SNAI1 (snail homolog 1 (Drosophila))
2010-08-01 Joerg Schwock , William R Geddie AffiliationUniversity of Toronto, Department of Laboratory Medicine, Pathobiology, Division of Anatomical Pathology, Toronto General Hospital, 200 Elizabeth Street, Room E11-219, M5G 2C4 Toronto, Ontario, Canada
Identity
HGNC
LOCATION
20q13.13
LOCUSID
ALIAS
SLUGH2,SNA,SNAH,SNAIL,SNAIL1,dJ710H13.1
FUSION GENES
DNA/RNA
Note
Human Snail homolog 1 (SNAI1, SLUGH2, SNA, SNAH, dJ710H13.1), homolog of the Drosophila gene sna, is localized on 20q13.13 (Paznekas et al., 1999; Twigg and Wilkie, 1999). Both publications describe a SNAI1-related pseudogene SNAI1P mapped to chromosome 2q33-37.
Description
SNAI1 has 3 exons (1: 143 bp, 2: 528 bp and 3: 1015 bp size) separated by intron 1-2 (682 bp) and intron 2-3 (3520 bp); spanning an approximately 6 kb region. A CpG island has been described upstream of the coding sequence.
Two silent single nucleotide polymorphisms, a T/C transition at position 783 and a G/A transition at position 1035, have been described (Twigg and Wilkie, 1999).
Two silent single nucleotide polymorphisms, a T/C transition at position 783 and a G/A transition at position 1035, have been described (Twigg and Wilkie, 1999).
Transcription
A single transcript of 1686 bp size gives rise to a protein of 264 aa and approximately 29.1 kDa.
Pseudogene
SNAI1P.
Proteins
Note
Charge 13.0, isoelectric point 8.7563, molecular weight 29082.97 Da (source: Uswest.Ensembl).
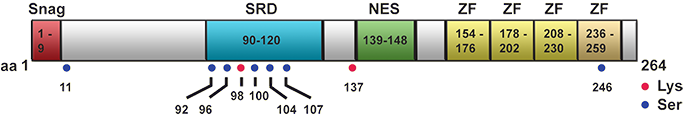
Snail protein structure.
Description
The N-terminal portion (aa 1-150) of the Snail protein contains a SNAG (SNAI1/GFI) domain (aa 1-9) which includes the consensus sequence PRSFLV found in all Snail family members. This motif is highly conserved among species and also found in several other transcription factors where it is associated with repressive functions. A serine-rich domain (SRD: aa 90-120) and a nuclear export sequence (NES: aa 139-148) are involved in the regulation of Snail protein stability and subcellular localization, respectively.
The C-terminal portion (aa 151-264) contains 3 typical (154-176, 178-202, 208-230) and one atypical (236-259) C2H2-type zinc finger (ZF) domains.
The C-terminal portion (aa 151-264) contains 3 typical (154-176, 178-202, 208-230) and one atypical (236-259) C2H2-type zinc finger (ZF) domains.
Expression
Nucleus.
Localisation
The human SNAI1 transcript has been detected in placenta, adult heart and lung. Lower levels were reported for adult brain, liver and skeletal muscle (Paznekas et al., 1999). Several human fetal tissues were reported to express the SNAI1 transcript, with the highest levels detected in kidney (Twigg and Wilkie, 1999).
Reliable high protein expression is present in the extravillous trophoblast of the human placenta which can be utilized as positive control (Rosivatz et al., 2006).
Reliable high protein expression is present in the extravillous trophoblast of the human placenta which can be utilized as positive control (Rosivatz et al., 2006).
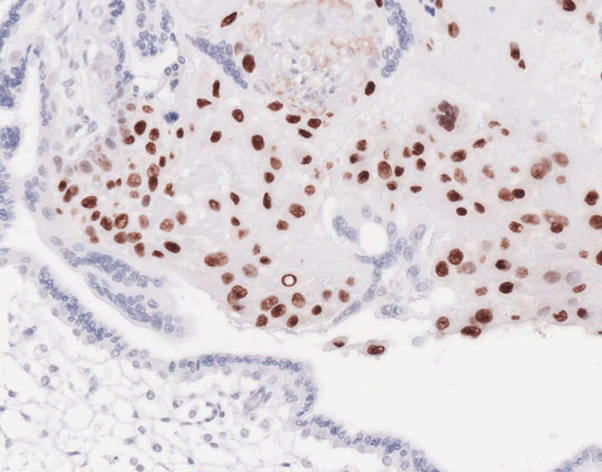
Snail protein expression in 1st trimester human placenta. Method and Antibody: Schwock et al., 2010.
Function
Snail protein (SNAI1) is part of a superfamily of transcription factors composed of the SNAI and the SCRATCH family (Nieto, 2002; Barrallo-Gimeno and Nieto, 2009). The SNAI family contains two more members: Slug (SNAI2 (Cohen et al., 1998)) and Smuc (SNAI3 (Katoh, 2003)) on chromosomes 8 and 16, respectively.
The Snail gene in Drosophila (sna), first identified during analysis of dorso-ventral patterning, is a zinc finger gene with repressor function required for mesoderm formation (Boulay et al., 1987; Leptin, 1991). Isolation of other Snail homologues in different species including the human indicated a high degree of conservation in coding sequence and predicted protein pointing towards a conserved role in early morphogenesis (Paznekas et al., 1999).
Snail protein functions as E-cadherin repressor and is essential during early developmental stages (Cano et al., 2000; LaBonne and Bronner-Fraser, 2000). Snail is re-expressed during adult life in tissue repair as well as in neoplasia, and, in the latter, thought to contribute to the acquisition of a metastatic potential in tumor cells (Batlle et al., 2000). The process by which Snail confers increased motility in individual cells is characterized by a down-regulation of epithelial (cell-cell adhesion, apical-basal polarity) and up-regulation of mesenchymal (cell-extracellular matrix interaction, front-back polarity) features associated with respective changes in molecular composition of the cell which include (among others) cell adhesion molecules and intermediate filaments. This process, termed epithelial-mesenchymal transition (EMT) (Hay, 1989; Thiery, 2002; Peinado et al., 2007), has been classified into 3 types: type 1 in the context of developmental processes, type 2 in inflammation, tissue repair and fibrosis, and type 3 in tumor invasion and metastasis (Kalluri and Weinberg, 2009).
To exert its function as repressor, Snail nuclear import is mediated by importins which recognize a nuclear localization signal that consists of basic residues situated in the zinc finger region (Mingot et al., 2009). Inside the nucleus Snail is required to form ternary complexes with co-repressors via the Snag domain. Different ternary complexes have been described which consist of Ajuba as mediator for the interaction with PRC2 (Herranz et al., 2008), 14-3-3 and PRMT5 (Hou et al., 2010), Sin3A for the interaction with HDAC1/HDAC2 (Peinado et al., 2004), and LSD1 for the interaction with CoREST (Lin et al., 2010). Binding to DNA occurs via E-box elements (5-CACCTG-3) found in the promoter region of different genes including the E-cadherin gene CDH1 (Batlle et al., 2000; Cano et al., 2000).
The regulation of Snail activity mainly involves the central part of the protein which contains most sites for post-translational modification: serine phosphorylation sites (Ser92, 96, 100, 104, 107) in the SRD as well as two lysine oxidation sites (Lys98 and 137), and the NES for Crm1-dependent nuclear export. Two additional serine phosphorylation sites are found N-terminal at Ser11 and C-terminal at Ser246. Phosphorylation of Ser 96, 100, 104 and 107 by GSK3beta is associated with nuclear export, ubiquitination by beta-TrCP1 or FBXL14 and proteasomal degradation (Dominguez et al., 2003; Zhou et al., 2004; Vinas-Castells et al., 2010). Snail is positively regulated by phosphorylation of Ser11, 92 and 246 and its protein stability is increased by interaction with SCP, PKA, CK2, PAK1 and LOXL2 (Peinado et al., 2005; Yang et al., 2005; Wu et al., 2009; MacPherson et al., 2010).
Limited information is available on the factors directly controlling the SNAI1 promoter. Snail up-regulation in cells has been reported as a result of diverse stimuli including cytokines (Interleukin-6), growth factors (TGFbeta, FGF, PDGF, EGF) and activation of their corresponding receptor tyrosine kinases as well as activation of developmental signaling pathways such as Wnt and Hedgehog. Notably, TGFbeta has been described as important EMT trigger leading to HMGA2 and Smad binding at the SNAI1 promoter (Thuault et al., 2008). Another example is Snail expression stimulated by HGF via the MAPK pathway and Egr-1 which also includes a negative feedback mechanism due to Snail binding at the Egr-1 promoter (Grotegut et al., 2006). Also, a conserved 3 enhancer element has been described which interacts with the SNAI1 promoter (Palmer et al., 2007). Furthermore, Snail has been found to control its own expression by binding to an E-box in its own promoter (Peiro et al., 2006). A more detailed overview over the complex signaling pathways regulating Snail expression is given in several recent review publications (Peinado et al., 2007; De Herreros et al., 2010).
Consequences of Snail up-regulation not only include the repression of E-cadherin transcription, but the negative as well as positive control of a series of genes involved in a range of biological functions such as cell-cell adhesion, cell-extracellular matrix interaction, cell polarity, cytoskeleton, cell cycle, survival, and angiogenesis leading to a phenotypic shift towards more mesenchymal cellular characteristics (De Craene et al., 2005; Higashikawa et al., 2008). In the context of tumor-associated EMT these mesenchymal-like characteristics have been correlated with a greater resistance to different therapeutic modalities (Kajita et al., 2004; Kurrey et al., 2009), escape from attack by the immune system (Kudo-Saito et al., 2009), and adoption of a cancer stem cell phenotype (Mani et al., 2008; Morel et al., 2008).
The importance and the exact biological implications of Snail expression in human tumors remain a focus of current research (Schwock et al., 2010). Some of the challenges in this area may be due to the transient and dynamic nature of tumor-associated EMT. Also, it has been proposed that Snail is required as initial trigger in EMT, whereas maintenance of the phenotype is taken over by other factors potentially leaving behind a mesenchymal cell devoid of Snail expression (Peinado et al., 2007). Another intriguing recent observation is the presence of Snail in tumor-associated stroma and its impact on tumor prognosis (Franci et al., 2009).
The Snail gene in Drosophila (sna), first identified during analysis of dorso-ventral patterning, is a zinc finger gene with repressor function required for mesoderm formation (Boulay et al., 1987; Leptin, 1991). Isolation of other Snail homologues in different species including the human indicated a high degree of conservation in coding sequence and predicted protein pointing towards a conserved role in early morphogenesis (Paznekas et al., 1999).
Snail protein functions as E-cadherin repressor and is essential during early developmental stages (Cano et al., 2000; LaBonne and Bronner-Fraser, 2000). Snail is re-expressed during adult life in tissue repair as well as in neoplasia, and, in the latter, thought to contribute to the acquisition of a metastatic potential in tumor cells (Batlle et al., 2000). The process by which Snail confers increased motility in individual cells is characterized by a down-regulation of epithelial (cell-cell adhesion, apical-basal polarity) and up-regulation of mesenchymal (cell-extracellular matrix interaction, front-back polarity) features associated with respective changes in molecular composition of the cell which include (among others) cell adhesion molecules and intermediate filaments. This process, termed epithelial-mesenchymal transition (EMT) (Hay, 1989; Thiery, 2002; Peinado et al., 2007), has been classified into 3 types: type 1 in the context of developmental processes, type 2 in inflammation, tissue repair and fibrosis, and type 3 in tumor invasion and metastasis (Kalluri and Weinberg, 2009).
To exert its function as repressor, Snail nuclear import is mediated by importins which recognize a nuclear localization signal that consists of basic residues situated in the zinc finger region (Mingot et al., 2009). Inside the nucleus Snail is required to form ternary complexes with co-repressors via the Snag domain. Different ternary complexes have been described which consist of Ajuba as mediator for the interaction with PRC2 (Herranz et al., 2008), 14-3-3 and PRMT5 (Hou et al., 2010), Sin3A for the interaction with HDAC1/HDAC2 (Peinado et al., 2004), and LSD1 for the interaction with CoREST (Lin et al., 2010). Binding to DNA occurs via E-box elements (5-CACCTG-3) found in the promoter region of different genes including the E-cadherin gene CDH1 (Batlle et al., 2000; Cano et al., 2000).
The regulation of Snail activity mainly involves the central part of the protein which contains most sites for post-translational modification: serine phosphorylation sites (Ser92, 96, 100, 104, 107) in the SRD as well as two lysine oxidation sites (Lys98 and 137), and the NES for Crm1-dependent nuclear export. Two additional serine phosphorylation sites are found N-terminal at Ser11 and C-terminal at Ser246. Phosphorylation of Ser 96, 100, 104 and 107 by GSK3beta is associated with nuclear export, ubiquitination by beta-TrCP1 or FBXL14 and proteasomal degradation (Dominguez et al., 2003; Zhou et al., 2004; Vinas-Castells et al., 2010). Snail is positively regulated by phosphorylation of Ser11, 92 and 246 and its protein stability is increased by interaction with SCP, PKA, CK2, PAK1 and LOXL2 (Peinado et al., 2005; Yang et al., 2005; Wu et al., 2009; MacPherson et al., 2010).
Limited information is available on the factors directly controlling the SNAI1 promoter. Snail up-regulation in cells has been reported as a result of diverse stimuli including cytokines (Interleukin-6), growth factors (TGFbeta, FGF, PDGF, EGF) and activation of their corresponding receptor tyrosine kinases as well as activation of developmental signaling pathways such as Wnt and Hedgehog. Notably, TGFbeta has been described as important EMT trigger leading to HMGA2 and Smad binding at the SNAI1 promoter (Thuault et al., 2008). Another example is Snail expression stimulated by HGF via the MAPK pathway and Egr-1 which also includes a negative feedback mechanism due to Snail binding at the Egr-1 promoter (Grotegut et al., 2006). Also, a conserved 3 enhancer element has been described which interacts with the SNAI1 promoter (Palmer et al., 2007). Furthermore, Snail has been found to control its own expression by binding to an E-box in its own promoter (Peiro et al., 2006). A more detailed overview over the complex signaling pathways regulating Snail expression is given in several recent review publications (Peinado et al., 2007; De Herreros et al., 2010).
Consequences of Snail up-regulation not only include the repression of E-cadherin transcription, but the negative as well as positive control of a series of genes involved in a range of biological functions such as cell-cell adhesion, cell-extracellular matrix interaction, cell polarity, cytoskeleton, cell cycle, survival, and angiogenesis leading to a phenotypic shift towards more mesenchymal cellular characteristics (De Craene et al., 2005; Higashikawa et al., 2008). In the context of tumor-associated EMT these mesenchymal-like characteristics have been correlated with a greater resistance to different therapeutic modalities (Kajita et al., 2004; Kurrey et al., 2009), escape from attack by the immune system (Kudo-Saito et al., 2009), and adoption of a cancer stem cell phenotype (Mani et al., 2008; Morel et al., 2008).
The importance and the exact biological implications of Snail expression in human tumors remain a focus of current research (Schwock et al., 2010). Some of the challenges in this area may be due to the transient and dynamic nature of tumor-associated EMT. Also, it has been proposed that Snail is required as initial trigger in EMT, whereas maintenance of the phenotype is taken over by other factors potentially leaving behind a mesenchymal cell devoid of Snail expression (Peinado et al., 2007). Another intriguing recent observation is the presence of Snail in tumor-associated stroma and its impact on tumor prognosis (Franci et al., 2009).
Homology
Human Snail protein is 97.3, 87.2, 87.5, 57.3 and 58.4 identical to SNAI1 in chimpanzee (Pan troglodytes), SNAI1 in dog (Canis lupus familiaris), Snai1 in mouse (Mus musculus), snai1a and snai1b in zebrafish (Danio rerio), respectively.
Implicated in
Entity name
Various cancers
Note
Involvement of Snail as a major factor in craniosynostosis was excluded (Paznekas et al., 1999; Twigg and Wilkie, 1999). Expression of SNAI1 at the transcript level has been detected in benign conditions such as tissue fibrosis (Sato et al., 2003; Yanez-Mo et al., 2003; Jayachandran et al., 2009), a range of malignant neoplasms (Cheng et al., 2001; Rosivatz et al., 2002; Takeno et al., 2004), and in normal tissue adjacent to tumor (Pena et al., 2009). Early studies based on detection of the SNAI1 transcript found associations with lymph node metastasis (Cheng et al., 2001; Blanco et al., 2002) and malignant effusion (Elloul et al., 2005) in breast cancer. A mouse model reported by Moody et al. (2005) implicated Snail expression with mammary cancer recurrence. Other studies described associations between elevated SNAI1 transcript levels and hypoxia in ovarian cancer (Imai et al., 2003), downregulation of Vitamin D Receptor in colon cancer (Palmer et al., 2004; Pena et al., 2005), invasion and distant metastasis in oesophageal squamous cell carcinoma (Takeno et al., 2004), invasiveness (Sugimachi et al., 2003) and poor prognosis (Miyoshi et al., 2005) in hepatocellular carcinoma, and spindle cell phenotype in synovial sarcoma (Saito et al., 2004). However, it has been pointed out that transcript levels may not correlate well with Snail protein which is tightly regulated and subject to a short half-life previously reported as approximately 25 minutes (Zhou et al., 2004). Also, transcript levels may be confounded by Snail expression in the stromal tumor component if no micro-dissection is performed (Peinado et al., 2007). Immunohistochemical detection of Snail has been documented for a range of cancers including the upper gastrointestinal tract (Rosivatz et al., 2006; Natsugoe et al., 2007; Usami et al., 2008; Kim et al., 2009), head and neck (Yang et al., 2007; Peinado et al., 2008; Yang et al., 2008; Zidar et al., 2008; Schwock et al., 2010), colorectum (Roy et al., 2005; Franci et al., 2009), neuroendocrine tumors of the ileum (Fendrich et al., 2007), uterine cervix (Franci et al., 2006), endometrium (Blechschmidt et al., 2007), ovary (Blechschmidt et al., 2008; Jin et al., 2009; Tuhkanen et al., 2009), prostate (Heeboll et al., 2009), breast (Zhou et al., 2004), bladder (Bruyere et al., 2009), adrenal gland (Waldmann et al., 2008), thyroid gland (Hardy et al., 2007), parathyroid gland (Fendrich et al., 2009) as well as pheochromocytoma (Waldmann et al., 2009) and sarcomas (Franci et al., 2006). Differences in immunoreactivity seem to depend on the individual tumor entity examined as well as technical issues (Schwock et al., 2010).
Entity name
Neoplasms of the gastro-intestinal tract
Note
Rosivatz et al. (2006) examined Snail expression in adenocarcinomas of the upper gastrointestinal tract. 7.9% (27/340) of their cases were reported with positive staining for Snail. There was no correlation between Snail and E-cadherin expression or Snail and clinicopathological parameters. Natsugoe et al. (2007) examined 194 cases with oesophageal squamous cell carcinoma. 61.7% (84/194) were reported with positive staining. Snail expression was associated with deep invasion, increased lymph node metastasis, and advanced stage. No correlation was found between Snail and E-cadherin expression. Usami et al. (2008) reported a cohort of 72 cases of oesophageal squamous cell carcinoma for which 38% (27/72) were considered positive. Elevated Snail expression was found at the invasion front, and was associated with lymphatic and venous vessel invasion, lymph node metastasis and tumor stage. Furthermore, a recent study by Kim et al. (2009) reported Snail positivity in 42.9% (245/571) of gastric carcinomas where it was associated with invasion and lymph node metastasis. Snail staining was an independent indicator of prognosis by multivariate analysis in this study. Fendrich et al., 2007 examined Snail expression in neuroendocrine tumors of the ileum. 59% (22/37) of the primary tumors and 6 of 7 liver metastases were reported with immunoreactivity for Snail. 53% (16/30) of the neuroendocrine tumors displayed positivity for Snail as well as Sonic Hedgehog. Roy et al. (2005) found a proportion of 78% (46/59) cases with positive staining in their study on colorectal cancers as well as a trend towards increased presence of Snail in tumors with distant metastasis. A more recent study on colorectal cancer by Franci et al. (2009) reported a similarly high proportion of 79% (128/162) of cases with Snail immunoreactivity. Interestingly, in this study a correlation between stromal Snail expression and decreased survival was found.
Entity name
Neoplasms of the head and neck
Note
Yang et al. (2007) reported a proportion of 37.4% (n=147) of primary head and neck squamous cell carcinomas with positive immunoreactivity for Snail. Snail expression was associated with lymph node metastasis, and co-expression with Nijmegen breakage syndrome 1 (NBS1) indicated short metastasis-free period and overall survival. Another study by Yang et al. (2008) reported a positive correlation between Snail and reduced metastasis-free and overall survival. Peinado et al. (2008) examined a large cohort of laryngeal squamous cell carcinomas for which 16% (40/251) were reported Snail positive including 3% (8/251) high-positive. They found a correlation between Snail and LOXL2 expression, but no association between Snail and disease-free or overall survival. Zidar et al. (2008) reported their findings on two cohorts of head and neck squamous cell carcinomas specifically distinguishing between spindle cell carcinomas and those of moderately differentiated phenotype. 19/30 of the spindle cell, but only 4/30 cases in the moderately differentiated group were found to display positive immunoreactivity. There was no correlation between Snail and E-cadherin expression. Schwock et al. (2010) examined a cohort of 46 cases of oral squamous cell carcinoma including corresponding metastases. Nuclear Snail-positivity equal or in excess of a 5% threshold was observed in 10 tumors and 5 metastases which corresponded to 12 cases. Individual Snail-positive tumor cells below this threshold, however, were present more frequently and found in primary tumors of 30 patients. Snail expression in tumor cells in excess of 10% was rare, but associated with poor outcome by univariate analysis.
Entity name
Neoplasms of the genitourinary tract
Note
87 primary endometrioid-type adenocarcinomas of the endometrium and 26 unrelated metastases were examined in a study by Blechschmidt et al. (2007). Among the primary tumors and the metastases a proportion of 28.7% and 53.8% were reported with positive Snail staining, respectively. Snail immunoreactivity in metastases was found to correlate with higher grade and reduced E-cadherin expression. A subsequent study by Blechschmidt et al. (2008) on 48 primary ovarian neoplasms and 50 metastases found Snail immunoreactivity in 37.5% and 52%, respectively. A borderline significant difference in overall survival with Snail expression in metastases was noted. There was no correlation between Snail and E-cadherin expression in this study. A similar study by Jin et al. (2009) examined 41 serous adenocarcinomas of the ovary with 14 matched metastases, 12 serous borderline tumors, 5 cystadenomas and 4 normal ovarian controls. There was a range of Snail immunoreactivity with increased nuclear positivity noted in the carcinoma group. Tuhkanen et al. (2009) compared 74 ovarian carcinomas with 24 borderline tumors, 21 benign ovarian neoplasms and 14 normal controls. Increased nuclear staining was noted with increasing malignancy both in the epithelial as well as the stromal compartment. 23% (17/74) of the ovarian carcinomas were reported to show focal Snail positivity. No association with clinicopathological factors was seen in this study. Heeboll et al., 2009 examined Snail in 327 prostate cancer specimens, 15 specimens with high-grade prostatic intraepithelial neoplasia (PIN), 30 specimens from patients with benign prostatic hyperplasia and 30 benign prostate tissue controls. Approximately 50% of the prostate cancers were found to have high Snail immunoreactivity compared to only 7% of the high-grade PIN specimens. Snail expression in this study was associated with Gleason score, but not with progression or prognosis. Bruyere et al., 2009 studied Snail expression in transitional cell carcinoma of the bladder using a microarray of 87 cases. Strong Snail positivity was found in 43.7% and weak positivity in the remainder of the cases. Snail immunoreactivity in this study was prognostic for tumor recurrence by uni- and multivariate analysis.
Entity name
Breast cancer
Note
Zhou et al. (2004) reported a study on Snail expression in breast cancer which found positive staining in 56% (72/129) of their cases; 17 with low and 55 with high Snail immunoreactivity. Snail correlated with GSK-3beta inhibition and E-cadherin downregulation, and clinically with metastasis in this study.
Entity name
Endocrine neoplasms
Note
Waldmann et al. (2008) reported their findings with Snail expression in adrenocortical carcinomas obtained in a study including 26 primary tumors as well as two lymph node and one liver metastases. 65% (17/26) primary tumors showed staining for Snail with strong positivity found at the invasion front of 7 tumors and in 2 of 3 metastases. Snail positivity was associated with advanced stage, decreased survival and higher risk for distant metastases. The same group reported a study on Snail in pheochromocytomas including 44 primary tumors, 3 lymph node and 2 peritoneal metastases (Waldmann et al., 2009). Snail positivity was reported for 28% (13/47) cases, and positive staining was associated with malignant behaviour. Hardy et al. (2007) published their results focused on thyroid neoplasms. 18/31 follicular and 28/32 papillary thyroid cancers as well as all of 4 lymph node metastases of papillary thyroid cancer were found to stain positive for Snail whereas normal thyroid tissue was negative. Snail staining was reported to be restricted to the invasive front and associated with a concomitant reduction in E-cadherin reactivity. The authors of this study also included their findings from a Combi-TA mouse model showing development of papillary thyroid carcinomas. Fendrich et al. (2009) recently published results of a study focused on parathyroid neoplasms including 9 cases of parathyroid carcinoma, 25 adenomas and 25 cases of hyperplasia. Snail staining was positive in all cases of hyperplasia and 22/25 adenomas. In carcinomas a change in staining pattern towards the invasion front was noted.
Entity name
Mesenchymal neoplasms
Note
Franci et al. (2006) studied Snail in a series of different neoplasm which included sarcomas and infantile fibromatosis as well as epithelial neoplasms (squamous cell carcinoma of the uterine cervix and adenocarcinoma of the colon). High Snail expression was present in fibrosarcomas and other sarcomas. Snail expression in neoplasms of epithelial origin was restricted to the tumor-stroma interface.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 19427053 | 2009 | Evolutionary history of the Snail/Scratch superfamily. | Barrallo-Gimeno A et al |
| 10655587 | 2000 | The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. | Batlle E et al |
| 12082640 | 2002 | Correlation of Snail expression with histological grade and lymph node status in breast carcinomas. | Blanco MJ et al |
| 18026186 | 2008 | The E-cadherin repressor Snail is associated with lower overall survival of ovarian cancer patients. | Blechschmidt K et al |
| 3683556 | 1987 | The Drosophila developmental gene snail encodes a protein with nucleic acid binding fingers. | Boulay JL et al |
| 19162513 | 2010 | Snail expression is an independent predictor of tumor recurrence in superficial bladder cancers. | Bruyere F et al |
| 10655586 | 2000 | The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. | Cano A et al |
| 11439345 | 2001 | Mechanisms of inactivation of E-cadherin in breast carcinoma: modification of the two-hit hypothesis of tumor suppressor gene. | Cheng CW et al |
| 9721220 | 1998 | Human SLUG gene organization, expression, and chromosome map location on 8q. | Cohen ME et al |
| 16024625 | 2005 | The transcription factor snail induces tumor cell invasion through modulation of the epithelial cell differentiation program. | De Craene B et al |
| 12832491 | 2003 | Phosphorylation regulates the subcellular location and activity of the snail transcriptional repressor. | Domínguez D et al |
| 15742334 | 2005 | Snail, Slug, and Smad-interacting protein 1 as novel parameters of disease aggressiveness in metastatic ovarian and breast carcinoma. | Elloul S et al |
| 19176646 | 2009 | Unique expression pattern of the EMT markers Snail, Twist and E-cadherin in benign and malignant parathyroid neoplasia. | Fendrich V et al |
| 19440385 | 2009 | Snail1 protein in the stroma as a new putative prognosis marker for colon tumours. | Francí C et al |
| 16568079 | 2006 | Expression of Snail protein in tumor-stroma interface. | Francí C et al |
| 16858414 | 2006 | Hepatocyte growth factor induces cell scattering through MAPK/Egr-1-mediated upregulation of Snail. | Grotegut S et al |
| 17724139 | 2007 | Snail family transcription factors are implicated in thyroid carcinogenesis. | Hardy RG et al |
| 2696597 | 1989 | Theory for epithelial-mesenchymal transformation based on the "fixed cortex" cell motility model. | Hay ED et al |
| 19245592 | 2009 | Snail1 is over-expressed in prostate cancer. | Heebøll S et al |
| 18519590 | 2008 | Polycomb complex 2 is required for E-cadherin repression by the Snail1 transcription factor. | Herranz N et al |
| 18329791 | 2008 | Gene expression profiling to identify genes associated with high-invasiveness in human squamous cell carcinoma with epithelial-to-mesenchymal transition. | Higashikawa K et al |
| 20501852 | 2010 | 14-3-3 binding sites in the snail protein are essential for snail-mediated transcriptional repression and epithelial-mesenchymal differentiation. | Hou Z et al |
| 14507651 | 2003 | Hypoxia attenuates the expression of E-cadherin via up-regulation of SNAIL in ovarian carcinoma cells. | Imai T et al |
| 19850962 | 2009 | SNAI transcription factors mediate epithelial-mesenchymal transition in lung fibrosis. | Jayachandran A et al |
| 19795442 | 2010 | Snail is critical for tumor growth and metastasis of ovarian carcinoma. | Jin H et al |
| 15314165 | 2004 | Aberrant expression of the transcription factors snail and slug alters the response to genotoxic stress. | Kajita M et al |
| 19487818 | 2009 | The basics of epithelial-mesenchymal transition. | Kalluri R et al |
| 12579345 | 2003 | Identification and characterization of human SNAIL3 (SNAI3) gene in silico. | Katoh M et al |
| 19309396 | 2009 | Prognostic importance of epithelial-mesenchymal transition-related protein expression in gastric carcinoma. | Kim MA et al |
| 19249678 | 2009 | Cancer metastasis is accelerated through immunosuppression during Snail-induced EMT of cancer cells. | Kudo-Saito C et al |
| 19544473 | 2009 | Snail and slug mediate radioresistance and chemoresistance by antagonizing p53-mediated apoptosis and acquiring a stem-like phenotype in ovarian cancer cells. | Kurrey NK et al |
| 10772801 | 2000 | Snail-related transcriptional repressors are required in Xenopus for both the induction of the neural crest and its subsequent migration. | LaBonne C et al |
| 1884999 | 1991 | twist and snail as positive and negative regulators during Drosophila mesoderm development. | Leptin M et al |
| 20389281 | 2010 | The SNAG domain of Snail1 functions as a molecular hook for recruiting lysine-specific demethylase 1. | Lin Y et al |
| 19923321 | 2010 | Phosphorylation of serine 11 and serine 92 as new positive regulators of human Snail1 function: potential involvement of casein kinase-2 and the cAMP-activated kinase protein kinase A. | MacPherson MR et al |
| 18485877 | 2008 | The epithelial-mesenchymal transition generates cells with properties of stem cells. | Mani SA et al |
| 19386897 | 2009 | Characterization of Snail nuclear import pathways as representatives of C2H2 zinc finger transcription factors. | Mingot JM et al |
| 15668718 | 2005 | Snail accelerates cancer invasion by upregulating MMP expression and is associated with poor prognosis of hepatocellular carcinoma. | Miyoshi A et al |
| 16169465 | 2005 | The transcriptional repressor Snail promotes mammary tumor recurrence. | Moody SE et al |
| 18682804 | 2008 | Generation of breast cancer stem cells through epithelial-mesenchymal transition. | Morel AP et al |
| 17273727 | 2007 | Snail plays a key role in E-cadherin-preserved esophageal squamous cell carcinoma. | Natsugoe S et al |
| 11994736 | 2002 | The snail superfamily of zinc-finger transcription factors. | Nieto MA et al |
| 15322538 | 2004 | The transcription factor SNAIL represses vitamin D receptor expression and responsiveness in human colon cancer. | Pálmer HG et al |
| 17616667 | 2007 | A 3' enhancer controls snail expression in melanoma cells. | Palmer MB et al |
| 10585766 | 1999 | Genomic organization, expression, and chromosome location of the human SNAIL gene (SNAI1) and a related processed pseudogene (SNAI1P). | Paznekas WA et al |
| 14673164 | 2004 | Snail mediates E-cadherin repression by the recruitment of the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. | Peinado H et al |
| 16096638 | 2005 | A molecular role for lysyl oxidase-like 2 enzyme in snail regulation and tumor progression. | Peinado H et al |
| 18559498 | 2008 | Lysyl oxidase-like 2 as a new poor prognosis marker of squamous cell carcinomas. | Peinado H et al |
| 17508028 | 2007 | Snail, Zeb and bHLH factors in tumour progression: an alliance against the epithelial phenotype? | Peinado H et al |
| 16617148 | 2006 | Snail1 transcriptional repressor binds to its own promoter and controls its expression. | Peiró S et al |
| 19802011 | 2009 | SNAI1 expression in colon cancer related with CDH1 and VDR downregulation in normal adjacent tissue. | Peña C et al |
| 16207734 | 2005 | Cancer development induced by graded expression of Snail in mice. | Pérez-Mancera PA et al |
| 16328348 | 2006 | Expression and nuclear localization of Snail, an E-cadherin repressor, in adenocarcinomas of the upper gastrointestinal tract. | Rosivatz E et al |
| 15712635 | 2005 | The transcriptional repressor SNAIL is overexpressed in human colon cancer. | Roy HK et al |
| 15467754 | 2004 | E-cadherin mutation and Snail overexpression as alternative mechanisms of E-cadherin inactivation in synovial sarcoma. | Saito T et al |
| 14617750 | 2003 | Targeted disruption of TGF-beta1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. | Sato M et al |
| 20181105 | 2010 | SNAI1 expression and the mesenchymal phenotype: an immunohistochemical study performed on 46 cases of oral squamous cell carcinoma. | Schwock J et al |
| 12855644 | 2003 | Transcriptional repressor snail and progression of human hepatocellular carcinoma. | Sugimachi K et al |
| 15272533 | 2004 | E-cadherin expression in patients with esophageal squamous cell carcinoma: promoter hypermethylation, Snail overexpression, and clinicopathologic implications. | Takeno S et al |
| 12189386 | 2002 | Epithelial-mesenchymal transitions in tumour progression. | Thiery JP et al |
| 18832382 | 2008 | HMGA2 and Smads co-regulate SNAIL1 expression during induction of epithelial-to-mesenchymal transition. | Thuault S et al |
| 19695091 | 2009 | Nuclear expression of Snail1 in borderline and malignant epithelial ovarian tumours is associated with tumour progression. | Tuhkanen H et al |
| 10543399 | 1999 | Characterisation of the human snail (SNAI1) gene and exclusion as a major disease gene in craniosynostosis. | Twigg SR et al |
| 18491351 | 2008 | Snail-associated epithelial-mesenchymal transition promotes oesophageal squamous cell carcinoma motility and progression. | Usami Y et al |
| 19955572 | 2010 | The hypoxia-controlled FBXL14 ubiquitin ligase targets SNAIL1 for proteasome degradation. | Viñas-Castells R et al |
| 19412634 | 2009 | Expression of the transcription factor snail and its target gene twist are associated with malignancy in pheochromocytomas. | Waldmann J et al |
| 19004823 | 2009 | Small C-terminal domain phosphatase enhances snail activity through dephosphorylation. | Wu Y et al |
| 12556543 | 2003 | Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. | Yáñez-Mó M et al |
| 16936774 | 2007 | Overexpression of NBS1 induces epithelial-mesenchymal transition and co-expression of NBS1 and Snail predicts metastasis of head and neck cancer. | Yang MH et al |
| 18297062 | 2008 | Direct regulation of TWIST by HIF-1alpha promotes metastasis. | Yang MH et al |
| 15833848 | 2005 | Pak1 phosphorylation of snail, a master regulator of epithelial-to-mesenchyme transition, modulates snail's subcellular localization and functions. | Yang Z et al |
| 15448698 | 2004 | Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial-mesenchymal transition. | Zhou BP et al |
| 18712413 | 2008 | Cadherin-catenin complex and transcription factor Snail-1 in spindle cell carcinoma of the head and neck. | Zidar N et al |
| 20455012 | 2010 | Snail family regulation and epithelial mesenchymal transitions in breast cancer progression. | de Herreros AG et al |
Other Information
Locus ID:
NCBI: 6615
MIM: 604238
HGNC: 11128
Ensembl: ENSG00000124216
Variants:
dbSNP: 6615
ClinVar: 6615
TCGA: ENSG00000124216
COSMIC: SNAI1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000124216 | ENST00000244050 | O95863 |
Expression (GTEx)
Pathways
| Pathway | Source | External ID |
|---|---|---|
| Adherens junction | KEGG | ko04520 |
| Adherens junction | KEGG | hsa04520 |
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37611675 | 2024 | Protein kinase D activity is a risk biomarker in prostate cancer that drives cell invasion by a Snail/ERK dependent mechanism. | 0 |
| 38043804 | 2024 | Dual role of Snail1 as transcriptional repressor and activator. | 1 |
| 38141763 | 2024 | The Snail signaling branch downstream of the TGF-β/Smad3 pathway mediates Rho activation and subsequent stress fiber formation. | 3 |
| 38195659 | 2024 | Decidual stromal cells-derived exosomes incurred insufficient migration and invasion of trophoblast by disturbing of β-TrCP-mediated snail ubiquitination and degradation in unexplained recurrent spontaneous abortion. | 1 |
| 38217587 | 2024 | KLF5 inhibits the migration and invasion in cervical cancer cell lines by regulating SNAI1. | 0 |
| 38250150 | 2024 | Ubiquitin-specific Protease 35 Promotes Gastric Cancer Metastasis by Increasing the Stability of Snail1. | 1 |
| 38281235 | 2024 | Co-expression of Twist and Snai1: predictor of poor prognosis and biomarker of treatment resistance in untreated prostate cancer. | 1 |
| 38358184 | 2024 | The clinical significance of SNAIL, TWIST, and E-Cadherin expression in gastric mesentery tumor deposits of advanced gastric cancer. | 0 |
| 38369181 | 2024 | Growth differentiation factor-11 upregulates matrix metalloproteinase 2 expression by inducing Snail in human extravillous trophoblast cells. | 0 |
| 38715171 | 2024 | CXCR2/Snail-1-Induced Epithelial-Mesenchymal Transition in the Formation and Progression of RCC with Inferior Vena Cava Tumour Thrombus. | 0 |
| 38815798 | 2024 | Energy stress-activated AMPK phosphorylates Snail1 and suppresses its stability and oncogenic function. | 1 |
| 38890750 | 2024 | TFAP2A downregulation mediates tumor-suppressive effect of miR-8072 in triple-negative breast cancer via inhibiting SNAI1 transcription. | 0 |
| 37611675 | 2024 | Protein kinase D activity is a risk biomarker in prostate cancer that drives cell invasion by a Snail/ERK dependent mechanism. | 0 |
| 38043804 | 2024 | Dual role of Snail1 as transcriptional repressor and activator. | 1 |
| 38141763 | 2024 | The Snail signaling branch downstream of the TGF-β/Smad3 pathway mediates Rho activation and subsequent stress fiber formation. | 3 |
Citation
Joerg Schwock ; William R Geddie
SNAI1 (snail homolog 1 (Drosophila))
Atlas Genet Cytogenet Oncol Haematol. 2010-08-01
Online version: http://atlasgeneticsoncology.org/gene/452/snai1
