or phorbol ester (Saito et al., 1997). It has also been described as a type II TGF-b receptor-dependent gene in lung adenocarcinoma subtypes (Borczuk et al., 2005). LOXL2 expression could be induced in MCF-7 breast cancer cells cultured on a collagen I matrix conditioned by fibroblasts (Kirschmann et al., 2002), in H292 human airway epithelial cells exposed to mite allergen (Vroling et al., 2007), and in MC3T3-E1 osteoblastic cells exposed to active vitamin D3 in culture (Nagaoka et al., 2008). LOXL2 is also induced by hypoxia. Hypoxia-inducible factor-1 alpha (HIF-1a) stimulates LOXL2 mRNA transcription in fibroblasts and renal tubular epithelial cells (Higgins et al., 2007; Salnikow et al., 2008). This upregulation was inhibited in fibroblasts by siRNA targeting the ETS1 transcription factor (Salnikow et al., 2008).
Transcription of the LOXL2 gene may also be affected by the methylation status of its CpG island (Fong et al., 2007).
Pseudogene
No known pseudogene.
Proteins
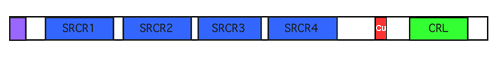
Figure 2. Lysyl oxidase-like 2 protein structure. All members of the lysyl oxidase family of proteins share two highly conserved domains: a unique copper-binding (Cu) domain containing four histidines, shaded in red; and a cytokine-receptor like (CRL) domain similar to type I cytokine receptors, shaded in green. LOXL2, LOXL3 and LOXL4 also contain four Scavenger Receptor Cysteine-Rich (SRCR) domains, shaded in blue, that are commonly found in cell surface receptors and adhesion molecules. The predicted signal sequence is shaded in purple.
Description
The LOXL2 transcript encodes for a 774-amino acid protein containing a predicted signal peptide of 22 amino acids, yielding an 87 kDa protein based on sequence analysis and produced by in vitro translation. In addition, there are three potential N-linked glycosylation sites (Saito et al., 1997). Western analysis of secreted proteins from LOXL2-transfected cells detected a protein of approximately 95 kDa, likely a glycosylated LOXL2 protein, and a protein of 63 kDa, which is an extracellular proteolytically processed form of LOXL2 (Akiri et al., 2003; Vadasz et al., 2005; Fong et al., 2007; Hollosi et al., 2009). Western analysis of cellular proteins isolated from LOXL2-transfected normal mammary epithelial cells revealed a 95 kDa protein similar to the secreted form and no processed form in the soluble cellular protein collection, with two additional LOXL2 proteins in the insoluble cellular protein collection: a 105 kDa protein that may be an alternately glycosylated full-length form, as well as a 50 kDa protein, which is another proteolytically processed form of LOXL2 (Hollosi et al., 2009). Although other LOX family members are processed extracellularly into their mature forms by bone morphogenetic protein-1 and related enzymes (Borel et al., 2001; Csiszar, 2001; Uzel et al., 2001), no potential processing site(s) or responsible enzymes have been identified for LOXL2.
Expression
Tissue expression of LOXL2 mRNA has been described in extracts of spleen, thymus, prostate, testis, uterus, small intestine, colon, heart, brain, placenta, lung, liver, kidney, pancreas, skeletal muscle and bone (Jourdan-Le Saux et al., 1999; Pires Martins et al., 2001). LOXL2 was more highly expressed in fetal heart compared to adult heart, and was found in the left and right atrium and ventricles, and the apex of the adult heart, as well as the aorta (Pires Martins et al., 2001; Molnar et al., 2003).
As for normal cell types, LOXL2 mRNA expression has been reported in fibroblasts (Saito et al., 1997; Pires Martins et al., 2001; Kirschmann et al., 2002), melanocytes and B-cells (Pires Martins et al., 2001), syncytiotrophoblasts and cytotrophoblasts (Jourdan-Le Saux et al., 1999; Hein et al., 2001), bone marrow stromal cells (Monticone et al., 2004), and astrocytes from the optic nerve head (Urban et al., 2007). LOXL2 protein has been detected in syncytiotrophoblasts and cytotrophoblasts (Hein et al., 2001), bone marrow stromal cells (Monticone et al., 2004), colonic enteroendocrine cells and esophageal squamous cells (Fong et al., 2007), and mammary epithelial cells (Hollosi et al., 2009).
As for normal cell types, LOXL2 mRNA expression has been reported in fibroblasts (Saito et al., 1997; Pires Martins et al., 2001; Kirschmann et al., 2002), melanocytes and B-cells (Pires Martins et al., 2001), syncytiotrophoblasts and cytotrophoblasts (Jourdan-Le Saux et al., 1999; Hein et al., 2001), bone marrow stromal cells (Monticone et al., 2004), and astrocytes from the optic nerve head (Urban et al., 2007). LOXL2 protein has been detected in syncytiotrophoblasts and cytotrophoblasts (Hein et al., 2001), bone marrow stromal cells (Monticone et al., 2004), colonic enteroendocrine cells and esophageal squamous cells (Fong et al., 2007), and mammary epithelial cells (Hollosi et al., 2009).
Localisation
In cell culture, LOXL2 has been detected extracellularly (Akiri et al., 2003; Vadasz et al., 2005; Fong et al., 2007; Hollosi et al., 2009) and intracellularly (Fong et al., 2007; Hollosi et al., 2009), and also localized to the perinuclear compartment (Peinado et al., 2005). In tissues, LOXL2 has been localized to the cytoplasm in colonic enteroendocrine cells and esophageal squamous cells towards the lumen surface (Fong et al., 2007), and associated with the luminal membrane surface in mammary gland acini (Hollosi et al., 2009).
Function
LOXL2, similar to its other LOX family members, has been reported to have amine oxidase activity, but unlike its family members, its enzyme activity was not inhibited by beta-aminoproprionitrile (BAPN) (Vadasz et al., 2005; Hollosi et al., 2009). Other LOX family members are known to oxidize peptidyl lysine and hydroxylysine residues in collagen and lysine residues in elastin to form the covalent cross-links that stabilize and insolubilize several fibrillar collagen types and elastin fibers (reviewed in Lucero and Kagan, 2006), and it has been reported that LOXL2 has catalytic activity against collagen type I (Vadasz et al., 2005). Treatment of MC3T3-E1 osteoblastic cells with active vitamin D3 resulted in increased LOXL2 expression (other LOX family members were not up-regulated), and increased collagen cross-links and acceleration of cross-link maturation (Nagaoka et al., 2008). As for its role in elastin fiber assembly, LOXL2 has been reported to interact with fibulin-5, which could tether LOXL2 onto microfibrils to facilitate elastic fiber assembly and maturation (Hirai et al., 2007). A single nucleotide polymorphism (SNP) in LOXL2 has been described to have an interactive effect with SNPs in the elastin/LIM kinase 1 locus in intracranial aneurysm susceptibility (Akagawa et al., 2007), and LOXL2 silencing repressed elastin gene transcription and eliminated elastic fiber formation by human umbilical vein endothelial cells (HUVEC) (Lelievre et al., 2008).
In diseases, LOXL2 has been reported to enhance the in vivo accumulation and deposition of collagen in breast tumors and glioma tumors formed by LOXL2-overexpressing cancer cells (Akiri et al., 2003). Increased LOXL2 has been described in hepatocytes from patients with Wilsons disease or primary biliary cirrhosis, which are fibrotic liver diseases (Vadasz et al., 2005); renal tubulointerstitial fibrosis from experimental unilateral ureteral obstruction and human renal samples of diabetic nephropathy, IgA nephropathy and hypertensive nephrosclerosis (Higgins et al., 2007); as well as a mouse model of chronic cholangitis which is similar to primary sclerosing cholangitis in humans (Nakken et al., 2007). Decreased LOXL2 mRNA expression was noted in human pelvic organ prolapse (Klutke et al., 2008).
LOXL2 is reduced in endothelial cells exposed to laminar shear stress, which induces reduction of atherogenicity (Chu and Peters, 2008). VE-statin/egfl7 that increases after arterial injury (Campagnolo et al., 2005), may modulate smooth muscle migration through interaction with the catalytic domain of LOXL2 and all LOX enzymes to inhibit enzyme activity (Lelievre et al., 2008). Increased LOXL2 has also been described in intracranial aneurysms, associated with a SNP in exon 5 (Akagawa et al., 2007).
LOXL2 is the only LOX family member not expressed in MC3T3-E1 osteoblastic cells (Atsawasuwan et al., 2005), and not up-regulated upon osteoblast differentiation (Kaku et al., 2007). Although expressed in bone marrow stromal cells (BMSC), LOXL2 was down-regulated in osteoblasts derived from BMSC (Qi et al., 2003; Monticone et al., 2004).
In addition, increased LOXL2 expression was observed in senescent fibroblasts, replicative senescence and stress-induced premature senescence (Saito et al., 1997; Pascal et al., 2005), and may be involved in primary open-angle glaucoma (Urban et al., 2007). LOXL2 also appears to have a role in cell proliferation (Akiri et al., 2003; Vadasz et al., 2005).
LOXL2 also has been described to have multiple roles in cancer, including epithelial-mesenchymal transition and the promotion of cancer cell adhesion, migration, invasion and metastases, and these are described in more detail in the following sections.
In diseases, LOXL2 has been reported to enhance the in vivo accumulation and deposition of collagen in breast tumors and glioma tumors formed by LOXL2-overexpressing cancer cells (Akiri et al., 2003). Increased LOXL2 has been described in hepatocytes from patients with Wilsons disease or primary biliary cirrhosis, which are fibrotic liver diseases (Vadasz et al., 2005); renal tubulointerstitial fibrosis from experimental unilateral ureteral obstruction and human renal samples of diabetic nephropathy, IgA nephropathy and hypertensive nephrosclerosis (Higgins et al., 2007); as well as a mouse model of chronic cholangitis which is similar to primary sclerosing cholangitis in humans (Nakken et al., 2007). Decreased LOXL2 mRNA expression was noted in human pelvic organ prolapse (Klutke et al., 2008).
LOXL2 is reduced in endothelial cells exposed to laminar shear stress, which induces reduction of atherogenicity (Chu and Peters, 2008). VE-statin/egfl7 that increases after arterial injury (Campagnolo et al., 2005), may modulate smooth muscle migration through interaction with the catalytic domain of LOXL2 and all LOX enzymes to inhibit enzyme activity (Lelievre et al., 2008). Increased LOXL2 has also been described in intracranial aneurysms, associated with a SNP in exon 5 (Akagawa et al., 2007).
LOXL2 is the only LOX family member not expressed in MC3T3-E1 osteoblastic cells (Atsawasuwan et al., 2005), and not up-regulated upon osteoblast differentiation (Kaku et al., 2007). Although expressed in bone marrow stromal cells (BMSC), LOXL2 was down-regulated in osteoblasts derived from BMSC (Qi et al., 2003; Monticone et al., 2004).
In addition, increased LOXL2 expression was observed in senescent fibroblasts, replicative senescence and stress-induced premature senescence (Saito et al., 1997; Pascal et al., 2005), and may be involved in primary open-angle glaucoma (Urban et al., 2007). LOXL2 also appears to have a role in cell proliferation (Akiri et al., 2003; Vadasz et al., 2005).
LOXL2 also has been described to have multiple roles in cancer, including epithelial-mesenchymal transition and the promotion of cancer cell adhesion, migration, invasion and metastases, and these are described in more detail in the following sections.
Homology
In the human lysyl oxidase protein family, there are five members, named LOX, LOXL1, LOXL2, LOXL3 and LOXL4. They all contain a lysine tyrosylquinone (LTQ), the only mammalian cofactor derived from the cross-linking of two amino acid side chains (Anthony, 1996), and which is unique to the LOX family. The other highly conserved motif that is unique to the LOX family is the copper-binding domain, which contains four histidines (Krebs and Krawetz, 1993). All LOX family members also contain a cytokine receptor-like (CRL) domain, which has part of the consensus sequence of Class 1 cytokine receptors (Bazan, 1990). LOXL2 has closest homology to LOXL3 and LOXL4, as these three members differ from LOX and LOXL1 by the presence of 4 SRCR domains, and may represent a separate subfamily (Asuncion et al., 2001; Maki et al., 2001).
Mutations
Note
Despite the implication of LOXL2 in several diseases and disorders, there are only sparse reports of any gene loss, polymorphisms or epigenetic alterations in the LOXL2 gene.
Somatic
Loss of heterozygosity has been documented in colon and esophageal cancers at a level lower than the loss of heterozygosity documented in chromosome 8p21.2 - p21.3 for these cancers, indicating that LOXL2 is unlikely to be a tumor suppressor (Fong et al., 2007).
Epigenetics
Increase in LOXL2 expression following treatment with the demethylating agent, 5-aza-2-deoxycytidine, in colon and breast cancer cell lines indicate that the demethylation of the LOXL2 CpG island is a possible mechanism for regulating LOXL2 gene expression (Fong et al., 2007; Hollosi et al., 2009).
Implicated in
Entity name
Blood cancers
Note
LOXL2 mRNA was reported to be barely detectable or absent in the erythroleukemia cell line K562 and the T-cell leukemia cell line Jurkat (Saito et al., 1997).
Entity name
Brain cancer
Note
LOXL2 mRNA was reported to be highly expressed in the cultured astrocytoma cell line U373MG (Saito et al., 1997).
C6-glioma cells, transfected with LOXL2, enhanced the in vivo accumulation and deposition of collagen in tumors (Akiri et al., 2003).
C6-glioma cells, transfected with LOXL2, enhanced the in vivo accumulation and deposition of collagen in tumors (Akiri et al., 2003).
Entity name
Breast cancer
Note
LOXL2 mRNA was demonstrated to be up-regulated in the invasive and metastatic cell lines, MDA-MB-231, Hs578T and MDA-MB435, compared to the poorly-invasive and non-metastatic cell lines, MCF-7 and T47D (Kirschmann et al., 2002; Akiri et al., 2003; Hollosi et al., 2009). MCF-7 cells, transfected with LOXL2, became more invasive in vivo and formed tumors with collagen deposition, and LOXL2 protein expression was more prevalent in poorly-differentiated compared to well-differentiated breast carcinomas (Akiri et al., 2003). LOXL2 mRNA was demonstrated to be present in 30 of 31 (97%) effusions due to breast cancer, compared to 17 of 23 (74%) primary breast carcinomas (Sebban et al., 2009). Increased LOXL2 protein expression was associated with decreased overall survival and decreased disease-free survival in patients with lymph node-negative breast adenocarcinomas (Peinado et al., 2008). LOXL2 mRNA expression may be regulated by methylation of its CpG island (Hollosi et al., 2009).
Entity name
Cervical cancer
Note
LOXL2 mRNA was reported to be highly expressed in the adherent cultured cervical adenocarcinoma cell line HeLa, but not the non-adherent cultured cervical adenocarcinoma cell line HeLa-S3 (Saito et al., 1997).
Entity name
Colon cancer
Note
LOXL2 was reported to be expressed in tumor-bearing tissue of the colon compared to absent expression in normal colon tissue using mining of the human EST database (Pires Martins et al., 2001). Increased LOXL2 mRNA expression has been shown in colon tumors (Fong et al., 2007; Macartney-Coxson et al., 2008). Increased LOXL2 protein expression is associated with less-differentiated colon cancers, which have more aggressive behavior and high incidence in metastasis (Fong et al., 2007), and has been shown in liver metastases due to colon cancer (Macartney-Coxson et al., 2008). LOXL2 mRNA expression may be regulated by methylation of its CpG island (Fong et al., 2007).
Entity name
Duodenal cancer
Note
LOXL2 mRNA was reported to be highly expressed in the duodenal adenocarcinoma cell line HuTu80 (Saito et al., 1997).
Entity name
Epithelial-mesenchymal transition (EMT)
Note
Increased LOXL2 mRNA expression was described in immortalized kidney epithelial cells that had undergone EMT (Kiemer et al., 2001). LOXL2 has been shown to interact with Snail through the SNAG domain with Snails lysine residues 98 and 137 as possible substrates, leading to increased stability of Snail, down-regulation of E-cadherin, up-regulation of fibronectin and induction of EMT (Peinado et al., 2005). LOXL2 is thought to contribute to Snail stability by impeding GSK3b-dependent Snail degradation (Peinado et al., 2005). LOXL2 also interacts with fibulin-5 (Hirai et al., 2007), which has been implicated in EMT in mammary epithelial cells (Lee et al., 2008).
Entity name
Esophageal cancer
Note
Increased LOXL2 mRNA expression has been reported in esophageal tumors (Fong et al., 2007).
Entity name
Fibrosarcoma
Note
LOXL2 mRNA was reported to be highly expressed in the cultured fibrosarcoma cell line HT 1080 (Saito et al., 1997).
Entity name
Gastric cancer
Note
LOXL2 mRNA was reported to be barely detectable or absent in the cultured gastric carcinoma cell line KATO III (Saito et al., 1997).
Entity name
Germ cell cancer
Note
LOXL2 was reported to be expressed in tumor-bearing tissue of germ cells using mining of the human EST database (Pires Martins et al., 2001).
Entity name
Head and neck squamous cell carcinoma (HNSCC)
Note
Reduced LOXL2 mRNA levels were noted in 9 of 10 HNSCC cell lines, but mean LOXL2 expression levels between malignantly transformed tissues and reference tissues were similar (Rost et al., 2003). LOXL2 was identified in poorly-differentiated HNSCC tumors with likely strong desmoplastic response or epithelial-mesenchymal transition, and as part of a gene set predictive of high risk of recurrence in HNSCC (Chung et al., 2004, Chung et al., 2006).
In oral squamous cell carcinoma (OSCC) tissues, there was significantly higher LOXL2 expression compared to paired normal matched tissues, and LOXL2 was part of a 25-gene predictor set to distinguish normal from OSCC tissues (Ziober et al., 2006; Shieh et al., 2007). However, there was higher LOXL2 expression in cultured normal oral mucosal cells than OSCC cells (Shieh et al., 2007).
Increased LOXL2 protein expression was reported to correlate with local recurrence, decreased overall survival and decreased disease-free survival in patients with laryngeal squamous cell carcinoma (Peinado et al., 2008).
In oral squamous cell carcinoma (OSCC) tissues, there was significantly higher LOXL2 expression compared to paired normal matched tissues, and LOXL2 was part of a 25-gene predictor set to distinguish normal from OSCC tissues (Ziober et al., 2006; Shieh et al., 2007). However, there was higher LOXL2 expression in cultured normal oral mucosal cells than OSCC cells (Shieh et al., 2007).
Increased LOXL2 protein expression was reported to correlate with local recurrence, decreased overall survival and decreased disease-free survival in patients with laryngeal squamous cell carcinoma (Peinado et al., 2008).
Entity name
Lung cancer
Note
LOXL2 was reported to be barely detectable or absent in the cultured lung small cell carcinoma cell line NCI-H69 (Saito et al., 1997). Repression of type II TGF-b receptor by siRNA in the lung carcinoma cell line H23, was associated with increased number of invasive cells and increased levels of LOXL2 mRNA (Borczuk et al., 2005). Increased LOXL2 protein expression was reported to be associated with decreased overall survival and decreased disease-free survival in patients with lung squamous cell carcinoma (Peinado et al., 2008).
Entity name
Malignant Mesothelioma
Note
LOXL2 mRNA expression was noted to be higher in effusions due to malignant mesothelioma than in effusions due to benign reactive mesothelium (Sebban et al., 2009).
Entity name
Melanoma
Note
LOXL2 mRNA was reported to be highly expressed in the lymph node melanoma cell line Hs294T (Saito et al., 1997). LOXL2 mRNA expression was also highly expressed in the highly invasive uveal melanoma cell lines M619 and C918, compared to absent expression in the poorly invasive uveal melanoma cell line OCM-1A (Kirschmann et al., 2002).
Entity name
Ovarian cancer
Note
Using gene expression profiling, LOXL2 mRNA expression was reported to be down-regulated in serous ovarian adenocarcinomas compared to the mucinous ovarian cancers (Hough et al., 2000; Ono et al., 2000). However, a more recent report found LOXL2 mRNA to be expressed in all types of primary ovarian cancer tissues, including serous, clear cell, endometrioid, undifferentiated and mixed type, as well as metastases and metastatic effusions (Sebban et al., 2009).
Entity name
Pancreatic cancer
Note
LOXL2 mRNA was upregulated in Panc1 pancreatic cancer cells compared to normal human pancreatic duct epithelial (HPDE) cells (Gronberg et al., 2006).
Entity name
Peripheral nerve sheath cancer
Note
LOXL2 mRNA was over-expressed in high-grade malignant peripheral nerve sheath tumors, which are highly aggressive (Kresse et al., 2008).
Entity name
Ras-transformation
Note
LOXL2 mRNA expression was down-regulated nearly 60 fold in ras-transformed fibroblasts compared to immortalized rat embryonic fibroblasts (Zuber et al., 2000).
Entity name
Skin cancer
Note
Knockdown of LOXL2 mRNA in the mouse malignant squamous cell carcinoma cell line HaCa4 and the spindle cell carcinoma cell line CarB resulted in decreased tumor growth, increased apoptosis, re-expression of E-cadherin, decreased expression of fibronectin, vimentin and MMP-9 (Peinado et al., 2005). LOXL2 knockdown in HaCa4 also resulted in increased cell-cell contacts, elimination of in vivo invasive ability and a skin-like epidermal differentiation program with re-expression and organization of E-cadherin at suprabasal layers. Expression of LOXL2 was correlated with malignant progression in a mouse skin carcinogenesis model (Peinado et al., 2008).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 17287949 | 2007 | Systematic screening of lysyl oxidase-like (LOXL) family genes demonstrates that LOXL2 is a susceptibility gene to intracranial aneurysms. | Akagawa H et al |
| 12670920 | 2003 | Lysyl oxidase-related protein-1 promotes tumor fibrosis and tumor progression in vivo. | Akiri G et al |
| 9003352 | 1996 | Quinoprotein-catalysed reactions. | Anthony C et al |
| 11691588 | 2001 | A novel human lysyl oxidase-like gene (LOXL4) on chromosome 10q24 has an altered scavenger receptor cysteine rich domain. | Asuncion L et al |
| 15652501 | 2005 | Expression of lysyl oxidase isoforms in MC3T3-E1 osteoblastic cells. | Atsawasuwan P et al |
| 2169613 | 1990 | Structural design and molecular evolution of a cytokine receptor superfamily. | Bazan JF et al |
| 15976377 | 2005 | Lung adenocarcinoma global profiling identifies type II transforming growth factor-beta receptor as a repressor of invasiveness. | Borczuk AC et al |
| 11684696 | 2001 | Lysyl oxidase-like protein from bovine aorta. Isolation and maturation to an active form by bone morphogenetic protein-1. | Borel A et al |
| 15972971 | 2005 | EGFL7 is a chemoattractant for endothelial cells and is up-regulated in angiogenesis and arterial injury. | Campagnolo L et al |
| 18505769 | 2008 | Serial analysis of the vascular endothelial transcriptome under static and shear stress conditions. | Chu TJ et al |
| 16912200 | 2006 | Gene expression profiles identify epithelial-to-mesenchymal transition and activation of nuclear factor-kappaB signaling as characteristics of a high-risk head and neck squamous cell carcinoma. | Chung CH et al |
| 11642359 | 2001 | Lysyl oxidases: a novel multifunctional amine oxidase family. | Csiszar K et al |
| 17394133 | 2007 | Lysyl oxidase-like 2 expression is increased in colon and esophageal tumors and associated with less differentiated colon tumors. | Fong SF et al |
| 16215274 | 2006 | Biomarker discovery from pancreatic cancer secretome using a differential proteomic approach. | Grønborg M et al |
| 11162352 | 2001 | Lysyl oxidases: expression in the fetal membranes and placenta. | Hein S et al |
| 18037992 | 2007 | Hypoxia promotes fibrogenesis in vivo via HIF-1 stimulation of epithelial-to-mesenchymal transition. | Higgins DF et al |
| 17371835 | 2007 | Fibulin-5/DANCE has an elastogenic organizer activity that is abrogated by proteolytic cleavage in vivo. | Hirai M et al |
| 11103784 | 2000 | Large-scale serial analysis of gene expression reveals genes differentially expressed in ovarian cancer. | Hough CD et al |
| 10212285 | 1999 | The LOXL2 gene encodes a new lysyl oxidase-like protein and is expressed at high levels in reproductive tissues. | Jourdan-Le Saux C et al |
| 17553463 | 2007 | Post-translational modifications of collagen upon BMP-induced osteoblast differentiation. | Kaku M et al |
| 11709702 | 2001 | Identification of genes involved in epithelial-mesenchymal transition and tumor progression. | Kiemer AK et al |
| 12154058 | 2002 | A molecular role for lysyl oxidase in breast cancer invasion. | Kirschmann DA et al |
| 18158636 | 2008 | Decreased endopelvic fascia elastin content in uterine prolapse. | Klutke J et al |
| 8104038 | 1993 | Lysyl oxidase copper-talon complex: a model. | Krebs CJ et al |
| 18522746 | 2008 | DNA copy number changes in high-grade malignant peripheral nerve sheath tumors by array CGH. | Kresse SH et al |
| 18713838 | 2008 | Fibulin-5 initiates epithelial-mesenchymal transition (EMT) and enhances EMT induced by TGF-beta in mammary epithelial cells via a MMP-dependent mechanism. | Lee YH et al |
| 18497746 | 2008 | VE-statin/egfl7 regulates vascular elastogenesis by interacting with lysyl oxidases. | Lelièvre E et al |
| 16909208 | 2006 | Lysyl oxidase: an oxidative enzyme and effector of cell function. | Lucero HA et al |
| 18590575 | 2008 | Metastatic susceptibility locus, an 8p hot-spot for tumour progression disrupted in colorectal liver metastases: 13 candidate genes examined at the DNA, mRNA and protein level. | Macartney-Coxson DP et al |
| 11691589 | 2001 | Cloning and characterization of a fifth human lysyl oxidase isoenzyme: the third member of the lysyl oxidase-related subfamily with four scavenger receptor cysteine-rich domains. | Mäki JM et al |
| 12686136 | 2003 | Structural and functional diversity of lysyl oxidase and the LOX-like proteins. | Molnar J et al |
| 15211571 | 2004 | Gene expression profile of human bone marrow stromal cells determined by restriction fragment differential display analysis. | Monticone M et al |
| 18930711 | 2008 | 1,25(OH)2D3 regulates collagen quality in an osteoblastic cell culture system. | Nagaoka H et al |
| 17852852 | 2007 | Multiple inflammatory-, tissue remodelling- and fibrosis genes are differentially transcribed in the livers of Abcb4 (-/ - ) mice harbouring chronic cholangitis. | Nakken KE et al |
| 11016619 | 2000 | Identification by cDNA microarray of genes involved in ovarian carcinogenesis. | Ono K et al |
| 15963989 | 2005 | Comparison of replicative senescence and stress-induced premature senescence combining differential display and low-density DNA arrays. | Pascal T et al |
| 16096638 | 2005 | A molecular role for lysyl oxidase-like 2 enzyme in snail regulation and tumor progression. | Peinado H et al |
| 18559498 | 2008 | Lysyl oxidase-like 2 as a new poor prognosis marker of squamous cell carcinomas. | Peinado H et al |
| 16294032 | 2005 | Switching on-off Snail: LOXL2 versus GSK3beta. | Peinado H et al |
| 11247664 | 2001 | Whole-body gene expression by data mining. | Pires Martins R et al |
| 12631704 | 2003 | Identification of genes responsible for osteoblast differentiation from human mesodermal progenitor cells. | Qi H et al |
| 12820424 | 2003 | Reduction of LOX- and LOXL2-mRNA expression in head and neck squamous cell carcinomas. | Rost T et al |
| 9079631 | 1997 | Regulation of a novel gene encoding a lysyl oxidase-related protein in cellular adhesion and senescence. | Saito H et al |
| 18381358 | 2008 | Regulation of hypoxia-inducible genes by ETS1 transcription factor. | Salnikow K et al |
| 19015874 | 2009 | Lysyl oxidase-like 4 is alternatively spliced in an anatomic site-specific manner in tumors involving the serosal cavities. | Sebban S et al |
| 17671119 | 2007 | Association of expression aberrances and genetic polymorphisms of lysyl oxidase with areca-associated oral tumorigenesis. | Shieh TM et al |
| 17591890 | 2007 | Population differences in elastin maturation in optic nerve head tissue and astrocytes. | Urban Z et al |
| 11313359 | 2001 | Multiple bone morphogenetic protein 1-related mammalian metalloproteinases process pro-lysyl oxidase at the correct physiological site and control lysyl oxidase activation in mouse embryo fibroblast cultures. | Uzel MI et al |
| 16023247 | 2005 | Abnormal deposition of collagen around hepatocytes in Wilson's disease is associated with hepatocyte specific expression of lysyl oxidase and lysyl oxidase like protein-2. | Vadasz Z et al |
| 17919147 | 2007 | Allergen induced gene expression of airway epithelial cells shows a possible role for TNF-alpha. | Vroling AB et al |
| 17062667 | 2006 | Identification of a gene signature for rapid screening of oral squamous cell carcinoma. | Ziober AF et al |
| 10655059 | 2000 | A genome-wide survey of RAS transformation targets. | Zuber J et al |
Other Information
Locus ID:
NCBI: 4017
MIM: 606663
HGNC: 6666
Ensembl: ENSG00000134013
Variants:
dbSNP: 4017
ClinVar: 4017
TCGA: ENSG00000134013
COSMIC: LOXL2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38520980 | 2024 | Simultaneous disturbance of NHE1 and LOXL2 decreases tumorigenicity of head and neck squamous cell carcinoma. | 0 |
| 38520980 | 2024 | Simultaneous disturbance of NHE1 and LOXL2 decreases tumorigenicity of head and neck squamous cell carcinoma. | 0 |
| 35428659 | 2023 | Macrophages direct cancer cells through a LOXL2-mediated metastatic cascade in pancreatic ductal adenocarcinoma. | 17 |
| 36512309 | 2023 | TRIM44 regulates tumor immunity in gastric cancer through LOXL2-dependent extracellular matrix remodeling. | 4 |
| 36573458 | 2023 | LOXL2 reduces 5-FU sensitivity through the Hedgehog/BCL2 signaling pathway in colorectal cancer. | 2 |
| 36894050 | 2023 | Hypoxia preconditioned DPSC-derived exosomes regulate angiogenesis via transferring LOXL2. | 4 |
| 37345575 | 2023 | Homozygous LOXL2 variant in individuals affected by non-syndromic occipital encephalocele. | 0 |
| 37458436 | 2023 | Lysyl oxidase like-2 in fibrosis and cardiovascular disease. | 3 |
| 37496288 | 2023 | Lysyl oxidase-like 2 promotes stemness and enhances antitumor effects of gefitinib in head and neck cancer via IFIT1 and IFIT3. | 3 |
| 37511503 | 2023 | Multiple Roles of LOXL2 in the Progression of Hepatocellular Carcinoma and Its Potential for Therapeutic Targeting. | 2 |
| 37737908 | 2023 | The role of LOXL2 induced by glucose metabolism-activated NF-κB in maintaining drug resistance through EMT and cancer stemness in gemcitabine-resistant PDAC. | 2 |
| 35428659 | 2023 | Macrophages direct cancer cells through a LOXL2-mediated metastatic cascade in pancreatic ductal adenocarcinoma. | 17 |
| 36512309 | 2023 | TRIM44 regulates tumor immunity in gastric cancer through LOXL2-dependent extracellular matrix remodeling. | 4 |
| 36573458 | 2023 | LOXL2 reduces 5-FU sensitivity through the Hedgehog/BCL2 signaling pathway in colorectal cancer. | 2 |
| 36894050 | 2023 | Hypoxia preconditioned DPSC-derived exosomes regulate angiogenesis via transferring LOXL2. | 4 |
Citation
Sheri FT Fong ; Keith SK Fong ; Katalin Csiszar
LOXL2 (lysyl oxidase-like 2)
Atlas Genet Cytogenet Oncol Haematol. 2009-02-01
Online version: http://atlasgeneticsoncology.org/gene/41192/loxl2
