TRIM37 (tripartite motif-containing 37)
2006-06-01 Elif Ayse Erson , .M Elizabeth Petty AffiliationBiology Department, Room: 141, Middle East Technical University, Ankara 06531, TURKEY
DNA/RNA
Description
Transcription
Both of these variants encode an identical protein product but they use different termination codons and have different 3 untranslated sequences. In the first transcript, all of the exon 24 sequence is included, whereas in the second one, only the first 79 nucleotides of exon 24 are included followed by nucleotides of exon 25, resulting in a shorter transcript.
A third "TRIM37adel23" transcript is detected as an alternatively spliced transcript of TRIM37a. This transcript lacks exon 23 (only 117 bp) with an in frame deletion of 39 amino acids that span the evolutionarily conserved DES (aspartate-glutamate-serine) rich motif at the C-terminus.
A 4.4 kb band representing the full-length transcript of TRIM37 is detected in RNA representing brain, placenta, lung, liver, skeletal muscle, kidney, pancreas, spleen, thymus, prostate, ovary, small intestine, colon and leukocyte samples by hybridization of several PCR-generated TRIM37 cDNA probes on a multi-tissue Northern blot. In addition, a strong signal of 3.9 kb is detected in the testis sample and a 2.6 kb band is noted in the heart sample.
In situ hybridization suggests TRIM37 expression patterns in multiple tissues during mouse and human embryogenesis. No Trim37 expression is detected up to E9.5. At E11.5, Trim37 expression is detected in cells lining the esophagus and bronchias well as the innermost cells of the optic cup adjacent to the lens. Between E12.5 and E14.5, TRIM37 is detected in different parts of ganglia and throughout liver. Intense expression is seen in gut epithelium of the midgut, stomach, esophagus and in the primitive seminiferous tubules of the developing testis at E14.5. Expression is also evident in the olfactory epithelium, epithelial lining of the bronchioles, surface ectoderm and in the developing eye lens epithelium, neural layer of the retina (but not in the optic nerve), epithelium of developing nephron, mesonephric duct, and epithelial pancreas cells. Similar to the E14.5 mouse, in 7 week old human embryos, TRIM37 expression can be detected in similar tissues including dorsal root ganglia, liver, submandibular gland and epithelial lining of the gut lumen. At 10 weeks, intense TRIM37 expression can be detected in dorsal root and trigeminal ganglia, epithelia in multiple tissues and liver. However, no TRIM37 transcript can be detected in migrating neural crest cells.
In another study, TaqMan PCR results suggest expression of TRIM37a and TRIM37b to be the highest in testis. In the brain, TRIM37a expression is 15-fold higher in adult and 20-fold higher in fetal tissue compared to the expression in heart as a reference. The lowest TRIM37a expression is detected in skeletal muscle with 0.3 and 0.8 times the expression of heart in adult and in fetal tissues.
Pseudogene
Proteins
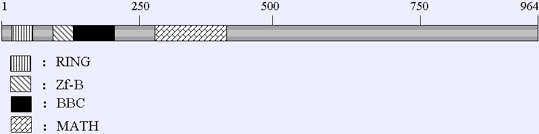
Description
Expression
In post-pubertal testis, a stage-specific cytoplasmic Trim37 staining of germ cells can be detected. Developing sperm from type B spermatogonia to early round spermatids show immunoreactivity. In post-pubertal ovary, intense Trim37 staining is observed in maturing oocytes as well as in the granulosa cells, luteal gland, and in the epithelium of the fallopian tubes.
Localisation
Function
Homology
P.troglodytes , LOC455163 similar to POB1, 705 aa.
C.familiaris , LOC480575 similar to tripartite motif protein 37, 962 aa.
M.musculus , Trim37 tripartite motif protein 37, 961 aa.
R.norvegicus , Trim37_predicted, 1075 aa.
G.gallus , TRIM37, 983 aa.
A.gambiae , ENSANGG00000009789, 153 aa.
Mutations
Germinal
2. c.2212delG : This "Finminor" mutation is a 1-bp deletion of a G at nucleotide c.2212 and results in a frameshift that predicts a stop codon 30 codons downstream. Finminor is found to be associated with a distinct haplotype that is found in 2 of 100 Finnish MUL chromosomes. This mutation is predicted to generate a truncated 767 aa protein. Two patients were found to be compound heterozygotes for the Finmajor and Finminor mutations.
3. c.838delACTTT : This homozygous "Czech" mutation found in a Czech patient is a 5-bp deletion of ACTTT at nucleotides c.838_842 leading to a frameshift that results in a stop codon 55 codons downstream. This mutation is predicted to generate a truncated 334 aa protein.
4. c.134insA : this "American" mutation is a homozygous 1-bp insertion of an A nucleotide after c.1346 in an American patient. The mutation disrupts the reading frame and results in a stop codon eight codons downstream. This mutation is predicted to generate a truncated 334 aa protein.
5. c.855_862delTGAATTAG : This mutation detected in a Turkish family is an 8-bp deletion. On the genomic level, aberrant splicing was implicated due to a transition at the splice acceptor (AG) at position c.855 1G>A. A cryptic splice site (AG, c.860) 8-bp downstream is activated, which leads to disruption of the open reading frame (ORF) through a premature stop codon (PTC, TGA) at position c.1045 1047 that translates into a truncated protein.
6. c.745C>T : This mutation detected in a Canadian patient is predicted to generate a truncated 249 aa protein.
7. c.965G>T : This mutation detected in a Canadian patient is predicted to generate a missense amino acid at the 322th position (Gly322Val).
8. c.1037_1040dupAGAT : This mutation detected in a Canadian patient is a four base-pair duplication in exon 13. It is predicted to generate a frame-shift at amino acid position 347, and truncation of the protein product after seven code-shifted amino acids.
9. c.1411C>T : This mutation detected in Tunusian-German and Canadian patients is predicted to generate a truncated 471 aa protein.
10. c.1314+507_1668-207del : This mutation detected in a Sicilian patient is predicted to generate a genomic deletion of 8603 bp with break points in introns 14 and 16 (c.1314+507_1668-207del), thus deleting exons 15 and 16. At the protein level this mutation leads to a frame-shift at 439th aa and truncation of the protein product after four code-shifted amino acids.
11. c.2056C>T : This mutation detected in a Saudi-Arabian patient is predicted to generate a truncated 686 aa protein.
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 10888877 | 2000 | Gene encoding a new RING-B-box-Coiled-coil protein is mutated in mulibrey nanism. | Avela K et al |
| 11774034 | 2001 | Overexpressed genes/ESTs and characterization of distinct amplicons on 17q23 in breast cancer cells. | Erson AE et al |
| 16310976 | 2006 | Characterisation of the mulibrey nanism-associated TRIM37 gene: transcription initiation, promoter region and alternative splicing. | Hämäläinen RH et al |
| 12754710 | 2003 | A novel splice site mutation in the TRIM37 gene causes mulibrey nanism in a Turkish family with phenotypic heterogeneity. | Jagiello P et al |
| 11704850 | 2001 | PML protein isoforms and the RBCC/TRIM motif. | Jensen K et al |
| 16514549 | 2006 | Tissue expression of the mulibrey nanism-associated Trim37 protein in embryonic and adult mouse tissues. | Kallijärvi J et al |
| 16306379 | 2005 | Insulin resistance syndrome in subjects with mutated RING finger protein TRIM37. | Karlberg N et al |
| 11578880 | 2001 | Expression of MUL, a gene encoding a novel RBCC family ring-finger protein, in human and mouse embryogenesis. | Lehesjoki AE et al |
| 11331760 | 2001 | Comprehensive copy number and gene expression profiling of the 17q23 amplicon in human breast cancer. | Monni O et al |
| 10339815 | 1999 | BLAST 2 Sequences, a new tool for comparing protein and nucleotide sequences. | Tatusova TA et al |
| 11279055 | 2001 | A diverse family of proteins containing tumor necrosis factor receptor-associated factor domains. | Zapata JM et al |
Other Information
Locus ID:
NCBI: 4591
MIM: 605073
HGNC: 7523
Ensembl: ENSG00000108395
Variants:
dbSNP: 4591
ClinVar: 4591
TCGA: ENSG00000108395
COSMIC: TRIM37
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 35163097 | 2022 | TRIM37 Promotes Pancreatic Cancer Progression through Modulation of Cell Growth, Migration, Invasion, and Tumor Immune Microenvironment. | 2 |
| 35220664 | 2022 | Liver pathology and biochemistry in patients with mutations in TRIM37 gene (Mulibrey nanism). | 1 |
| 35864973 | 2022 | TRIM37 Augments AP-2γ Transcriptional Activity and Cellular Localization via K63-linked Ubiquitination to Drive Breast Cancer Progression. | 3 |
| 35921902 | 2022 | TRIM37 promotes the aggressiveness of ovarian cancer cells and increases c-Myc expression by binding to HUWE1. | 1 |
| 35163097 | 2022 | TRIM37 Promotes Pancreatic Cancer Progression through Modulation of Cell Growth, Migration, Invasion, and Tumor Immune Microenvironment. | 2 |
| 35220664 | 2022 | Liver pathology and biochemistry in patients with mutations in TRIM37 gene (Mulibrey nanism). | 1 |
| 35864973 | 2022 | TRIM37 Augments AP-2γ Transcriptional Activity and Cellular Localization via K63-linked Ubiquitination to Drive Breast Cancer Progression. | 3 |
| 35921902 | 2022 | TRIM37 promotes the aggressiveness of ovarian cancer cells and increases c-Myc expression by binding to HUWE1. | 1 |
| 32572790 | 2021 | ASB16-AS1 up-regulated and phosphorylated TRIM37 to activate NF-κB pathway and promote proliferation, stemness, and cisplatin resistance of gastric cancer. | 28 |
| 33491649 | 2021 | TRIM37 prevents formation of centriolar protein assemblies by regulating Centrobin. | 10 |
| 33728810 | 2021 | Circular RNA circRNA_101996 promoted cervical cancer development by regulating miR-1236-3p/TRIM37 axis. | 13 |
| 33839419 | 2021 | TRIM37 negatively regulates inflammatory responses induced by virus infection via controlling TRAF6 ubiquitination. | 7 |
| 33983387 | 2021 | TRIM37 prevents formation of condensate-organized ectopic spindle poles to ensure mitotic fidelity. | 7 |
| 34130705 | 2021 | TRIM37 orchestrates renal cell carcinoma progression via histone H2A ubiquitination-dependent manner. | 17 |
| 32572790 | 2021 | ASB16-AS1 up-regulated and phosphorylated TRIM37 to activate NF-κB pathway and promote proliferation, stemness, and cisplatin resistance of gastric cancer. | 28 |
Citation
Elif Ayse Erson ; .M Elizabeth Petty
TRIM37 (tripartite motif-containing 37)
Atlas Genet Cytogenet Oncol Haematol. 2006-06-01
Online version: http://atlasgeneticsoncology.org/gene/42703/trim37
