Currarino syndrome
2007-06-01 Sally Ann Lynch AffiliationNewcastle upon Tyne Hospitals NHS Trust, Newcastle upon Tyne, NE1 4LP, UK
Identity
Name
Currarino syndrome
Alias
Currarino triad , Anal atresia, sacral anomalies, presacral mass (ASP) , Sacral defect with anterior meningocoele
Note
Currarino syndrome is a multiple congenital anomalies syndrome characterized by partial agenesis of the sacrum in association with pelvic malformation. Anal atresia and the presence of a pre-sacral mass (teratoma and\/or anterior meningocoele) make up the so called Currarino triad. Other malformations, such as renal (35%) and gynaecological (19%) are common. Hirschsprungs diseasehas also been recorded. Other rarer complications include pelvic abcess, malignant degeneration of a pre-sacral teratoma, E Coli ascending meningitis and spinal cord tethering. Recurrence of a benign teratoma has been recorded. Urinary incontinence, dysmenorrhoea, dyspareunia, poor sphincter control, sacral anaesthesia and headaches precipitated by coughing or straining have been reported.
Inheritance
Autosomal Dominant. De novo mutations have been recorded and account for approximately 15% of cases.
Omim
176450
Mesh
C536221
Orphanet
1552 Currarino triad
Umls
C1531773
Clinics
Note
Currarino syndrome was described as a triad by Guido Currarino, an American radiologist in 1981. The triad consists of:
Sickle shaped sacrum Pre-sacral mass (meningocoele and\/or tumour (teratoma; hamartoma) Anal atresia
The term Currarino syndrome is preferred as it is now known that there are other components to this genetic disorder. Bowel obstruction in infancy or chronic constipation in childhood are the commonest presenting symptoms. Gynaecological and renal malformation are commonly described. Hirschsprungs disease and meningitis (often E Coli ascending meningitis) have been recorded. Perianal sepsis, which is found in at least 10% of patients, can indicate the presence of an underlying presacral mass. Tumour development within the teratoma has been documented.
A report describing Currarino syndrome with ventriculomegaly due to a Arnold-Chiari type II malformation must be interpreted with caution. The affected child was dysmorphic with hypertelorism, a short enlarged neck and camptodactyly. The mother and brother also had Currarino syndrome without these added features. It is quite possible that this child had two co-existing disorders, one the Currarino inherited from a mother with typical features and secondly a de novo Arnold-Chiari type II malformation.
As with many dominant disorders the phenotype is very variable. It is estimated that approximately 50% of those who inherit the gene will present with the severe phenotype (requiring surgery), 25% will have symptoms albeit milder and 25% will be asymptomatic heterozygotes.
The term Currarino syndrome is preferred as it is now known that there are other components to this genetic disorder. Bowel obstruction in infancy or chronic constipation in childhood are the commonest presenting symptoms. Gynaecological and renal malformation are commonly described. Hirschsprungs disease and meningitis (often E Coli ascending meningitis) have been recorded. Perianal sepsis, which is found in at least 10% of patients, can indicate the presence of an underlying presacral mass. Tumour development within the teratoma has been documented.
A report describing Currarino syndrome with ventriculomegaly due to a Arnold-Chiari type II malformation must be interpreted with caution. The affected child was dysmorphic with hypertelorism, a short enlarged neck and camptodactyly. The mother and brother also had Currarino syndrome without these added features. It is quite possible that this child had two co-existing disorders, one the Currarino inherited from a mother with typical features and secondly a de novo Arnold-Chiari type II malformation.
As with many dominant disorders the phenotype is very variable. It is estimated that approximately 50% of those who inherit the gene will present with the severe phenotype (requiring surgery), 25% will have symptoms albeit milder and 25% will be asymptomatic heterozygotes.
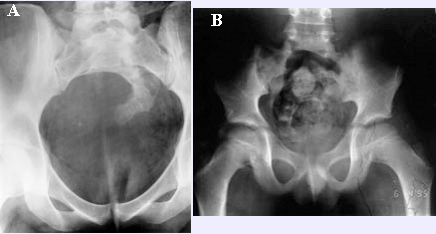
Figure A: Pelvic x ray of adult female showing a scimitar sacrum with a right sided defect. In our cohort, 38% had a sickle shaped sacrum with a left sided defect and 37% had a similar sickle shaped sacrum with a right sided defect.
Figure B: Pelvic x ray of male child showing a left sided sacral defect with associated faecal loading. Courtesy Sally Ann Lynch, reprinted from J Med Genet. 2000; 37:561-566 by permission of the publisher BMJ Publishing Group Ltd of the BMA.
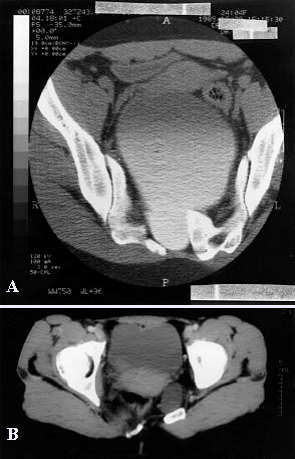
Figure A: CT scan of pelvis showing a large anterior meningocele filling with contrast (this is the same patient as shown in fig 1A).
Figure B: CT of pelvis of adult female showing gross distortion of pelvic anatomy with an abnormal sacral bone and a presacral mass.Courtesy Sally Ann Lynch, reprinted from J Med Genet. 2000; 37:561-566 by permission of the publisher BMJ Publishing Group Ltd of the BMA.
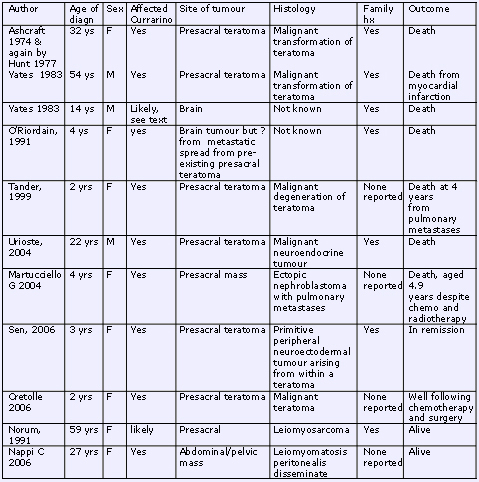
Table 1 summary of the malignant tumours reported in the literature.
Neoplastic risk
The presacral mass associated with the Currarino triad may be a teratoma, hamartoma, neuroenteric cyst, anterior meningocoele or a combination of these three.
A teratoma is a neoplasm originating from primordial germ cells and is derived from all three embryonal germ layers.
A hamartoma is a mass produced by disorganized local tissue growth and can contain all three germ layers.
A teratoma carries a risk for germ cell malignancy, a hamartoma does not.
It has been argued that the reason the risk for malignant degeneration in Currarino syndrome is low is because most of these pre-sacral masses which have previously been classified as teratomas are more likely to be hamartomas. In those rare instances where malignancy has been recorded it is possible that this could have resulted from a secondary carcinoma developing in a chronically inflamed hamartoma rather than a germ cell malignancy.
Malignant change of the pre-sacral tumour has been described but is a rare occurance. The histological appearances are varied and often poorly described particularly in the older literature.
A 32 year old women, who was known to have a pre-sacral teratoma, died from metastatic spread of the tumour. Histological findings were not included in the report (Ashcraft 1973; same patient also reported by Hunt, 1977).
A 54 year old male from a known Currarino family was diagnosed with a presacral mass. This was shown to be a teratoma which had undergone malignant change. A relative of this man, aged 14 years, who had been treated for a rectal stricture, died from a brain tumour, no histology was available. (Yates et al 1983).
A child who died from probable brain metastases was reported in a large Irish pedigree (ORiordain et al 1991). Histology was not available and the child did not have a post mortem. This child had presented at birth with a pre-sacral mass and had surgery. The presacral teratoma recurred at aged 4 years and she died from a brain tumour shortly afterwards.
A female infant who had complete excision of a benign pre-sacral teratoma aged 14 months had a recurrence of a pre-sacral mass at aged 2 ( Tander et al 1999). Histology confirmed recurrence of the teratoma with malignant transformation. The AFP level was extremely high. Treatment was by excision and chemotherapy (bleomycin, etopside and cis-platin) but unfortunately the tumour recurred after 12 months She responded to the same course of chemotherapy but an aggressive recurrence at age 4 years did not respond to treatment and she died from pulmonary metastases.
A 22 year old male was reported who developed a malignant neuroectodermal tumour arising from a pre-sacral mass, (Urioste 2004). He had previously been asymptomatic. He presented with a painless abdominal swelling. A CT scan showed multiple cysts in the liver and it was thought he had isolated liver cystic disease. Lytic pelvic lesions were identified 1 year later on CT and a biopsy of one of these lesions confirmed a well differentiated neuroendocrine tumour. A presacral mass was identified in follow up scans suggestive of a presacral mass with malignant transformation. Biopsy of the mass and the liver confirmed the presence of a neuroendocrine tumour. Chemotherapy was ineffective and he subsequently died. Autopsy confirmed tumour invasion of the liver, pancreas, kidneys and mesenteric lympth nodes. The pre-sacral mass was comprised of neoplastic tissue, teratoma, muscle and fibroadipous tissue.
A 4 year old girl was reported with Currarino syndrome who developed an ectopic nephroblastoma and pulmonary metastases from a presacral mass (Martucciello G 2004). She died despite chemo and radiotherapy.
A 2 year old girl developed a malignant teratoma from a pre-existing presacral mass (Cretolle et al 2006). She was well following chemotherapy.
A 3 year old girl was reported with a malignant tumour arising within a teratoma (Sen et al 2006). Histology revealed the presence of two components; residual mature teratoma and a necrotic malignant tumour resembling a primitive peripheral neuroectodermal tumour. A chemotherapy regimen according to the Euro Ewing 99 protocol was given in addition to radiotherapy. She remained well 8 months following her treatment.
One report of leiomyosarcoma in a grandmother of an affected child with Currarino has been reported (Norum 1991).
In addition, a further case of Leiomyomatosis peritonealis disseminate (LPD) in a 27 year old female with Currarino syndrome has been described in the literature (Nappi 2006). Malignant degeneration of LPD to a leiomyosarcoma has been reported but is rare.
A sacrococcygeal teratoma has been described with a deletion of chromosome 7q, partial trisomy 2p. Malignant degeneration had not occurred and the child died from post-operative complications (Le Caignec 2003). This teratoma was a posterior teratoma and similar to the type often identified antenatally. It is thought to have a different genetic basis to the teratomas seen in the Currarino syndrome. No mutations in the HLXB9 gene have been found in such cases. It is therefore interesting that this child had deleted the HLXB9 gene suggesting some genetic relationship between both conditions.
In classical Currarino syndrome the pre-sacral mass tends to be anterior to the sacrum. The sacrococcygeal teratomas have appeared to be a different group in that they present in the posterior coccygeal region, are often sporadic, occur more commonly in females and commonly become malignant. Mutational analysis of the HLXB9 gene has been negative in a number of cases. This recent case is interesting as it does suggest that HLXB9 is involved in some cases of posterior teratomas (the gene was shown to be deleted in this case) in some cases.
A teratoma is a neoplasm originating from primordial germ cells and is derived from all three embryonal germ layers.
A hamartoma is a mass produced by disorganized local tissue growth and can contain all three germ layers.
A teratoma carries a risk for germ cell malignancy, a hamartoma does not.
It has been argued that the reason the risk for malignant degeneration in Currarino syndrome is low is because most of these pre-sacral masses which have previously been classified as teratomas are more likely to be hamartomas. In those rare instances where malignancy has been recorded it is possible that this could have resulted from a secondary carcinoma developing in a chronically inflamed hamartoma rather than a germ cell malignancy.
Malignant change of the pre-sacral tumour has been described but is a rare occurance. The histological appearances are varied and often poorly described particularly in the older literature.
A 32 year old women, who was known to have a pre-sacral teratoma, died from metastatic spread of the tumour. Histological findings were not included in the report (Ashcraft 1973; same patient also reported by Hunt, 1977).
A 54 year old male from a known Currarino family was diagnosed with a presacral mass. This was shown to be a teratoma which had undergone malignant change. A relative of this man, aged 14 years, who had been treated for a rectal stricture, died from a brain tumour, no histology was available. (Yates et al 1983).
A child who died from probable brain metastases was reported in a large Irish pedigree (ORiordain et al 1991). Histology was not available and the child did not have a post mortem. This child had presented at birth with a pre-sacral mass and had surgery. The presacral teratoma recurred at aged 4 years and she died from a brain tumour shortly afterwards.
A female infant who had complete excision of a benign pre-sacral teratoma aged 14 months had a recurrence of a pre-sacral mass at aged 2 ( Tander et al 1999). Histology confirmed recurrence of the teratoma with malignant transformation. The AFP level was extremely high. Treatment was by excision and chemotherapy (bleomycin, etopside and cis-platin) but unfortunately the tumour recurred after 12 months She responded to the same course of chemotherapy but an aggressive recurrence at age 4 years did not respond to treatment and she died from pulmonary metastases.
A 22 year old male was reported who developed a malignant neuroectodermal tumour arising from a pre-sacral mass, (Urioste 2004). He had previously been asymptomatic. He presented with a painless abdominal swelling. A CT scan showed multiple cysts in the liver and it was thought he had isolated liver cystic disease. Lytic pelvic lesions were identified 1 year later on CT and a biopsy of one of these lesions confirmed a well differentiated neuroendocrine tumour. A presacral mass was identified in follow up scans suggestive of a presacral mass with malignant transformation. Biopsy of the mass and the liver confirmed the presence of a neuroendocrine tumour. Chemotherapy was ineffective and he subsequently died. Autopsy confirmed tumour invasion of the liver, pancreas, kidneys and mesenteric lympth nodes. The pre-sacral mass was comprised of neoplastic tissue, teratoma, muscle and fibroadipous tissue.
A 4 year old girl was reported with Currarino syndrome who developed an ectopic nephroblastoma and pulmonary metastases from a presacral mass (Martucciello G 2004). She died despite chemo and radiotherapy.
A 2 year old girl developed a malignant teratoma from a pre-existing presacral mass (Cretolle et al 2006). She was well following chemotherapy.
A 3 year old girl was reported with a malignant tumour arising within a teratoma (Sen et al 2006). Histology revealed the presence of two components; residual mature teratoma and a necrotic malignant tumour resembling a primitive peripheral neuroectodermal tumour. A chemotherapy regimen according to the Euro Ewing 99 protocol was given in addition to radiotherapy. She remained well 8 months following her treatment.
One report of leiomyosarcoma in a grandmother of an affected child with Currarino has been reported (Norum 1991).
In addition, a further case of Leiomyomatosis peritonealis disseminate (LPD) in a 27 year old female with Currarino syndrome has been described in the literature (Nappi 2006). Malignant degeneration of LPD to a leiomyosarcoma has been reported but is rare.
A sacrococcygeal teratoma has been described with a deletion of chromosome 7q, partial trisomy 2p. Malignant degeneration had not occurred and the child died from post-operative complications (Le Caignec 2003). This teratoma was a posterior teratoma and similar to the type often identified antenatally. It is thought to have a different genetic basis to the teratomas seen in the Currarino syndrome. No mutations in the HLXB9 gene have been found in such cases. It is therefore interesting that this child had deleted the HLXB9 gene suggesting some genetic relationship between both conditions.
In classical Currarino syndrome the pre-sacral mass tends to be anterior to the sacrum. The sacrococcygeal teratomas have appeared to be a different group in that they present in the posterior coccygeal region, are often sporadic, occur more commonly in females and commonly become malignant. Mutational analysis of the HLXB9 gene has been negative in a number of cases. This recent case is interesting as it does suggest that HLXB9 is involved in some cases of posterior teratomas (the gene was shown to be deleted in this case) in some cases.
Treatment
Symptomatic: those children who present at birth with imperforate anus will require emergency surgery. Surgery for the pre-sacral mass is usually advised. The mass can cause an obstruction or other symptoms such as ascending meningitis if an enteric fistula is present. There is also the risk of malignant transformation. Treatment of the mass if it undergoes malignant transformation involves both a surgical and chemothereputic approach. Bleomycin, etoposide and carboplatin are now the gold standard chemotherapeutic treatments. Carboplatin has recently replaced cisplatin as it is less nephro and ototoxic.
Some patients may develop symptoms as a result of their renal or gynaecological malformation and these need to be managed appropriately. Spinal cord tethering is recognized and may recur requiring long term surveillance. Many individuals suffer from chronic constipation. Patients with a pre-sacral mass, particularly those with an anterior meningocoele, can develop headaches if pressure is increased within the abdominal cavity from coughing or sneezing or by lying on their abdomens.
Some patients may develop symptoms as a result of their renal or gynaecological malformation and these need to be managed appropriately. Spinal cord tethering is recognized and may recur requiring long term surveillance. Many individuals suffer from chronic constipation. Patients with a pre-sacral mass, particularly those with an anterior meningocoele, can develop headaches if pressure is increased within the abdominal cavity from coughing or sneezing or by lying on their abdomens.
Evolution
In those children where this condition is diagnosed at birth, a good clinical plus radiological assessment (including pelvic MRI) will identify most of the malformations. Surgery should be considered if a presacral mass is present.
Many of the complications listed above can be avoided if appropriate action is taken in early childhood.
Many of the complications listed above can be avoided if appropriate action is taken in early childhood.
Prognosis
Generally very good. In some patients a colostomy bag is required for life. Some women can present symptoms from their gynaecological malformation and require assessment and occasionally treatment for this. Spinal cord tethering can be another debilitating problem that can recur despite treatment.
If malignant transformation occurs chemotherapy can be effective but recurrences can occur which are resistant to treatment. There are no case reports recording a long remission but as the case reports are so few it is not possible to give definitive prognostic indicators. People have reported recurrence of a teratoma and therefore it is possible that malignancy could occur even if surgery has removed a presacral teratoma. However, the malignancy risk, which is already very small must be reduced even further by elective removal of any pre-sacral mass.
If malignant transformation occurs chemotherapy can be effective but recurrences can occur which are resistant to treatment. There are no case reports recording a long remission but as the case reports are so few it is not possible to give definitive prognostic indicators. People have reported recurrence of a teratoma and therefore it is possible that malignancy could occur even if surgery has removed a presacral teratoma. However, the malignancy risk, which is already very small must be reduced even further by elective removal of any pre-sacral mass.
Cytogenetics
Note
Cytogenetic analysis will be normal in most cases. However, some cases are associated with a 7q36 deletion or a translocation involving this region. It is worth considering FISH analysis of 7q36 in cases where Currarino syndrome is associated with developmental delay.
Cancer cytog
There is no information on the cytogenetic analysis of the pre-sacral tumours.
Genes involved and Proteins
Alias
HLXB9 HB9
Note
HLXB9 is a Homeobox gene. Proteins encoded by homeobox-containing genes are sequence-specific DNA binding proteins implicated in the control of gene expression in both developing and adult tissues.
Description
The HLXB9 gene has 3 exons. The phenotype results from haploinsufficiency of this gene.
Expression
In humans, studies have shown expression in the basal plate of the spinal cord and hindbrain and in the pharynx, oesophagus, stomach and pancreas and lymphoid tissue. Significant spatial and temporal expression differences were evident when expression of the gene in human and mouse were compared which may explain the phenotypic disparities observed between the two species.
Tailbud expression has been noted in Xenopus laevis. Studies on human embryos demonstrated expression in the sacral region during embryogenesis albeit predominantly in the anterior horn regions of the spinal cord. In adults the HLXB9 gene is predominantly expressed in the pancreas.
A mouse model of the Currarino triad demonstrated that abnormal differentiation of the tail bud mesenchyme led to defects of the tailgut and neural tube. Etretinate (the teratogenic agent used) disrupted secondary neurulation and resulted in malformations resembling the Currarino triad. The cloacal plate appeared to play a critical role in the development of the anorectum and deficiencies of this caused anorectal malformation.
Tailbud expression has been noted in Xenopus laevis. Studies on human embryos demonstrated expression in the sacral region during embryogenesis albeit predominantly in the anterior horn regions of the spinal cord. In adults the HLXB9 gene is predominantly expressed in the pancreas.
A mouse model of the Currarino triad demonstrated that abnormal differentiation of the tail bud mesenchyme led to defects of the tailgut and neural tube. Etretinate (the teratogenic agent used) disrupted secondary neurulation and resulted in malformations resembling the Currarino triad. The cloacal plate appeared to play a critical role in the development of the anorectum and deficiencies of this caused anorectal malformation.
Function
The HLXB9 gene functions as a transcription factor regulating gene expression in both developing and adult tissues. Little is known about target genes or protein partners.
Homology
The HLXB9 sequence shows up to 96%, 91% and 80% identity to orthologues in mouse, chick and Xenopus respectively. Sequence homology to Drosphilia homeobox genes and to the human homeobox gene PDX-1 suggest that some mutations within the HLXB9 homeodomain may alter the DNA binding specificity while other mutations may reduce nuclear translocation of the mutated protein.
Description
There is a polyalanine region consisting of 16 alanines. There are population differences in the polyalanine domain but little correlation between the presence of disease or the variable penetrance of disease and the number of GCC repeats in patients.
Note
Nonsense, frameshift and missense mutations have been identified. Most mutations are located within exon 1 and the homeodomain. Most missense mutations are clustered in the homeodomain whereas nonsense and frameshift mutations are mostly on the NH2 terminus of the protein. There is poor genotype phenotype correlation. Reduced penetrance has also been recorded by several groups. It appears, from in vitro assays that the missense mutations disrupt the binding of the homeodomain to the target DNA motif TAAT.
Whilst mutations have been identified in almost all the familial cases, it appears that mutations are only found in 30% of sporadic cases. Somatic mosaicism is most favoured explanation. It is also possible that mutations might be present outside of the coding region and genetic heterogeneity remains a possiblity.
Whilst mutations have been identified in almost all the familial cases, it appears that mutations are only found in 30% of sporadic cases. Somatic mosaicism is most favoured explanation. It is also possible that mutations might be present outside of the coding region and genetic heterogeneity remains a possiblity.
Germinal
There have been no recorded cases of germ line mosaicism.
Somatic
There have been no recorded cases of somatic mosaicism.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 4418917 | 1974 | Hereditary presacral teratoma. | Ashcraft KW et al |
| 17352395 | 2007 | Currarino syndrome shown by prenatal onset ventriculomegaly and spinal dysraphism. | Crétolle C et al |
| 6789651 | 1981 | Triad of anorectal, sacral, and presacral anomalies. | Currarino G et al |
| 16254195 | 2006 | Population differences in the polyalanine domain and 6 new mutations in HLXB9 in patients with Currarino syndrome. | Garcia-Barceló M et al |
| 10749657 | 2000 | Mutation analysis and embryonic expression of the HLXB9 Currarino syndrome gene. | Hagan DM et al |
| 830336 | 1977 | Radiography of hereditary presacral teratoma. | Hunt PT et al |
| 11528505 | 2001 | Spectrum of mutations and genotype-phenotype analysis in Currarino syndrome. | Köchling J et al |
| 10415286 | 1999 | Urodynamic evaluation in boys treated for posterior urethral valves. | Lal R et al |
| 14663834 | 2003 | Prenatal diagnosis of sacrococcygeal teratoma with constitutional partial monosomy 7q/trisomy 2p. | Le Caignec C et al |
| 12682745 | 2003 | Sharing of the same embryogenic pathway in anorectal malformations and anterior sacral myelomeningocele formation. | Liu Y et al |
| 10922380 | 2000 | Autosomal dominant sacral agenesis: Currarino syndrome. | Lynch SA et al |
| 15359381 | 2004 | Currarino syndrome: proposal of a diagnostic and therapeutic protocol. | Martucciello G et al |
| 16686944 | 2006 | Leiomyomatosis peritonealis disseminata in association with Currarino syndrome? | Nappi C et al |
| 1777251 | 1991 | Incomplete Currarino syndrome with a presacral leiomyosarcoma. | Norum J et al |
| 2059799 | 1991 | Hereditary sacral agenesis with presacral mass and anorectal stenosis: the Currarino triad. | O'Riordain DS et al |
| 9843207 | 1998 | A homeobox gene, HLXB9, is the major locus for dominantly inherited sacral agenesis. | Ross AJ et al |
| 16685735 | 2008 | Familial Currarino syndrome presenting with peripheral primitive neuroectodermal tumour arising with a sacral teratoma. | Sen G et al |
| 15216552 | 2004 | Malignant degeneration of presacral teratoma in the Currarino anomaly. | Urioste M et al |
| 10594140 | 2000 | "Teratomas" in the Currarino triad: a misnomer. | Weinberg AG et al |
| 6822928 | 1983 | Anterior sacral defects: an autosomal dominantly inherited condition. | Yates VD et al |
External Links
Citation
Sally Ann Lynch
Currarino syndrome
Atlas Genet Cytogenet Oncol Haematol. 2007-06-01
Online version: http://atlasgeneticsoncology.org/cancer-prone-disease/10082/currarino-syndrome
