NKX2-1 (NK2 homeobox 1)
2010-04-01 Theresia Wilbertz , Sebastian Maier , Sven Perner AffiliationInstitute of Pathology, University Hospital Tubingen, Germany
Identity
DNA/RNA
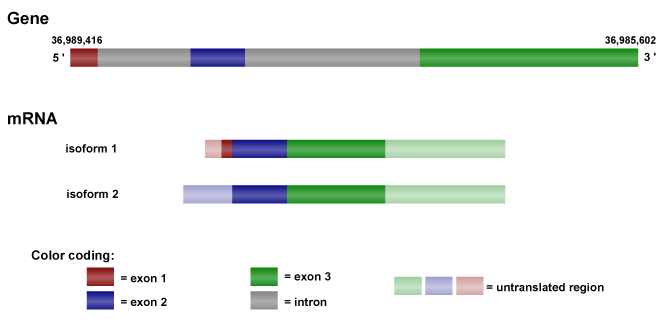
Description
Transcription
Proteins
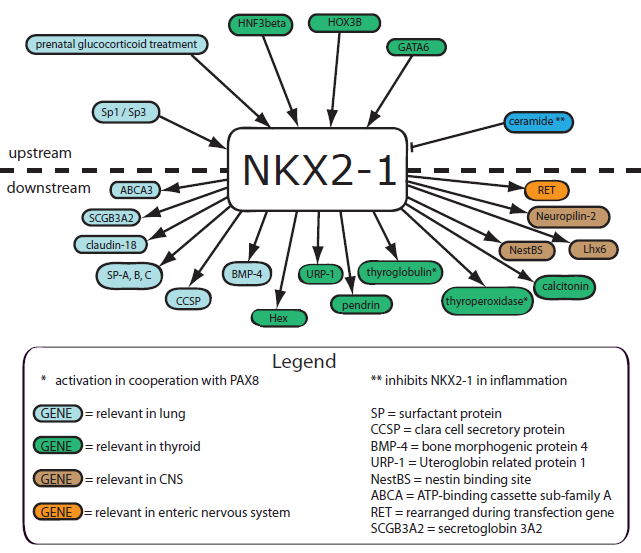
Description
Expression
NKX2-1 expression is also found in thyroid follicular cells and both normal and hyperplastic C cells where it activates calcitonin gene expression.
NKX2-1 is not expressed in adult neurons of the basal ganglia.
During embryonic and fetal development, NKX2-1 expression is found in various tissues (e.g. brain, lung, thyroid), for details see "function" → "Embryonic and fetal development".
Localisation
Function
It cooperates with C/EBPalpha in transactivating CCSP.
In the transcription of SP-C, NKX2-1 interacts with nuclear factor I to differentially regulate the transcription. The longer NKX2-1 isoform reduces transactivation of SP-C, probably due to some kind of interference.
NKX2-1 is a key activator of SP-B gene expression having at least two binding sites at the SP-B promoter and enhancer. The transactivation capacity of NKX2-1 regarding the expression of SP-B is controlled by the sphingolipid ceramide which is produced in inflammation and reduces NKX2-1 binding capacity to the SP-B promoter. SP-B transcription is also inhibited by TGFbeta1-mediated interaction of smad3 with NKX2-1. Moreover, NKX2-1 interacts with retinoic acid receptor (RAR), nuclear receptor coactivators (p160, CBP/p300) and signal transducers and activators of transcription 3 (STAT3) in regulation of SP-B expression.
Furthermore, NKX2-1 regulates the expression of the secretoglobulin 3A2 gene (SCGB3A2) in mouse airways in cooperation with CAATT/enhancer binding proteins alpha and delta as well as the expression of ABCA3 which encodes for a lipid transporter critical for surfactant function at birth and formation of lamellar bodies.
NKX2-1 also plays an important role in the endocrine system: it regulates the expression of the thyroid-specific genes thyroglobulin, thyroid peroxidase, thyrotropin receptor and sodium-iodide-symporter, therefore being crucial for proper thyroid hormone synthesis.
Deletion of NKX2-1 in differentiated neurons of the hypothalamus in mice causes delayed puberty, reduced reproductive capacity and a shorter reproductive span in female mice, suggesting that NKX2-1 plays an important role in juvenile and adult endocrine function.
During embryonic and fetal development, NKX2-1 is active in various organs, especially lung, thyroid and brain.
As a crucial factor for lung development, NKX2-1 is expressed in the ventral foregut endoderm at a very early stage functioning as a signal which is essential for specification of a pulmonary cell fate instead of a liver cell fate. At a later stage, NKX2-1 is critical to the formation of distal pulmonary structures (whereas proximal lung differentiation is NKX2-1-independent), a function in which it is inhibited by TGF-beta.
In addition to that NKX2-1 regulates surfactant protein genes that are important for the development of alveolar stability at birth. It induces SP-A gene expression in fetal lung type II cells through increased binding of NKX2-1 (mediated by cAMP) and the NFkappa-B proteins p50 and p65. Supporting the notion of NKX2-1-dependent SP-expression, lung and associated respiratory dysfunction in neonates caused by SP-B-deficiency are partly induced by down-regulation of NKX2-1. The main therapeutical option, prenatal glucocorticoid treatment, induces the expression of NKX2-1. NKX2-1 regulates expression of uteroglobin-related protein-1 and claudin-18 during lung development.
During thyroid gland organogenesis NKX2-1 is expressed in the ultimobranchial body (UBB) and in the thyroid diverticulum. It is important for the survival of UBB-cells and eventually their dissemination into the thyroid diverticulum and for the formation of the UBB-derived vesicular structure. Pendrin and thyroglobulin are downstream targets of NKX2-1 during thyroid differentiation. The transactivational activity of NKX2-1 during thyroid development can be inhibited by NKX2-5.
In the course of brain development, NKX2-1 expression is found in both telencephalic and diencephalic domains. It cooperates with Gsh2 to pattern the ventral telencephalon. Lack of functional NKX2-1 protein in neurons impairs developmental differentiation and organization of basal ganglia and basal forebrain. NKX2-1 upregulates the transcription of nestin, an intermediate filament protein expressed in multipotent neuroepithelial cells, by direct binding to a HRE-CRE-like site (NestBS) within a CNS-specific enhancer, which indicates that nestin might be at least one of the effectors of NKX2-1 during forebrain development.
NKX2-1 expression occurs in neurons of the arcuate nucleus of the hypothalamus and in glia cells (tanycytes) in neonatal and adult mice, as well as in fetal and adult pituicytes suggesting that NKX2-1 is essential for proper development of the hypothalamus. Lack of NKX2-1 causes aberrant trajectory of the dopaminergic pathway in the developing hypothalamus (mouse-model), development of GABAergic and cholinergic neurons is also impaired in NKX2-1 defective mice. Furthermore, NKX2-1 regulates the specification of oligodendrocytes and controls the postmitotic migration of interneurons originating in the medial ganglionic eminence to either the cortex (downregulation of NKX2-1) or the striatum (maintenance of NKX2-1 expression and thus direct transcriptional activation of neuropilin-2, a guidance receptor in postmitotic cells). By directly activating Lhx6 during embryonic development NKX2-1 plays an essential role for the specification of cortical interneurons which express parvalbumin or somatostatin.
In accordance with the findings concerning the role of NKX2-1 in the development of the above-mentioned organs, NKX2-1-defective mice die at birth due to a characteristic set of malformations and functional impairments: hypoplastic lungs and insufficient surfactant production, defective hypothalamus, absence of thyroid and pituitary gland, delayed development of dopaminergic, GABAergic and cholinergic neurons.
Mutations
Note
| Brain-lung-thyroid syndrome congenital hypothyreoidism, infant respiratory distress syndrome, benign hereditary chorea | |||
| SNP | bp 523 | G → T | premature stop codon at postition 175 |
| SNP | bp 609 | C → A | premature stop codon at position 145 |
| SNP | bp 1320 | C → A | premature stop codon at position 75 |
| SNP | bp 2626 | G → T | missense mutation: valine → phenylalanine at position 14 of DNA-binding-domain |
| SNP | splice acceptor site of intron 2 | A → T | altered mRNA structure => incorrect removal of introns |
| Deletion | 14q11.2-q13.3 | ||
| Insertion | bp 2595 | insertion of GG frameshift mutation: causes truncated protein lacking the entire third helix of the homeodomain | |
| Cancer predisposition can contribute to predisposition for multinodular goiter and papillary thyroid carcinoma. | |||
| SNP | bp 1016 | C → T | missense mutation: A339V |
Germinal
A heterozygous substitution at position 1016 in the coding sequence (C → T) leads to a mutant NKX2-1 protein (A339V) and can contribute to a predisposition for multinodular goiter and papillary thyroid carcinoma.
For other heterozygous NKX2-1 mutations in humans, phenotypes vary widely.
Thyroid dysfunction ranges from mild hypothyrotrophinaemia to severe congenital hypothyroidism due to thyroid hypoplasia or even agenesis. Implication of the lung ranges from a slight increase in pulmonary infections to severe neonatal respiratory distress syndrome.
Homozygous NKX2-1 mutations in humans are probably not viable.
Implicated in
| Consistently expressed | Occasionally expressed | Not expressed | |
| Thyroid | - Papillary carcinoma - Follicular carcinoma - Medullary carcinoma - Hurthle cell carcinoma - Follicular adenoma - Hyperplastic follicular cells | - Undifferentiated thyroid carcinomas | |
| Lung | - Adenocarcinoma - Small cell lung cancer (SCLC) - Pleural effusions of SCLC - Pulmonary sclerosing hemangioma - Bronchioloalveolar carcinoma (except for mucinous parts) - Non-neuroendocrine large-cell carcinoma - Signet-ring cell carcinomas of lung origin | - Pulmonary carcinoids (50%) | - Squamous cell lung cancer - Pleural mesothelioma - Bronchioloalveolar carcinomas (just mucinous parts) - Basaloid carcinoma of the lung |
| Gastrointestinal system | - Small cell cancer of the esophagus | - Colorectal carcinoma | |
| Genitourinary system | - Small cell carcinoma of the urine bladder - Nephroblastoma - Endometrial carcinoma - Endocervical carcinoma | ||
| Thymus | - Thymic carcinoma - Thymoma | ||
| Skin | - Merkel cell carcinoma | ||
| Neuroectodermal | - Ependymoma - Glioblastoma | - Astrocytoma - Oligodendroglioma - Medulloblastoma - Paraganglioma - Ganglioglioma | |
| Neuroendocrine (carcinoid tumorlets, neuroendocrine cell hyperplasia, typical carcinoids, atypical carcinoids, large cell neuroendocrine carcinomas) | - Thyroid origin - Pulmonary origin | - Thymic origin - Gastrointestinal origin - Pancreatic origin - Ovarian origin - Parathyroid adenoma - Pituitary adenoma - Pheochromocytoma | |
| Body cavity fluids | - Lung origin (adenocarcinoma) | - Genitourinary origin - Gastrointestinal origin - Breast origin - Ovarian origin |
Table 2. Expression of NKX2-1 in different tumor entities.
Small cell lung cancer, as well as pulmonary carcinoids and non-neuroendocrine large-cell carcinomas partly exhibit NKX2-1 protein expression.
For a subset of lung adenocarcinomas (especially those which are derived from the terminal respiratory unit) sustained expression of NKX2-1 has been shown to be crucial for the survival of tumor cells. In these tumors RNAi inhibition of NKX2-1 induces proliferation inhibition, growth inhibition and apoptosis (lineage-specific dependency model).
Interestingly, NKX2-1 is also an activator of HOP (Hsp70/Hsp90 Organizing Protein), a potential tumor suppressor gene in lung cancer, and it inhibits EMT (epithelial to mesenchymal transition). NKX2-1 restores epithelial phenotypes in lung adenocarcinomas, acting as an adversary of the EMT-stimulating TGF-beta and a suppressor of tumor progression and invasiveness. TGF-beta inhibits the expression of NKX2-1 and thus lung morphogenesis.
Moreover, NKX2-1 is expressed in most metastatic lung adenocarcinomas.
Concerning parafollicular cells, NKX2-1 expression can be found in normal and hyperplastic c-cells, as well as in medullary thyroid carcinomas. However, the signal intensity is much weaker and less homogenous than observed in tumors originating from follicular thyroid cells.
Non-malignant branchiogenic cysts can easily be confounded with papillary thyroid carcinomas. Since positive immunostaining for NKX2-1 has been found in a subset of these non-malignant cervical cysts, NKX2-1 cannot serve to distinguish between both entities.
A heterozygous germline mutation, which leads to a mutant NKX2-1 protein has been shown to be associated with increased cell proliferation. Consequently, it might contribute to a predisposition for multinodular goiter and papillary thyroid carcinoma (for details see section mutations).
NKX2-1 expression can be found in benign tubal and endometrial epithelia, as well as in benign tumors originating from these tissues. In addition, malignant tumors of the female genital tract, such as endocervical adenocarcinomas, small cell carcinomas of the uterine cervix, endometrioid adenocarcinomas, serous carcinomas, clear cell carcinomas, and uterine malignant mixed Mullerian tumors show positivity for NKX2-1. Staining morphology in these tumors differs from rare positive cells to a diffusely positive staining pattern.
Concerning small cell carcinomas, NKX2-1 expression is not specific for small cell lung cancer, as NKX2-1 expression can also be found in small cell carcinomas originating from the esophagus, prostate, bladder or uterine cervix.
Sellar tumors, including pituicytomas, atypical pituicytomas, granular cell tumors and spindle cell oncocytomas can show positive immunostaining for NKX2-1.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 10757334 | 2000 | Thyroid transcription factor-1 is expressed in extrapulmonary small cell carcinomas but not in other extrapulmonary neuroendocrine tumors. | Agoff SN et al |
| 18682709 | 2008 | Induction of sodium iodide symporter gene and molecular characterisation of HNF3 beta/FoxA2, TTF-1 and C/EBP beta in thyroid carcinoma cells. | Akagi T et al |
| 14633512 | 2004 | Glucocorticoid inhibition of SP-A gene expression in lung type II cells is mediated via the TTF-1-binding element. | Alcorn JL et al |
| 17951193 | 2007 | Large cell and small cell neuroendocrine bladder carcinoma: immunohistochemical and outcome study in a single institution. | Alijo Serrano F et al |
| 15955952 | 2005 | A novel TITF-1 mutation causes benign hereditary chorea with response to levodopa. | Asmus F et al |
| 14645514 | 2003 | Nuclear factor I/thyroid transcription factor 1 interactions modulate surfactant protein C transcription. | Bachurski CJ et al |
| 15614737 | 2004 | TTF-1 and surfactant-B as co-adjuvants in the diagnosis of lung adenocarcinoma and pleural mesothelioma. | Bakir K et al |
| 16980598 | 2006 | Thyroid transcription factor 1--a new prognostic factor in lung cancer: a meta-analysis. | Berghmans T et al |
| 17890326 | 2007 | Sterol response element binding protein and thyroid transcription factor-1 (Nkx2.1) regulate Abca3 gene expression. | Besnard V et al |
| 19011567 | 2009 | TTF-1 expression in nephroblastoma. | Bisceglia M et al |
| 18786356 | 2008 | The requirement of Nkx2-1 in the temporal specification of cortical interneuron subtypes. | Butt SJ et al |
| 19336474 | 2009 | Five new TTF1/NKX2.1 mutations in brain-lung-thyroid syndrome: rescue by PAX8 synergism in one case. | Carré A et al |
| 12161423 | 2002 | Synergistic transactivation of the differentiation-dependent lung gene Clara cell secretory protein (secretoglobin 1a1) by the basic region leucine zipper factor CCAAT/enhancer-binding protein alpha and the homeodomain factor Nkx2.1/thyroid transcription factor-1. | Cassel TN et al |
| 15547625 | 2005 | Linkage and mutational analysis of familial thyroid dysgenesis demonstrate genetic heterogeneity implicating novel genes. | Castanet M et al |
| 17417779 | 2007 | Homeobox gene HOP has a potential tumor suppressive activity in human lung cancer. | Chen Y et al |
| 11175640 | 2001 | Immunostaining for thyroid transcription factor 1 and cytokeratin 20 aids the distinction of small cell carcinoma from Merkel cell carcinoma, but not pulmonary from extrapulmonary small cell carcinomas. | Cheuk W et al |
| 16224162 | 2005 | Immunoexpression of HBME-1, high molecular weight cytokeratin, cytokeratin 19, thyroid transcription factor-1, and E-cadherin in thyroid carcinomas. | Choi YL et al |
| 18413286 | 2008 | Expression of thyroid transcription factor-1 in brain metastases: a useful indicator of pulmonary origin. | Chuang WY et al |
| 12930780 | 2003 | Combinatorial function of the homeodomain proteins Nkx2.1 and Gsh2 in ventral telencephalic patterning. | Corbin JG et al |
| 16260629 | 2005 | Pendrin is a novel in vivo downstream target gene of the TTF-1/Nkx-2.1 homeodomain transcription factor in differentiated thyroid cells. | Dentice M et al |
| 15257545 | 2004 | TTF-1 expression is specific for lung primary in typical and atypical carcinoids: TTF-1-positive carcinoids are predominantly in peripheral location. | Du EZ et al |
| 18339674 | 2008 | NKX2.1 specifies cortical interneuron fate by activating Lhx6. | Du T et al |
| 19262164 | 2009 | Transcription factors make a turn into migration. | Fazzari P et al |
| 18788921 | 2008 | A novel NKX2.1 mutation in a family with hypothyroidism and benign hereditary chorea. | Ferrara AM et al |
| 17562073 | 2007 | TTF-1 staining in glioblastoma multiforme. | Galloway M et al |
| 12378596 | 2002 | Thyroid transcription factor 1: a marker for lung adenoarinoma in body cavity fluids. | Gomez-Fernandez C et al |
| 15718293 | 2005 | Thyroid transcription factor 1 rescues PAX8/p300 synergism impaired by a natural PAX8 paired domain mutation with dominant negative activity. | Grasberger H et al |
| 15554885 | 2004 | Molecular genetic defects in congenital hypothyroidism. | Grüters A et al |
| 19581346 | 2009 | Role of NKX2-1 in N-bis(2-hydroxypropyl)-nitrosamine-induced thyroid adenoma in mice. | Hoshi S et al |
| 19279207 | 2009 | Characterizing the developmental pathways TTF-1, NKX2-8, and PAX9 in lung cancer. | Hsu DS et al |
| 11777274 | 2001 | Utility of thyroid transcription factor-1 and cytokeratin 7 and 20 immunostaining in the identification of origin in malignant effusions. | Jang KY et al |
| 16084939 | 2005 | Thyroid transcription factor 1 expression in small cell carcinoma of the urinary bladder: an immunohistochemical profile of 44 cases. | Jones TD et al |
| 15264213 | 2004 | Nkx2.1 transcription factor in lung cells and a transforming growth factor-beta1 heterozygous mouse model of lung carcinogenesis. | Kang Y et al |
| 10824930 | 2000 | Thyroid transcription factor-1 in normal, hyperplastic, and neoplastic follicular thyroid cells examined by immunohistochemistry and nonradioactive in situ hybridization. | Katoh R et al |
| 10746684 | 2000 | Expression of thyroid transcription factor-1 (TTF-1) in human C cells and medullary thyroid carcinomas. | Katoh R et al |
| 12821380 | 2003 | Aberrant trajectory of ascending dopaminergic pathway in mice lacking Nkx2.1. | Kawano H et al |
| 17925434 | 2007 | Oncogenic cooperation and coamplification of developmental transcription factor genes in lung cancer. | Kendall J et al |
| 8557195 | 1996 | The T/ebp null mouse: thyroid-specific enhancer-binding protein is essential for the organogenesis of the thyroid, lung, ventral forebrain, and pituitary. | Kimura S et al |
| 12891678 | 2003 | Benign hereditary chorea: clinical, genetic, and pathological findings. | Kleiner-Fisman G et al |
| 16960125 | 2007 | Thyroid transcription factor in differentiating type II cells: regulation, isoforms, and target genes. | Kolla V et al |
| 19506552 | 2009 | Epigenetic silencing of TTF-1/NKX2-1 through DNA hypermethylation and histone H3 modulation in thyroid carcinomas. | Kondo T et al |
| 15947946 | 2005 | Thyroid transcription factor 1 expression in cystic lesions of the neck: an immunohistochemical investigation of thyroglossal duct cysts, branchial cleft cysts and metastatic papillary thyroid cancer. | Kreft A et al |
| 11854319 | 2002 | Choreoathetosis, hypothyroidism, and pulmonary alterations due to human NKX2-1 haploinsufficiency. | Krude H et al |
| 18246044 | 2008 | Thyroid transcription factor-1 expression in ovarian epithelial neoplasms. | Kubba LA et al |
| 16355925 | 2005 | Growth factors in lung development. | Kumar VH et al |
| 16601074 | 2006 | Thyroid-specific enhancer-binding protein/NKX2.1 is required for the maintenance of ordered architecture and function of the differentiated thyroid. | Kusakabe T et al |
| 18212743 | 2008 | Genomic profiling identifies TITF1 as a lineage-specific oncogene amplified in lung cancer. | Kwei KA et al |
| 12011259 | 2002 | Expression of thyroid transcription factor-1, cytokeratin 7, and cytokeratin 20 in bronchioloalveolar carcinomas: an immunohistochemical evaluation of 67 cases. | Lau SK et al |
| 19525896 | 2009 | Thyroid transcription factor 1 expression in sellar tumors: a histogenetic marker? | Lee EB et al |
| 10753648 | 2000 | Two functionally distinct forms of NKX2.1 protein are expressed in the pulmonary epithelium. | Li C et al |
| 10684967 | 2000 | A novel DNA element mediates transcription of Nkx2.1 by Sp1 and Sp3 in pulmonary epithelial cells. | Li C et al |
| 12408771 | 2002 | Thyroid transcription factor-1 in the histogenesis of plumonary sclerosing hemangioma. | Lin D et al |
| 18060877 | 2008 | Thyroglobulin and human thyroid cancer. | Lin JD et al |
| 18091383 | 2007 | Diagnostic value of CDX-2 and TTF-1 expressions in separating metastatic neuroendocrine neoplasms of unknown origin. | Lin X et al |
| 10830305 | 2000 | Down-regulation of thyroid transcription factor-1 gene expression in fetal lung hypoplasia is restored by glucocorticoids. | Losada A et al |
| 18067638 | 2008 | Immunohistochemical expression of TTF-1 in various cytological subtypes of primary lung adenocarcinoma, with special reference to intratumoral heterogeneity. | Maeshima AM et al |
| 15979605 | 2005 | Molecular interactions coordinating the development of the forebrain and face. | Marcucio RS et al |
| 17182767 | 2006 | Deletion of the Ttf1 gene in differentiated neurons disrupts female reproduction without impairing basal ganglia function. | Mastronardi C et al |
| 11717541 | 2001 | Primary signet-ring cell carcinoma of lung: immunohistochemical study and comparison with non-pulmonary signet-ring cell carcinomas. | Merchant SH et al |
| 11923479 | 2002 | The synergistic activity of thyroid transcription factor 1 and Pax 8 relies on the promoter/enhancer interplay. | Miccadei S et al |
| 15188024 | 2004 | The role of TTF-1 in differentiating primary and metastatic lung adenocarcinomas. | Moldvay J et al |
| 15459176 | 2004 | Pituitary hypoplasia and respiratory distress syndrome in Prop1 knockout mice. | Nasonkin IO et al |
| 19176457 | 2009 | A germline mutation (A339V) in thyroid transcription factor-1 (TITF-1/NKX2.1) in patients with multinodular goiter and papillary thyroid carcinoma. | Ngan ES et al |
| 11682631 | 2001 | UGRP1, a uteroglobin/Clara cell secretory protein-related protein, is a novel lung-enriched downstream target gene for the T/EBP/NKX2.1 homeodomain transcription factor. | Niimi T et al |
| 11585919 | 2001 | claudin-18, a novel downstream target gene for the T/EBP/NKX2.1 homeodomain transcription factor, encodes lung- and stomach-specific isoforms through alternative splicing. | Niimi T et al |
| 18786357 | 2008 | Postmitotic Nkx2-1 controls the migration of telencephalic interneurons by direct repression of guidance receptors. | Nóbrega-Pereira S et al |
| 11395561 | 2001 | Thyroid transcription factor-1 distinguishes metastatic pulmonary from well-differentiated neuroendocrine tumors of other sites. | Oliveira AM et al |
| 14652817 | 2003 | Expression of calretinin and other mesothelioma-related markers in thymic carcinoma and thymoma. | Pan CC et al |
| 18033672 | 2008 | TTF-1/NKX2.1 up-regulates the in vivo transcription of nestin. | Pelizzoli R et al |
| 18932182 | 2009 | TTF1 expression in non-small cell lung carcinoma: association with TTF1 gene amplification and improved survival. | Perner S et al |
| 11121867 | 2001 | Combined analysis of MIB-1 and thyroid transcription factor-1 predicts survival in non-small cell lung carcinomas. | Puglisi F et al |
| 12655000 | 2003 | Thyroid-specific transcription factors control Hex promoter activity. | Puppin C et al |
| 14745718 | 2004 | Prognostic significance of thyroid transcription factor-1 expression in both early-stage conventional adenocarcinoma and bronchioloalveolar carcinoma of the lung. | Saad RS et al |
| 19293183 | 2009 | Thyroid transcription factor-1 inhibits transforming growth factor-beta-mediated epithelial-to-mesenchymal transition in lung adenocarcinoma cells. | Saito RA et al |
| 15716236 | 2005 | Usefulness of CDX2 and TTF-1 in differentiating gastrointestinal from pulmonary carcinoids. | Saqi A et al |
| 15576401 | 2005 | Different thresholds of fibroblast growth factors pattern the ventral foregut into liver and lung. | Serls AE et al |
| 18059234 | 2007 | Thyroid transcription factor-1 expression in endometrial and endocervical adenocarcinomas. | Siami K et al |
| 16183668 | 2006 | Ceramide decreases surfactant protein B gene expression via downregulation of TTF-1 DNA binding activity. | Sparkman L et al |
| 15485815 | 2004 | Interleukin-10 induces uteroglobin-related protein (UGRP) 1 gene expression in lung epithelial cells through homeodomain transcription factor T/EBP/NKX2.1. | Srisodsai A et al |
| 18373697 | 2009 | Identification of carcinoma origin by thyroid transcription factor-1 immunostaining of fine needle aspirates of metastases. | Strojan Flezar M et al |
| 11567220 | 2001 | Thyroid transcription factor 1 and cytokeratins 1, 5, 10, 14 (34betaE12) expression in basaloid and large-cell neuroendocrine carcinomas of the lung. | Sturm N et al |
| 17412341 | 2007 | Functional analysis of Nkx2.1 and Pax9 for calcitonin gene transcription. | Suzuki M et al |
| 12515971 | 2002 | Immunocytochemical profile of malignant pleural effusions of small-cell lung cancer. | Tamiolakis D et al |
| 12827614 | 2003 | Thyroid transcription factor-1 expression prevalence and its clinical implications in non-small cell lung cancer: a high-throughput tissue microarray and immunohistochemistry study. | Tan D et al |
| 17616654 | 2007 | Lineage-specific dependency of lung adenocarcinomas on the lung development regulator TTF-1. | Tanaka H et al |
| 18632661 | 2008 | CAATT/enhancer-binding proteins alpha and delta interact with NKX2-1 to synergistically activate mouse secretoglobin 3A2 gene expression. | Tomita T et al |
| 16463270 | 2006 | Comparison of the immunophenotypes of signet-ring cell carcinoma, solid adenocarcinoma with mucin production, and mucinous bronchioloalveolar carcinoma of the lung characterized by the presence of cytoplasmic mucin. | Tsuta K et al |
| 17982442 | 2007 | Characterizing the cancer genome in lung adenocarcinoma. | Weir BA et al |
| 9813380 | 1998 | Regulation of surfactant protein gene transcription. | Whitsett JA et al |
| 16138374 | 2005 | Cytology applications of p63 and TTF-1 immunostaining in differential diagnosis of lung cancers. | Wu M et al |
| 18602626 | 2008 | Mechanisms of TGFbeta inhibition of LUNG endodermal morphogenesis: the role of TbetaRII, Smads, Nkx2.1 and Pten. | Xing Y et al |
| 12023581 | 2002 | TTF-1 expression in pulmonary adenocarcinomas. | Yatabe Y et al |
| 19711380 | 2009 | Arcuate nucleus expression of NKX2.1 and DLX and lineages expressing these transcription factors in neuropeptide Y(+), proopiomelanocortin(+), and tyrosine hydroxylase(+) neurons in neonatal and adult mice. | Yee CL et al |
| 10706142 | 2000 | Inhibition of distal lung morphogenesis in Nkx2.1(-/-) embryos. | Yuan B et al |
| 17335582 | 2007 | Primary small cell carcinoma of the esophagus: clinicopathological and immunohistochemical features of 21 cases. | Yun JP et al |
| 15452173 | 2004 | Expression of thyroid transcription factor 1 in primary brain tumours. | Zamecnik J et al |
| 19047914 | 2009 | TTF-1 expression in ovarian and uterine epithelial neoplasia and its potential significance, an immunohistochemical assessment with multiple monoclonal antibodies and different secondary detection systems. | Zhang PJ et al |
| 17164424 | 2007 | GATA and Nkx factors synergistically regulate tissue-specific gene expression and development in vivo. | Zhang Y et al |
| 14960358 | 2004 | NKX2.1 regulates transcription of the gene for human bone morphogenetic protein-4 in lung epithelial cells. | Zhu NL et al |
| 18022953 | 2008 | Comparative functional analysis provides evidence for a crucial role for the homeobox gene Nkx2.1/Titf-1 in forebrain evolution. | van den Akker WM et al |
Other Information
Locus ID:
NCBI: 7080
MIM: 600635
HGNC: 11825
Ensembl: ENSG00000136352
Variants:
dbSNP: 7080
ClinVar: 7080
TCGA: ENSG00000136352
COSMIC: NKX2-1
RNA/Proteins
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37935886 | 2024 | Functional characterization of two novel NKX2-1 frameshift variants that cause pulmonary surfactant dysfunction. | 0 |
| 38677751 | 2024 | Diagnostic Value of Immunostaining for Thyroid Transcription Factor 1 (TTF1) and Paired Box 8 (PAX8) in Distinguishing Pulmonary Metastases of Mesonephric and Mesonephric-like Adenocarcinomas from Primary Lung Adenocarcinomas. | 0 |
| 37935886 | 2024 | Functional characterization of two novel NKX2-1 frameshift variants that cause pulmonary surfactant dysfunction. | 0 |
| 38677751 | 2024 | Diagnostic Value of Immunostaining for Thyroid Transcription Factor 1 (TTF1) and Paired Box 8 (PAX8) in Distinguishing Pulmonary Metastases of Mesonephric and Mesonephric-like Adenocarcinomas from Primary Lung Adenocarcinomas. | 0 |
| 36634573 | 2023 | Expression landscapes in non-small cell lung cancer shaped by the thyroid transcription factor 1. | 4 |
| 36732655 | 2023 | Relationship between brain metastasis and thyroid transcription factor 1. | 0 |
| 36758103 | 2023 | Exploring the values, preferences, and information needs of patients with NKX2-1-related disorders: A qualitative study protocol. | 0 |
| 36870059 | 2023 | Genotype-phenotype mapping of a patient-derived lung cancer organoid biobank identifies NKX2-1-defined Wnt dependency in lung adenocarcinoma. | 7 |
| 36932191 | 2023 | Transcription factor NKX2-1 drives serine and glycine synthesis addiction in cancer. | 5 |
| 36634573 | 2023 | Expression landscapes in non-small cell lung cancer shaped by the thyroid transcription factor 1. | 4 |
| 36732655 | 2023 | Relationship between brain metastasis and thyroid transcription factor 1. | 0 |
| 36758103 | 2023 | Exploring the values, preferences, and information needs of patients with NKX2-1-related disorders: A qualitative study protocol. | 0 |
| 36870059 | 2023 | Genotype-phenotype mapping of a patient-derived lung cancer organoid biobank identifies NKX2-1-defined Wnt dependency in lung adenocarcinoma. | 7 |
| 36932191 | 2023 | Transcription factor NKX2-1 drives serine and glycine synthesis addiction in cancer. | 5 |
| 34524427 | 2022 | SRGN-Triggered Aggressive and Immunosuppressive Phenotype in a Subset of TTF-1-Negative Lung Adenocarcinomas. | 16 |
Citation
Theresia Wilbertz ; Sebastian Maier ; Sven Perner
NKX2-1 (NK2 homeobox 1)
Atlas Genet Cytogenet Oncol Haematol. 2010-04-01
Online version: http://atlasgeneticsoncology.org/gene/44015/nkx2-1
