RPL26 (ribosomal protein L26)
2010-09-01 Kristy Boggs , Michael Kastan AffiliationDepartment of Oncology, St Jude Childrens Research Hospital, Memphis, TN 38105, USA
Identity

DNA/RNA
Note
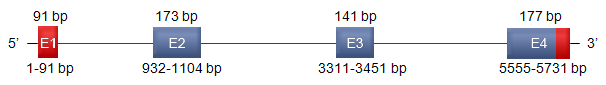
Description
Transcription
It has been reported that ribosomal protein L26 mRNA is preferentially expressed in normal early placenta (NEP) compared to L26 mRNA levels in gestational trophoblastic diseases (GTDs).
Pseudogene
An RPL26 processed pseudogene has been found within the intron of a functional RPS2 gene.
Proteins

Description
Expression
Localisation
Function
Translational regulation of mRNA: protein stability has largely been attributed to increases in p53 protein levels following DNA damage however, more recent reports show an increase in translation of p53 mRNA also contributes to p53 protein levels and is necessary for optimal induction of p53 following irradiation. In particular, the RPL26 protein binds to the 5UTR of p53 mRNA following irradiation enhancing the association of p53 mRNA with polysomes thereby increasing translation of p53 mRNA. Furthermore, RPL26-mediated enhancement of p53 translation not only resulted in an increase in p53 protein levels but also in the transactivation of downstream p53 targets as demonstrated by the induction of G1 cell-cycle arrest and enhanced apoptosis.
More recently, it has been shown that RPL26 binding to p53 mRNA following DNA damage requires not only the 5UTR of p53 mRNA but also the 3UTR. The interaction between complimentary sequences found within the 5 and 3UTR of p53 mRNA creates a double-stranded RNA structure necessary for RPL26 binding to p53 mRNA. Disruption of this double-stranded RNA structure, either by base mutations in the two complementary UTR sequences or with a single-stranded oligonucleotide targeting the 5-3UTR base pairing, abolishes binding of RPL26 to the p53 mRNA, RPL26-enhancement of p53 translation and p53-mediated apoptosis.
Ribosomal proteins are an integral component of protein synthesis however, evidence is emerging that ribosomal proteins have additional functions outside of protein synthesis. Translational regulation of RPL26 mRNA has been shown to be an important feature of LPS-activation of human dendritic cells (DCs). During the early phase of DC maturation via LPS-activation, RPL26 mRNA is translated while during late phase of DC maturation, RPL26 mRNA is translationally down-regulated as demonstrated by a reduction in polysomal-bound RPL26 mRNA and overall RPL26 protein levels. Transcription of the RPL26 gene remained unchanged in all phases of LPS-activated DC maturation.
Interaction with MDM2: regulation of p53 protein levels within the cell has largely been attributed to changes in p53 half-life. In the absence of DNA damage, p53 protein levels are kept low via p53 interaction with MDM2, an E3-ubiquitin ligase that targets p53 for proteasomal degradation. After DNA damage, MDM2-mediated proteasomal degradation of p53 is abrogated allowing p53 protein levels to rapidly accumulate. Recently is has been reported that MDM2 can also regulate p53 protein levels by targeting RPL26-mediated translation of p53 mRNA. In the absence of DNA damage, MDM2 binds to and polyubiquitinates RPL26 targeting the RPL26 protein for proteasomal degradation thereby attenuating RPL26-enhancement of p53 translation. Following DNA damage by irradiation, the inhibitory effect of MDM2 on RPL26-enhancement of p53 translation is abrogated resulting in RPL26-mediated translation of p53 mRNA and accumulation of p53 protein.
In addition to translational regulation of p53 mRNA, it has also been reported that overexpressed RPL26 can regulate p53 protein stability through interaction with MDM2. The interaction of RPL26 with MDM2 stabilizes total p53 protein levels by blocking MDM2-mediated ubiquitination of p53 protein and its subsequent proteasomal degradation.
Homology
HomoloGene: 113207. Gene conserved in Eukaryota (HomoloGene).
Mutations
Note
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|
Other Information
Locus ID:
NCBI: 6154
MIM: 603704
HGNC: 10327
Ensembl: ENSG00000161970
Variants:
dbSNP: 6154
ClinVar: 6154
TCGA: ENSG00000161970
COSMIC: RPL26
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 28288992 | 2017 | Identification of a DNA Damage-Induced Alternative Splicing Pathway That Regulates p53 and Cellular Senescence Markers. | 51 |
| 28288992 | 2017 | Identification of a DNA Damage-Induced Alternative Splicing Pathway That Regulates p53 and Cellular Senescence Markers. | 51 |
| 27825141 | 2016 | p73 expression is regulated by ribosomal protein RPL26 through mRNA translation and protein stability. | 11 |
| 27825141 | 2016 | p73 expression is regulated by ribosomal protein RPL26 through mRNA translation and protein stability. | 11 |
| 26687066 | 2015 | The Six1 oncoprotein downregulates p53 via concomitant regulation of RPL26 and microRNA-27a-3p. | 27 |
| 26687066 | 2015 | The Six1 oncoprotein downregulates p53 via concomitant regulation of RPL26 and microRNA-27a-3p. | 27 |
| 22431104 | 2012 | Frameshift mutation in p53 regulator RPL26 is associated with multiple physical abnormalities and a specific pre-ribosomal RNA processing defect in diamond-blackfan anemia. | 75 |
| 22433872 | 2012 | Interactions of nucleolin and ribosomal protein L26 (RPL26) in translational control of human p53 mRNA. | 62 |
| 22868929 | 2012 | Silencing expression of ribosomal protein L26 and L29 by RNA interfering inhibits proliferation of human pancreatic cancer PANC-1 cells. | 27 |
| 22431104 | 2012 | Frameshift mutation in p53 regulator RPL26 is associated with multiple physical abnormalities and a specific pre-ribosomal RNA processing defect in diamond-blackfan anemia. | 75 |
| 22433872 | 2012 | Interactions of nucleolin and ribosomal protein L26 (RPL26) in translational control of human p53 mRNA. | 62 |
| 22868929 | 2012 | Silencing expression of ribosomal protein L26 and L29 by RNA interfering inhibits proliferation of human pancreatic cancer PANC-1 cells. | 27 |
| 18951086 | 2008 | Mdm2 regulates p53 mRNA translation through inhibitory interactions with ribosomal protein L26. | 130 |
| 18951086 | 2008 | Mdm2 regulates p53 mRNA translation through inhibitory interactions with ribosomal protein L26. | 130 |
Citation
Kristy Boggs ; Michael Kastan
RPL26 (ribosomal protein L26)
Atlas Genet Cytogenet Oncol Haematol. 2010-09-01
Online version: http://atlasgeneticsoncology.org/gene/47470/rpl26
