TP63 (tumor protein p63)
2012-12-01 Austin Mattox , Zhong Chen , Carter Van Waes AffiliationClinical Genomics Unit, Tumor Biology Section, Head, Neck Surgery Branch, National Institute on Deafness, Other Communication Disorders, NIH, Bethesda, MD, 20892, USA
Identity
DNA/RNA
Note
Description
Proteins
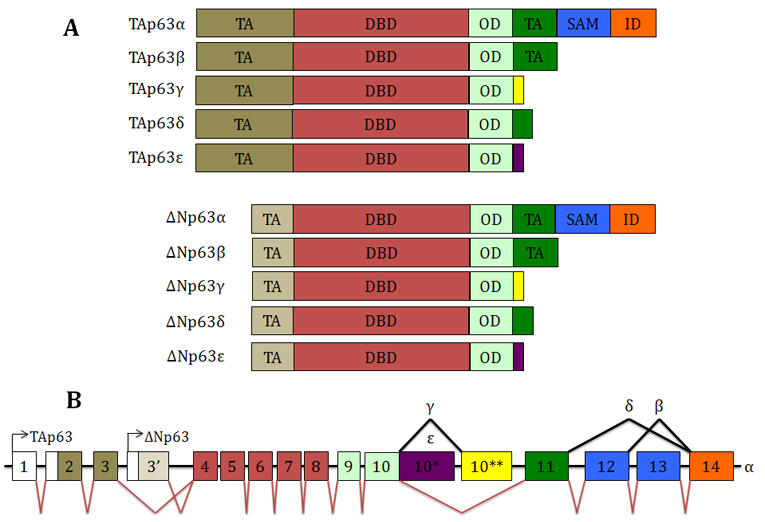
Description
Structure
TP63 binds DNA as either a homo- or a heterotetramer, with isoform composition of the tetramer possibly determining transactivation activity. p63 may also form mixed dimers or tetramers with p73 at relatively higher affinity than with p53, suggesting functional cross talk to regulate transcriptional activity (Davison et al., 1999; Natan and Joerger, 2012). p63 appears to form a dimer of dimers, with monomers consisting of a β-strand followed by two helices (H1 and H2) that adopt a z-shaped double-hairpin conformation with little intramolecular contact between structural elements (Natan and Joerger, 2012). Monomers dimerize via intermolecular antiparallel β-sheet interactions and antiparallel packing of the H1 helices, with important hydrophobic contacts made by key leucine, valine, tyrosine, methionine, and isoleucine residues (Natan and Joerger, 2012). Tetramers are formed by hydrophobic H1-H1 interactions and H2-mediated contact where the H2 helices from the primary dimer clasps the adjacent dimer, packing the tetramer in an orthogonal fashion via H1 helices arrangement (Natan and Joerger, 2012). Analysis of the DBD of TP63 shows higher similarity to that of p73 than p53, and appears to bind a 10-bp DNA sequence containing a "CATG" motif with A/T-rich flanking regions (Chen et al., 2011).
Expression
Localisation
Function
While the interactomes of TP53 and TP73 have been systematically analyzed and cataloged, until recently, no such information had been compiled for TP63 (Tozluoglu et al., 2008; Collavin et al., 2010). Recent protein chip analysis has elucidated 144 proteins that specifically interact with TP63 and are implicated in cell growth/death/survival, chromatin remodeling and gene regulation, RNA processing, protein trafficking and degradation, and other and epithelial differentiation (Huang et al., 2012). A representative protein and its function from each of the aforementioned categories are described below, while a complete list may be found in the supplemental data of Huang et al. (2012).
Cell growth/survival: Signal Transducer and Activator of Transcription 3 (STAT3) - STAT3 is a latent cytoplasmic transcription factor activated in response to various interleukins and growth factors that binds the promoter regions of IL-6-induced plasma protein and intermediate-early genes (Akira et al., 1994; McLoughlin et al., 2005; Huang et al., 2012).
Chromatin remodeling: SWI/SNF Regulator of Chromatin 1 (SMARCC1) - SMARCC1 is a component of the larger SWI/SNF complex responsible for chromatin remodeling necessary for transcriptional activation of certain genes (Wang et al., 1996; Ring et al., 1998; Huang et al., 2012).
Gene regulation/expression: Eukaryotic Translation Initiation Factor 4A2 (EIF4A2) - EIF4A2 plays an important role in the binding of mRNA to the 43S pre-initiation complex during the beginning of protein synthesis (Sudo et al., 1995; Huang et al., 2012).
RNA processing: Splicing Factor 3b, Subunit 4 (SF3B4) - SF3B4 interacts with other spliceosome proteins to help cross-link a 29-nucleotide region in the pre-mRNA near the branch-point sequence in the A complex (Champion-Arnaud and Reed, 1994; Huang et al., 2012).
Protein trafficking: Trafficking Protein Particle Complex 2-Like (TRAPPC2L) - TRAPPC2L is part of the TRAPP multi-subunit tethering complex involved in intracellular vesicle trafficking (Scrivens et al., 2011; Huang et al., 2012).
Protein degradation: Ubiquitin Conjugation Factor E4 B Isoform 1 (UFD2A) - UFD2A has been shown to promote monoubiquitination of p53 and, in combination with MDM2, to promote p53 polyubiquitination (Wu and Leng, 2011; Wu et al., 2011; Huang et al., 2012).
Epithelial differentiation: Keratin 1 (KRT1) - KRT1 is a marker for terminal differentiation in the mammalian epidermis (Lessin et al., 1988; Huang et al., 2012).
Through protein-protein interactions with other transcription and cofactors, TP63 also contributes to the transcriptional regulation of genes involved in cellular differentiation, proliferation/survival, growth suppression, apoptosis, adhesion, inflammation, and metabolism (Perez and Pietenpol, 2007; Viganò and Mantovani, 2007; Yang et al., 2011). Recent work by our laboratory has identified protein-protein interactions between ΔNp63α, TAp73, and c-REL that function to regulate key genes involved in growth arrest and apoptosis of mutant p53 head and neck squamous cell carcinoma (HNSCC) (Lu et al., 2011). Functionally important genes representing each of the aforementioned categories are described below, while more comprehensive reviews may be found in Perez and Pietenpol (2007) and Viganò and Mantovani (2007).
Cellular differentiation: Jagged 1 (JAG1) - JAG1 is the ligand of the Notch receptor. Its binding causes a proteolytic cleavage cascade, leading to translocation of the intracellular component of the Notch receptor and subsequent activation of transcription factors with key roles in cell differentiation and morphogenesis (Gray et al., 1999; Sasaki et al., 2002; Guarnaccia et al., 2004). Overexpression of p63γ has been shown to dramatically upregulate JAG1 protein levels in colorectal cancer, osteogenic sarcoma, lung cancer, hepatocellular carcinoma, and glioma (Sasaki et al., 2002).
Proliferation/survival: Epidermal Growth Factor Receptor (EGFR) - EGFR is the receptor for epidermal growth factor and is involved in modulating cellular functions such as cell proliferation, differentiation, and survival by activating various intracellular signaling cascades such as RAS and STAT that transcribe target genes important in cellular proliferation and survival (Carpenter, 1984; Jamnongjit et al., 2005; Testoni et al., 2006). Knockdown of ΔNp63α has been shown to reduce expression of EGFR in keratinocytes (Testoni et al., 2006).
Growth suppression: Cyclin-Dependent Kinase Inhibitor 1A (CDKN1A) - CDKN1A (p21) associates with cyclins A, D, and E to prevent the G1-S phase transition in mammals (el-Deiry et al., 1993; Westfall et al., 2003). Overexpression of ΔNp63α has been shown to reduce expression of p21 in HEK cells (Westfall et al., 2003).
Apoptosis: p53-Upregulated Modulator of Apoptosis (PUMA) - PUMA binds to BCL2 to induce rapid and profound apoptosis by cytochrome c release (Yu et al., 2001; Flores et al., 2002). Additionally, recent work has shown that a complex of ΔNp63α, with TAp73 or c-REL, can modify expression of key growth arrest and apoptotic genes such as p21WAF1, NOXA, and PUMA (Lu et al., 2011; Yang et al., 2011).
Adhesion: Integrin Alpha 6 (ITGA6) - ITGA6 is a cell surface adhesion molecule that may help regulate migration and layer formation, especially in epithelial cells. Mouse models deficient in ITGA6 display severe blistering of the skin and other epithelia and die shortly after birth (Georges-Labouesse et al., 1996; Carroll et al., 2006). Loss of endogenous p63 in mammary epithelial cells has been shown to induce detachment and cell death, presumably because p63 regulates expression of key adhesion molecules such as ITGA6, ITGB1, ITGB4 other ECM components, and cadherins-catenins (Carroll et al., 2006; Yang et al., 2011).
Inflammation: ΔNp63 overexpression has been observed in head and neck squamous cell carcinoma (HNSCC), which are associated with inflammation. TNF-α, a potent pro-inflammatory cytokine, promoted NF-κB, c-REL and RELA complexes with nuclear ΔNp63, to promote a broad-spectrum production of cytokines and chemokines (Lu et al., 2011; Yang et al., 2011). In addition, in squamous epithelia of ΔNp63α transgenic mice, severe inflammation, skin lesions and erythema were observed after ΔNp63 expression was induced. Microscopically, hyperplastic and hyperproliferative epidermis with diffuse infiltration of inflammatory cells in the dermis was observed (Yang et al., 2011). NF-κB family proteins, c-Rel and RelA, and numerous inflammatory cytokines and chemokines were significantly upregulated in ΔNp63 transgenic mice.
Metabolism: Fatty Acid Synthase (FASN) - FASN catalyzes the conversion of Acetyl-CoA and Malonyl-CoA into long-chain saturated fatty acids with the help of NADPH (Wakil, 1989; DErchia et al., 2006). ΔNp63α (and TAp73α) expression has also been shown to induce promoter and enhancer activity in human FASN gene by binding to p53 response elements (DErchia et al., 2006).
Mouse models TP63 knockout mice are born alive but have striking developmental defects including absent or truncated limbs, lack of stratified epithelia, and lack hair follicles, teeth, and mammary glands. Thus, TP63 is essential for epidermal-mesenchymal interactions during embryonic development, including regenerative limb proliferation, craniofacial and epithelial development, and differentiation of squamous epithelia (Yang et al., 1998; Mills et al., 1999; McKeon, 2004; Laurikkala et al., 2006). Recently, ΔNp63α transgenic mice have been developed using tissue-specific tetracycline inducible expression. Mice expressing ΔNp63α under a tissue-specific promoter (SPC) for lung epithelium exhibited Keratin 5 and 14 induction, trans-differentiation to an epidermal cell lineage, and squamous metaplasia. Overexpression of ΔNp63α under a K5 promoter in wild-type epidermis results in severe defects in hair follicle development and cycling, leading to severe hair loss and a depleted hair follicle stem-cell niche (Romano et al., 2009; Romano et al., 2010). In addition, ΔNp63α overexpression induced marked skin inflammation, and skin hyperplasia (Yang et al., 2011). A more detailed summary of the phenotypes of different p63 knockout and transgenic mouse models has recently been published (Vanbokhoven et al., 2011).
Homology
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12838557 | 2003 | EEC syndrome type 3 with a heterozygous germline mutation in the P63 gene and B cell lymphoma. | Akahoshi K et al |
| 7512451 | 1994 | Molecular cloning of APRF, a novel IFN-stimulated gene factor 3 p91-related transcription factor involved in the gp130-mediated signaling pathway. | Akira S et al |
| 9799841 | 1998 | Cloning and chromosomal mapping of the human p53-related KET gene to chromosome 3q27 and its murine homolog Ket to mouse chromosome 16. | Augustin M et al |
| 6327062 | 1984 | Properties of the receptor for epidermal growth factor. | Carpenter G et al |
| 16715076 | 2006 | p63 regulates an adhesion programme and cell survival in epithelial cells. | Carroll DK et al |
| 7958871 | 1994 | The prespliceosome components SAP 49 and SAP 145 interact in a complex implicated in tethering U2 snRNP to the branch site. | Champion-Arnaud P et al |
| 21464285 | 2011 | Structures of p63 DNA binding domain in complexes with half-site and with spacer-containing full response elements. | Chen C et al |
| 15752259 | 2005 | Expression of p63 protein and mRNA in oral epithelial dysplasia. | Chen YK et al |
| 20379196 | 2010 | p53-family proteins and their regulators: hubs and spokes in tumor suppression. | Collavin L et al |
| 16582625 | 2006 | The fatty acid synthase gene is a conserved p53 family target from worm to human. | D'Erchia AM et al |
| 10373484 | 1999 | p73 and p63 are homotetramers capable of weak heterotypic interactions with each other but not with p53. | Davison TS et al |
| 20484388 | 2010 | p63 and p73, the ancestors of p53. | Dötsch V et al |
| 22076464 | 2012 | Identification of germline susceptibility loci in ETV6-RUNX1-rearranged childhood acute lymphoblastic leukemia. | Ellinghaus E et al |
| 11932750 | 2002 | p63 and p73 are required for p53-dependent apoptosis in response to DNA damage. | Flores ER et al |
| 8673141 | 1996 | Absence of integrin alpha 6 leads to epidermolysis bullosa and neonatal death in mice. | Georges-Labouesse E et al |
| 10079256 | 1999 | Human ligands of the Notch receptor. | Gray GE et al |
| 15358557 | 2004 | Exon 6 of human Jagged-1 encodes an autonomously folding unit. | Guarnaccia C et al |
| 946410 | 1976 | The syndrome of ankyloblepharon, ectodermal defects and cleft lip and palate: an autosomal dominant condition. | Hay RJ et al |
| 14647429 | 2004 | Gadd45a regulates matrix metalloproteinases by suppressing DeltaNp63alpha and beta-catenin via p38 MAP kinase and APC complex activation. | Hildesheim J et al |
| 21725308 | 2011 | A genome-wide association study identifies two new lung cancer susceptibility loci at 13q12.12 and 22q12.2 in Han Chinese. | Hu Z et al |
| 22672905 | 2012 | Global tumor protein p53/p63 interactome: making a case for cisplatin chemoresistance. | Huang Y et al |
| 16260720 | 2005 | Epidermal growth factor receptor signaling is required for normal ovarian steroidogenesis and oocyte maturation. | Jamnongjit M et al |
| 16524929 | 2006 | p63 regulates multiple signalling pathways required for ectodermal organogenesis and differentiation. | Laurikkala J et al |
| 17446929 | 2007 | The p63/p73 network mediates chemosensitivity to cisplatin in a biologically defined subset of primary breast cancers. | Leong CO et al |
| 2461420 | 1988 | Chromosomal mapping of human keratin genes: evidence of non-linkage. | Lessin SR et al |
| 21933882 | 2011 | TNF-α promotes c-REL/ΔNp63α interaction and TAp73 dissociation from key genes that mediate growth arrest and apoptosis in head and neck cancer. | Lu H et al |
| 8737655 | 1996 | EEC syndrome and genitourinary anomalies: an update. | Maas SM et al |
| 19700772 | 2009 | Identification and functional characterization of two new transcriptional variants of the human p63 gene. | Mangiulli M et al |
| 11159940 | 2001 | Hay-Wells syndrome is caused by heterozygous missense mutations in the SAM domain of p63. | McGrath JA et al |
| 15037544 | 2004 | p63 and the epithelial stem cell: more than status quo? | McKeon F et al |
| 15976028 | 2005 | IL-6 trans-signaling via STAT3 directs T cell infiltration in acute inflammation. | McLoughlin RM et al |
| 10227293 | 1999 | p63 is a p53 homologue required for limb and epidermal morphogenesis. | Mills AA et al |
| 15280445 | 2004 | p63 and p73: roles in development and tumor formation. | Moll UM et al |
| 16641997 | 2006 | The DNA sequence, annotation and analysis of human chromosome 3. | Muzny DM et al |
| 22100306 | 2012 | Structure and kinetic stability of the p63 tetramerization domain. | Natan E et al |
| 19142959 | 2009 | Comprehensive mutational analysis and mRNA isoform quantification of TP63 in normal and neoplastic human prostate cells. | Parsons JK et al |
| 12086851 | 2002 | DeltaNp63 induces beta-catenin nuclear accumulation and signaling. | Patturajan M et al |
| 17297308 | 2007 | Transcriptional programs regulated by p63 in normal epithelium and tumors. | Perez CA et al |
| 8456838 | 1993 | ADULT-syndrome: an autosomal-dominant disorder with pigment anomalies, ectrodactyly, nail dysplasia, and hypodontia. | Propping P et al |
| 17041931 | 2006 | Further phenotypic and genetic variation in ADULT syndrome. | Reisler TT et al |
| 9693044 | 1998 | Five SWI/SNF-related, matrix-associated, actin-dependent regulator of chromatin (SMARC) genes are dispersed in the human genome. | Ring HZ et al |
| 16724007 | 2006 | Delineation of the ADULT syndrome phenotype due to arginine 298 mutations of the p63 gene. | Rinne T et al |
| 16413471 | 2006 | p63 mediates survival in squamous cell carcinoma by suppression of p73-dependent apoptosis. | Rocco JW et al |
| 19461998 | 2009 | An active role of the DeltaN isoform of p63 in regulating basal keratin genes K5 and K14 and directing epidermal cell fate. | Romano RA et al |
| 20972438 | 2010 | A multi-stage genome-wide association study of bladder cancer identifies multiple susceptibility loci. | Rothman N et al |
| 11641404 | 2002 | The p53 family member genes are involved in the Notch signal pathway. | Sasaki Y et al |
| 21525244 | 2011 | C4orf41 and TTC-15 are mammalian TRAPP components with a role at an early stage in ER-to-Golgi trafficking. | Scrivens PJ et al |
| 11971180 | 2002 | TAp63gamma (p51A) and dNp63alpha (p73L), two major isoforms of the p63 gene, exert opposite effects on the vascular endothelial growth factor (VEGF) gene expression. | Senoo M et al |
| 21798893 | 2011 | The mutational landscape of head and neck squamous cell carcinoma. | Stransky N et al |
| 19570515 | 2009 | TAp63 prevents premature aging by promoting adult stem cell maintenance. | Su X et al |
| 8521730 | 1995 | Isolation and mapping of the human EIF4A2 gene homologous to the murine protein synthesis initiation factor 4A-II gene Eif4a2. | Sudo K et al |
| 17172858 | 2006 | Identification of new p63 targets in human keratinocytes. | Testoni B et al |
| 18660513 | 2008 | Cataloging and organizing p73 interactions in cell cycle arrest and apoptosis. | Tozluoğlu M et al |
| 21471985 | 2011 | p63, a story of mice and men. | Vanbokhoven H et al |
| 17297297 | 2007 | Hitting the numbers: the emerging network of p63 targets. | Viganò MA et al |
| 2669958 | 1989 | Fatty acid synthase, a proficient multifunctional enzyme. | Wakil SJ et al |
| 8804307 | 1996 | Diversity and specialization of mammalian SWI/SNF complexes. | Wang W et al |
| 15033906 | 2004 | p63: Molecular complexity in development and cancer. | Westfall MD et al |
| 21558803 | 2011 | UBE4B, a ubiquitin chain assembly factor, is required for MDM2-mediated p53 polyubiquitination and degradation. | Wu H et al |
| 21317885 | 2011 | UBE4B promotes Hdm2-mediated degradation of the tumor suppressor p53. | Wu H et al |
| 9774969 | 1998 | p63, a p53 homolog at 3q27-29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities. | Yang A et al |
| 21576089 | 2011 | ΔNp63 versatilely regulates a Broad NF-κB gene program and promotes squamous epithelial proliferation, migration, and inflammation. | Yang X et al |
| 11463391 | 2001 | PUMA induces the rapid apoptosis of colorectal cancer cells. | Yu J et al |
| 20225093 | 2010 | p63 short isoforms are found in invasive carcinomas only and not in benign breast conditions. | de Biase D et al |
| 8242752 | 1993 | WAF1, a potential mediator of p53 tumor suppression. | el-Deiry WS et al |
| 11462173 | 2001 | p63 Gene mutations in eec syndrome, limb-mammary syndrome, and isolated split hand-split foot malformation suggest a genotype-phenotype correlation. | van Bokhoven H et al |
Other Information
Locus ID:
NCBI: 8626
MIM: 603273
HGNC: 15979
Ensembl: ENSG00000073282
Variants:
dbSNP: 8626
ClinVar: 8626
TCGA: ENSG00000073282
COSMIC: TP63
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37158316 | 2024 | Absent meibomian glands and cone dystrophy in ADULT syndrome: identification by whole exome sequencing of pathogenic variants in two causal genes TP63 and CNGB3. | 0 |
| 38012539 | 2024 | Tumour 63 protein (p63) in breast pathology: biology, immunohistochemistry, diagnostic applications, and pitfalls. | 1 |
| 38281558 | 2024 | A spectrum of TP63-related disorders with eight affected individuals in five unrelated families. | 0 |
| 38509096 | 2024 | Reciprocal inhibition between TP63 and STAT1 regulates anti-tumor immune response through interferon-γ signaling in squamous cancer. | 2 |
| 38528613 | 2024 | TP63 truncating mutation causes increased cell apoptosis and premature ovarian insufficiency by enhanced transcriptional activation of CLCA2. | 1 |
| 38823762 | 2024 | ΔNp63-restricted viral mimicry response impedes cancer cell viability and remodels tumor microenvironment in esophageal squamous cell carcinoma. | 0 |
| 38880601 | 2024 | Identification of HTRA4 as a Transcriptional Target of p63 in Trophoblast. | 0 |
| 38891051 | 2024 | Distinct Effects of Respiratory Viral Infection Models on miR-149-5p, IL-6 and p63 Expression in BEAS-2B and A549 Epithelial Cells. | 0 |
| 37158316 | 2024 | Absent meibomian glands and cone dystrophy in ADULT syndrome: identification by whole exome sequencing of pathogenic variants in two causal genes TP63 and CNGB3. | 0 |
| 38012539 | 2024 | Tumour 63 protein (p63) in breast pathology: biology, immunohistochemistry, diagnostic applications, and pitfalls. | 1 |
| 38281558 | 2024 | A spectrum of TP63-related disorders with eight affected individuals in five unrelated families. | 0 |
| 38509096 | 2024 | Reciprocal inhibition between TP63 and STAT1 regulates anti-tumor immune response through interferon-γ signaling in squamous cancer. | 2 |
| 38528613 | 2024 | TP63 truncating mutation causes increased cell apoptosis and premature ovarian insufficiency by enhanced transcriptional activation of CLCA2. | 1 |
| 38823762 | 2024 | ΔNp63-restricted viral mimicry response impedes cancer cell viability and remodels tumor microenvironment in esophageal squamous cell carcinoma. | 0 |
| 38880601 | 2024 | Identification of HTRA4 as a Transcriptional Target of p63 in Trophoblast. | 0 |
Citation
Austin Mattox ; Zhong Chen ; Carter Van Waes
TP63 (tumor protein p63)
Atlas Genet Cytogenet Oncol Haematol. 2012-12-01
Online version: http://atlasgeneticsoncology.org/gene/365/tp63
