NAT2 (N-acetyltransferase 2 (arylamine N-acetyltransferase))
2009-01-01 Jhon D Ruiz , José AG Agúndez , Carmen Martínez , Elena García-Martín AffiliationDepartment of Pharmacology, Medical School, University of Extremadura, Badajoz, Spain (JDR, JA, CM); Department of Biochemistry & Molecular biology & Genetics, School of Biological Sciences, Badajoz, Spain (EGM)
Identity
HGNC
LOCATION
8p22
IMAGE

LEGEND
Picture from Genetics Home Reference; January 2009.
LOCUSID
ALIAS
AAC2,NAT-2,PNAT
DNA/RNA
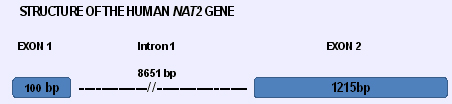
Transcription
The human NAT2 gene has two exons but the coding region, spanning 870 bp is located in exon 2. Functionally active NAT2 enzyme can be obtained after transient heterologous transfection of the open reading frame only, indicating that exon 1 is not necessary to obtain functional enzyme.
Pseudogene
In humans the NAT locus has a pseudogene designated as NATP.
Proteins
Note
NAT enzymes have been identified in several vertebrate and microorganism species. NAT2 proteins differ among species. However, common features include an 83 amino acid N-terminal domain containing five alpha-helices and a short beta-strand; a second domain consisting of nine beta-strands and two short helices; and a third alpha/beta lid domain with four beta-strands and an alpha-helix.
Expression
NAT2 has a restricted expression profile with the highest levels of protein and mRNA being detected in the liver, small intestine and colon. The transcription start site for human NAT2 has been recently localised between 30 and 101 bp upstream of the non-coding exon, with the most frequent TSS located at position -64 relative to exon 1. The region containing the NAT2 transcription start site shares an 85% sequence homology to the region of human NAT1 containing the major transcription start site for NAT1. The functional elements of the NAT2 promoter sequence have not been characterised to date. In addtion the promoter sequence appears to be highly polymorphic.
Localisation
Arylamine N-acetyltransferase 2 is a cytosolic enzyme.
Function
NAT2 is a phase II enzyme that participates in the metabolism of numerous primary arylamines and hydrazine drugs and carcinogens. In addition to their N-acetylation catalytic activity, NAT enzymes have also O-acetylation activity towards N-hydroxyarylamines.
Homology
NAT1 and NAT2 share 87% nucleotide homology in the coding region, whereas NAT1 and NAT2 proteins share 81% amino-acid sequence identity.
Mutations
Note
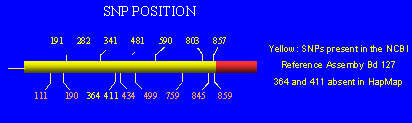
Seven major single nucleotide polymorphisms that occur isolated or combined have been described in the NAT2 gene. These affect the positions 191, 282, 341, 481, 590, 803 and 857. In addition, rare SNPs affecting the positions 111, 190, 364, 411, 434, 499, 795, 845 and 859 have been described although their frequencies are unknown. For details on NAT2 SNPs and haplotypes, see http://louisville.edu/medschool/pharmacology/Human.NAT2.pdf. Critical gene variants leading to slow acetylation capacity contains mutations at positions 191, 341, 590 or 857. Since some genotypes can be due to the presence of different combinations of haplotypes leading to ambiguous phenotype prediction, haplotype reconstruction is often necessary to clarify ambiguous genotype data.
Implicated in
Note
Determination of the NAT2 genotype or phenotype has been proposed to predict adverse reactions in patients with tuberculosis receiving isoniazid, prior to the concomitant administration of drug combinations such as procainamide-phenytoin or doxycyline-rifampin. In addition, several human diseases have been related to NAT2 polymorphism. There are described below.
Entity name
Brain cancer
Prognosis
Preliminary findings argue for association of a trend towards higher risk in individuals classified as NAT2 homozygous rapid acetylators in patients with astrocytoma or meningioma.
Entity name
Lung cancer
Prognosis
Several studies based on an initial hypothesis that slow acetylation may increase the risk of developing lung cancer have been conducted. This hypothesis has been reinforced by studies indicating that slow acetylation, especially if it is associated to defect genotypes for other phase II enzymes, may confer increased susceptibility to the formation of adducts. Several studies have concluded that the NAT2 slow acetylation genotype causes a marginally increased risk of developing lung cancer. In spite of these findings, present evidence suggests that the NAT2 polymorphism alone does not constitute a relevant risk factor for lung cancer. However this polymorphism may reinforce the effect of other genetic and/or environmental factors.
Entity name
Liver cancer
Prognosis
A role for xenobiotic-metabolising enzymes in liver carcinogenesis is to be expected among patients with environmentally-related liver cancer since, besides viral hepatitis, liver cancer may be related to environmental substances. The findings obtained in patients with primary liver cancer not related to viral hepatitis are consistent and indicate a minor, but relevant, association of the slow NAT2 acetylation status and predisposition to liver cancer.
Entity name
Colorectal cancer
Prognosis
The hypothesis that acetylator status may predispose to a determined cancer risk is based on a differential effect of N-acetylation as a potential detoxification step and O-acetylation as a potential carcinogen-activation step. In the case of colorectal cancer it was hypothesized that O-acetylation is more relevant that N-acetylation, and therefore the rapid acetylation genotype is the putative risk status associated with colorectal cancer. Sufficient evidence is available to rule out a relevant association of NAT genotypes alone with colorectal cancer risk. However, the putative interaction of meat consumption and the NAT2 genotype deserves particular attention.
Entity name
Bladder cancer
Prognosis
Despite the large number of studies and meta-analyses performed in several human populations, current evidence is not sufficient to confirm unambiguously an association of NAT2 polymorphism to overall bladder cancer risk. A general association of the slow acetylation status with bladder cancer risk has not been fully confirmed, although meta-analyses have obtained positive findings for a modest association of the slow NAT2 acetylation genotype with bladder cancer risk, with odds ratio values between 1.3 and 1.5. Furthermore, the biological basis for the putative association is uncertain. In diverse independent studies, mutagenicity in urine was tested in individuals exposed to urban pollution, smoking, red meat intake or textile dyes. In all cases, no higher mutagenicity in slow NAT2 acetylators could be established when compared to these or rapid acetylators, and in fact among individuals exposed to urban pollution, rapid acetylators showed a higher mutagenicity in urine than slow acetylators. In a study investigating the influence of NAT genotypes in the association between permanent hair dyes and bladder cancer, a significant association of the slow NAT2 acetylation genotype was identified. However these findings could not be replicated in other studies.
Entity name
Breast cancer
Prognosis
After dozens of studies involving several thousands of breast cancer patients, as well as meta-analyses, today it is obvious that no major association of NAT2 polymorphism and breast cancer risk exists.
Entity name
Head and neck cancer
Prognosis
Since chemical compounds present in tobacco are inactivated by phase II enzymes, it has been proposed that head and neck cancer risk could be modified by NAT genotypes. However, overall findings indicate that no relevant association between NAT2 polymorphism and head and neck cancer risk is to be expected.
Entity name
Other diseases
Disease
Although a relation of risk may be definitely discarded for systemic lupus erythematosus (SLE), inflammatory bowel disease and endometriosis, more research is needed for rheumatoid arthritis, Parkinsons, Alzheimers, Behçets and periodontal diseases, as current results are inconclusive but suggest a possible relation with NAT2 polymorphism. In diabetes mellitus the possible relation with the rapid phenotype may be due to acquired metabolic changes and more genotyping studies are needed. NAT2 slow metabolizers are more prone to the side effects of polymorphically acetylated drugs, as is the SLE-like syndrome induced by hydralazine and procainamide, the side effects due to sulphasalazine and the skin rash secondary to many sulphonamides.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18664443 | 2008 | Unraveling ambiguous NAT2 genotyping data. | Agúndez JA et al |
| 18680465 | 2008 | N-acetyltransferases: lessons learned from eighty years of research. | Agúndez JA et al |
| 18680475 | 2008 | N-acetyltransferases as markers for asthma and allergic/atopic disorders. | Batra J et al |
| 1675794 | 1991 | Molecular mechanism of slow acetylation of drugs and carcinogens in humans. | Blum M et al |
| 2340091 | 1990 | Human arylamine N-acetyltransferase genes: isolation, chromosomal localization, and functional expression. | Blum M et al |
| 18680469 | 2008 | Regulation of arylamine N-acetyltransferases. | Butcher NJ et al |
| 2859977 | 1985 | Genetically determined variability in acetylation and oxidation. Therapeutic implications. | Clark DW et al |
| 7695265 | 1994 | Possible implications of doxycycline-rifampin interaction for treatment of brucellosis. | Colmenero JD et al |
| 515773 | 1979 | Agranulocytosis during combined procainamide and phenytoin therapy. | Crook JE et al |
| 13820968 | 1960 | Genetic control of isoniazid metabolism in man. | EVANS DA et al |
| 13697556 | 1960 | The determination of the isoniazid inactivator phenotype. | EVANS DA et al |
| 18680468 | 2008 | Interethnic and intraethnic variability of NAT2 single nucleotide polymorphisms. | García-Martín E et al |
| 2734109 | 1989 | Nucleotide sequence of an intronless gene for a human arylamine N-acetyltransferase related to polymorphic drug acetylation. | Grant DM et al |
| 2924904 | 1989 | Evidence for two closely related isozymes of arylamine N-acetyltransferase in human liver. | Grant DM et al |
| 18680466 | 2008 | Structures of human arylamine N-acetyltransferases. | Grant DM et al |
| 8081359 | 1994 | Molecular genetics of human polymorphic N-acetyltransferase: enzymatic analysis of 15 recombinant wild-type, mutant, and chimeric NAT2 allozymes. | Hein DW et al |
| 18680473 | 2008 | Influence of polymorphic N-acetyltransferases on non-malignant spontaneous disorders and on response to drugs. | Ladero JM et al |
| 18680474 | 2008 | Human N-acetyltransferases and drug-induced hepatotoxicity. | Makarova SI et al |
| 18680470 | 2008 | Effect of environmental substances on the activity of arylamine N-acetyltransferases. | Rodrigues-Lima F et al |
| 18680471 | 2008 | Arylamine N-acetyltransferases in mycobacteria. | Sim E et al |
| 10876241 | 2000 | Structure of arylamine N-acetyltransferase reveals a catalytic triad. | Sinclair JC et al |
| 18680467 | 2008 | Structure/function evaluations of single nucleotide polymorphisms in human N-acetyltransferase 2. | Walraven JM et al |
| 5678219 | 1968 | The mechanism of isoniazid acetylation by human N-acetyltransferase. | Weber WW et al |
| 17656365 | 2007 | Structural basis of substrate-binding specificity of human arylamine N-acetyltransferases. | Wu H et al |
Other Information
Locus ID:
NCBI: 10
MIM: 612182
HGNC: 7646
Ensembl: ENSG00000156006
Variants:
dbSNP: 10
ClinVar: 10
TCGA: ENSG00000156006
COSMIC: NAT2
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000156006 | ENST00000286479 | P11245 |
| ENSG00000156006 | ENST00000286479 | A4Z6T7 |
| ENSG00000156006 | ENST00000520116 | E7EWF9 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA10715 | sulfamethoxazole / trimethoprim | Chemical | LabelAnnotation | associated | |||
| PA164712505 | Antivirals for treatment of HIV infections, combinations | Chemical | VariantAnnotation | associated | PK | PD | |
| PA164712708 | Drugs For Treatment Of Tuberculosis | Chemical | ClinicalAnnotation, VariantAnnotation | ambiguous | PK | PD | 10751073, 11595069, 11915035, 12668988, 16246623, 16677176, 16770646, 17950035, 18023090, 18330759, 18421452, 18713495, 19686464, 19761367, 20392357, 21047300, 21261721, 21753138, 21856096, 22012226, 22020825, 22162992, 22283902, 22506592, 22788240, 22947533, 23190413, 23277397, 23394127, 23875638, 24188272, 24465778, 24637014, 24888881, 26616266, 27340556, 28799976, 29036176, 29781872 |
| PA164749650 | pyridoxal phosphate | Chemical | VariantAnnotation | not associated | PK | PD | 16770646 |
| PA164779050 | sulfapyridine | Chemical | VariantAnnotation | associated | PK | 18167504, 19560446, 20040334 | |
| PA164784021 | ethambutol | Chemical | ClinicalAnnotation, VariantAnnotation | ambiguous | PK | PD | 10751073, 11595069, 12668988, 17950035, 18330759, 18421452, 19686464, 19891553, 20392357, 21047300, 21261721, 21753138, 21856096, 22020825, 22506592, 22947533, 23190413, 23875638, 24188272, 26911349, 29036176 |
| PA164925725 | Toxic liver disease | Disease | ClinicalAnnotation, VariantAnnotation, VipGene | ambiguous | PK | PD | 10751073, 11915035, 12668988, 16246623, 16677176, 16770646, 17950035, 18023090, 18330759, 18421452, 18713495, 19761367, 20392357, 21047300, 21261721, 21753138, 21856096, 22012226, 22020825, 22162992, 22283902, 22506592, 22788240, 22947533, 23190413, 23277397, 23394127, 23875638, 24188272, 24465778, 24637014, 24888881, 26616266, 26911349, 27340556, 28799976, 29781872 |
| PA165108622 | Drug interaction with drug | Disease | ClinicalAnnotation | associated | PD | ||
| PA165816579 | CYP2D6*4 | Haplotype | LabelAnnotation | associated | |||
| PA165817016 | Kidney Transplantation | Disease | VariantAnnotation | associated | PK | ||
| PA165820585 | 1,7-dimethylxanthine | Chemical | VariantAnnotation | associated | PK | 17011540 | |
| PA165949098 | NAT2*4 | Haplotype | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166048714 | Pregnancy | Disease | VariantAnnotation | not associated | PD | 11755554 | |
| PA166119041 | drug-induced liver injury | Disease | VariantAnnotation, VipGene | ambiguous | PK | PD | 24888881, 29781872 |
| PA166119201 | daniquidone | Chemical | Literature, MultilinkAnnotation | associated | 23912694 | ||
| PA166120927 | drug reaction with eosinophilia and systemic symptoms | Disease | VariantAnnotation | not associated | PD | 29458119 | |
| PA166120929 | severe cutaneous adverse reactions | Disease | VariantAnnotation | associated | PD | 11191886 | |
| PA166128206 | dipyrone | Chemical | ClinicalAnnotation, VariantAnnotation | associated | PK | PD | 25241292, 26111152 |
| PA166157267 | rs776746 | Variant | LabelAnnotation | associated | |||
| PA166157540 | rs1208 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157549 | rs1041983 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157560 | rs1495741 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157563 | rs1799929 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157564 | rs1799930 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157565 | rs1799931 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157570 | rs1801280 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157597 | rs4271002 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166157603 | rs4646244 | Variant | VipGene | associated | 15558239, 18330759, 18421452, 19356010, 19686464, 19891553, 20038957, 20392357, 20602614, 21479500, 21856096, 22506592, 22676187, 23407048, 23875638, 7273597, 11595069 | ||
| PA166165147 | solithromycin | Chemical | VariantAnnotation | associated | PK | ||
| PA166176017 | hydralazine / isosorbide dinitrate | Chemical | LabelAnnotation | associated | |||
| PA166176022 | isoniazid / pyrazinamide / rifampin | Chemical | LabelAnnotation | associated | |||
| PA166182002 | amifampridine phosphate | Chemical | LabelAnnotation | associated | |||
| PA166185150 | amifampridine | Chemical | LabelAnnotation | associated | |||
| PA443227 | Abortion, Spontaneous | Disease | VariantAnnotation | not associated | PD | 11755554 | |
| PA443250 | Acquired Immunodeficiency Syndrome | Disease | VariantAnnotation | associated | PD | 11191886 | |
| PA443434 | Arthritis, Rheumatoid | Disease | VariantAnnotation | ambiguous | PD | 18398952, 24394199 | |
| PA443450 | Asthma | Disease | ClinicalAnnotation | associated | PD | 20602614 | |
| PA443512 | Urinary Bladder Neoplasms | Disease | VipGene | associated | |||
| PA443937 | Drug Toxicity | Disease | ClinicalAnnotation, VipGene | associated | PK | PD | 10751073, 11915035, 12668988, 16246623, 16677176, 16770646, 17950035, 18023090, 18330759, 18421452, 18713495, 19761367, 20392357, 21047300, 21261721, 21753138, 21856096, 22012226, 22020825, 22162992, 22283902, 22506592, 22788240, 22947533, 23190413, 23277397, 23394127, 23875638, 24188272, 24465778, 24637014, 24888881, 26616266, 27340556, 29781872 |
| PA444153 | Fatigue | Disease | VariantAnnotation | associated | PK | 30520338 | |
| PA444435 | Hepatitis | Disease | VariantAnnotation | associated | PK | PD | 11915035 |
| PA444442 | Hepatitis, Toxic | Disease | ClinicalAnnotation, VariantAnnotation | ambiguous | PK | PD | 10751073, 11915035, 12668988, 16246623, 16677176, 16770646, 17950035, 18023090, 18330759, 18421452, 18713495, 19761367, 20392357, 21047300, 21261721, 21753138, 21856096, 22012226, 22020825, 22162992, 22283902, 22506592, 22788240, 22947533, 23190413, 23277397, 23394127, 23875638, 24188272, 24465778, 24637014, 24888881, 26616266, 27340556, 29781872 |
| PA444546 | Hypersensitivity | Disease | VariantAnnotation | ambiguous | PD | 12043950, 26111152, 8689813 | |
| PA444552 | Hypertension | Disease | ClinicalAnnotation | associated | PD | 1136859, 21781652, 2231320, 24444407, 318493, 5026380 | |
| PA444626 | Insulin Resistance | Disease | VariantAnnotation | associated | |||
| PA444819 | Lupus erythematosus | Disease | VipGene | associated | |||
| PA444822 | Lupus Erythematosus, Systemic | Disease | VariantAnnotation | associated | PK | 16531626 | |
| PA445204 | Ovarian Neoplasms | Disease | ClinicalAnnotation | associated | PD | 24533712 | |
| PA445253 | Paresthesia | Disease | VariantAnnotation | associated | PK | 30520338 | |
| PA445355 | Pneumonia | Disease | VariantAnnotation | associated | PD | 9923584 | |
| PA445425 | Prostatic Neoplasms | Disease | ClinicalAnnotation | associated | PD | 20038957 | |
| PA445676 | Skin Diseases | Disease | VariantAnnotation | not associated | PD | 29458119 | |
| PA445941 | Tuberculosis | Disease | ClinicalAnnotation, VariantAnnotation | ambiguous | PK | PD | 10751073, 11595069, 11915035, 12668988, 15855489, 16182272, 16246623, 16531626, 16677176, 16770646, 17950035, 18023090, 18330759, 18421452, 18544910, 18713495, 19686464, 19761367, 19891553, 20392357, 21047300, 21261721, 21479500, 21558457, 21753138, 21856096, 22012226, 22020825, 22162992, 22283902, 22506592, 22788240, 22947533, 23099118, 23190413, 23277397, 23394127, 23875638, 24188272, 24383060, 24465778, 24533708, 24637014, 24888881, 26616266, 26911349, 27340556, 28799976, 29036176, 29781872 |
| PA447230 | HIV | Disease | VariantAnnotation | ambiguous | PK | PD | 11186133, 12043950, 8689813 |
| PA448497 | aspirin | Chemical | ClinicalAnnotation | associated | PD | 20602614 | |
| PA448710 | caffeine | Chemical | VariantAnnotation, VipGene | ambiguous | PK | PD | 11755554, 22105431 |
| PA449014 | cisplatin | Chemical | ClinicalAnnotation | associated | PD | 24533712 | |
| PA449050 | clonazepam | Chemical | Pathway, VariantAnnotation, VipGene | associated | PK | 30520338 | |
| PA449165 | cyclophosphamide | Chemical | ClinicalAnnotation | associated | PD | 24533712 | |
| PA449211 | dapsone | Chemical | VariantAnnotation | not associated | PD | 29458119 | |
| PA449383 | docetaxel | Chemical | ClinicalAnnotation | associated | PD | 20038957 | |
| PA449441 | efavirenz | Chemical | Literature, MultilinkAnnotation | associated | 23990572 | ||
| PA449894 | hydralazine | Chemical | ClinicalAnnotation, VariantAnnotation, VipGene | ambiguous | PK | PD | 1136859, 1396201, 17761710, 21781652, 2231320, 24444407, 24702251, 318493, 445966, 5026380, 7284051, 7298112, 7408395, 7438695, 7471604 |
| PA450112 | isoniazid | Chemical | ClinicalAnnotation, LabelAnnotation, Literature, MultilinkAnnotation, VariantAnnotation, VipGene | ambiguous | PK | PD | 10751073, 11595069, 11915035, 12668988, 15855489, 16182272, 16246623, 16531626, 16677176, 16770646, 17950035, 18023090, 18330759, 18421452, 18544910, 18713495, 19686464, 19761367, 19891553, 20392357, 21047300, 21261721, 21479500, 21558457, 21753138, 21856096, 22012226, 22020825, 22162992, 22283902, 22506592, 22788240, 22947533, 23099118, 23190413, 23277397, 23394127, 23875638, 24188272, 24383060, 24465778, 24533708, 24637014, 24798717, 24888881, 26616266, 26911349, 27340556, 29036176, 29781872 |
| PA450947 | phenytoin | Chemical | ClinicalAnnotation | associated | PD | ||
| PA451108 | procainamide | Chemical | LabelAnnotation | associated | |||
| PA451182 | pyrazinamide | Chemical | ClinicalAnnotation, VariantAnnotation | ambiguous | PK | PD | 11595069, 12668988, 16677176, 17950035, 18330759, 18421452, 19686464, 19891553, 20392357, 21047300, 21261721, 21753138, 21856096, 22020825, 22283902, 22506592, 23099118, 23190413, 23394127, 23875638, 24188272, 26911349, 29036176 |
| PA451250 | rifampin | Chemical | ClinicalAnnotation, VariantAnnotation | ambiguous | PK | PD | 10751073, 11595069, 12668988, 16246623, 16677176, 17950035, 18330759, 18421452, 19686464, 19891553, 20392357, 21047300, 21261721, 21753138, 21856096, 22020825, 22283902, 22506592, 22788240, 22947533, 23099118, 23190413, 23394127, 23875638, 24188272, 26911349, 29036176 |
| PA451512 | streptomycin | Chemical | VariantAnnotation | ambiguous | PK | PD | 10751073, 21753138, 22947533 |
| PA451542 | sulfamethazine | Chemical | VariantAnnotation | associated | PK | ||
| PA451544 | sulfamethoxazole | Chemical | VariantAnnotation, VipGene | ambiguous | PK | PD | 11186133, 11191886, 12043950, 8689813, 9923584 |
| PA451547 | sulfasalazine | Chemical | LabelAnnotation, VariantAnnotation, VipGene | ambiguous | PK | PD | 18167504, 18398952, 19560446, 24394199 |
| PA451644 | thalidomide | Chemical | ClinicalAnnotation | associated | PD | 20038957 | |
| PA451788 | trimethoprim | Chemical | VariantAnnotation | ambiguous | PD | 11191886, 12043950, 8689813, 9923584 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37728483 | 2024 | Evaluating interactions of polygenic risk scores and NAT2 genotypes with tobacco smoking in bladder cancer risk. | 1 |
| 38131213 | 2024 | Relevance of NAT2 genotype and clinical factors to risk for antituberculosis drug-induced liver injury. | 0 |
| 38287694 | 2024 | Influence of N-acetyltransferase 2 polymorphisms and clinical variables on liver function profile of tuberculosis patients. | 0 |
| 37728483 | 2024 | Evaluating interactions of polygenic risk scores and NAT2 genotypes with tobacco smoking in bladder cancer risk. | 1 |
| 38131213 | 2024 | Relevance of NAT2 genotype and clinical factors to risk for antituberculosis drug-induced liver injury. | 0 |
| 38287694 | 2024 | Influence of N-acetyltransferase 2 polymorphisms and clinical variables on liver function profile of tuberculosis patients. | 0 |
| 36048272 | 2023 | The rs1801280 SNP is associated with non-small cell lung carcinoma by exhibiting a highly deleterious effect on N-acetyltransferase 2. | 2 |
| 36138126 | 2023 | N-acetyltransferase 2 genetic polymorphism modifies genotoxic and oxidative damage from new psychoactive substances. | 2 |
| 36963735 | 2023 | Association of NAT2, GSTT1, and GSTM1 gene polymorphisms withprostate cancer risk in Bangladeshi population. | 0 |
| 37023111 | 2023 | NAT2 global landscape: Genetic diversity and acetylation statuses from a systematic review. | 7 |
| 37306342 | 2023 | N -acetyltransferase 2 haplotype modifies risks for both dyslipidemia and urinary bladder cancer. | 0 |
| 37423373 | 2023 | Induction of glucose production by heterocyclic amines is dependent on N-acetyltransferase 2 genetic polymorphism in cryopreserved human hepatocytes. | 2 |
| 37478088 | 2023 | The impact of IDH and NAT2 gene polymorphisms in acute myeloid leukemia risk and overall survival in an Arab population: A case-control study. | 0 |
| 37595067 | 2023 | Discovery of a novel genetic variant in the N-acetyltransferase2 (NAT2) gene that is associated with bladder cancer risk. | 0 |
| 37715989 | 2023 | A case-control study in NAT2 gene polymorphism studies in patients diagnosed with acute myeloid leukemia. | 1 |
Citation
Jhon D Ruiz ; José AG Agúndez ; Carmen Martínez ; Elena García-Martín
NAT2 (N-acetyltransferase 2 (arylamine N-acetyltransferase))
Atlas Genet Cytogenet Oncol Haematol. 2009-01-01
Online version: http://atlasgeneticsoncology.org/gene/41498/nat2
