Frasier syndrome (FS)
2007-05-01 Mariana M Cajaiba , Miguel Reyes-Múgica AffiliationProgram of Pediatric, Developmental Pathology, Yale University School of Medicine, 430 Congress Avenue, New Haven, CT 06520-8023, USA (MR-M)
Identity
Name
Frasier syndrome (FS)
Inheritance
Sporadic occurrence, with possible cases of autosomal dominant inheritance
Omim
136680
Mesh
D052159
Orphanet
347 Frasier syndrome
Umls
C0950122
Clinics
Phenotype and clinics
- Exceptional cases in younger children; youngest example at 6 months of age.
- XY karyotype:
- Streak (dysgenetic) gonads with gonadoblastoma.
- Normal external female genitalia; clitoris enlargement and ambiguous genitalia may be present.
- Small uterus (often with an inactive\/atrophic endometrium) and fallopian tubes.
- Focal and segmental glomerulosclerosis; in later stages of renal disease, only chronic, nonspecific findings may be present in kidney biopsy.
- Normal and functioning female genitalia.
- Clinically present only with renal disease.
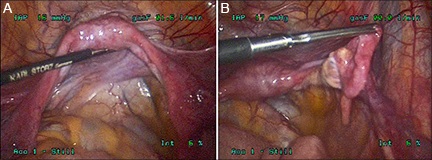
Figure 1. Surgical appearance of the internal genitalia in a patient with Frasier syndrome. A) Small but normally shaped uterus. B) A streak gonad is seen at the tip of the surgical instrument (courtesy of Dr. Masoud Azodi, Yale University School of Medicine).
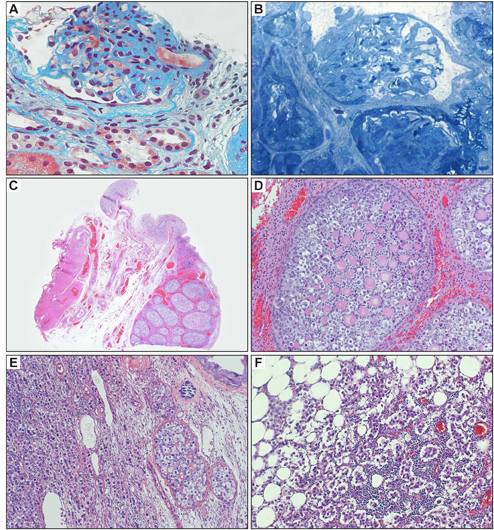
Figure 2. A-B: Kidney biopsy showing focal and segmental glomerulosclerosis (FSGS). A: Masson trichrome stain (400X). B: Semi-thin section stained with toluidine blue (400X). C-D: Gonadectomy specimens. C: Streak gonad with gonadoblastoma (H&E 40X). D: Gonadoblastoma nodule displaying Call-Exner body-like structures, surrounded by Leydig-like cells (H&E 200X). E-F: Histological aspect of dysgerminoma arising in gonadoblastoma. E: Gonadectomy specimen showing both, gonadoblastoma and dysgerminoma (H&E 100X). F: Peritoneal metastasis of dysgerminoma (H&E 200X).
Neoplastic risk
Gonadoblastomas are present in virtually all XY patients; usually bilateral.
Germ cell tumors (dysgerminomas) may arise from gonadoblastoma within dysgenetic gonads.
Wilms tumors are exceptional, and in these cases the diagnosis of FS is controversial (differential diagnosis with Denys-Drash syndrome; vide infra).
Germ cell tumors (dysgerminomas) may arise from gonadoblastoma within dysgenetic gonads.
Wilms tumors are exceptional, and in these cases the diagnosis of FS is controversial (differential diagnosis with Denys-Drash syndrome; vide infra).
Treatment
Prophylactic bilateral gonadectomy (may be laparoscopic if there is no evidence of overgrowth by a germ cell tumor); hysterectomy is not necessary.
The renal disease is usually steroid-resistant, requiring dialysis and renal transplantation.
Chemotherapy may be needed in cases with germ cell tumors.
In XY patients, menstruation can be induced with cyclic hormone replacement therapy; there are reported cases of successful pregnancy following in vitro fertilization procedures in these patients.
The renal disease is usually steroid-resistant, requiring dialysis and renal transplantation.
Chemotherapy may be needed in cases with germ cell tumors.
In XY patients, menstruation can be induced with cyclic hormone replacement therapy; there are reported cases of successful pregnancy following in vitro fertilization procedures in these patients.
Evolution
The end-stage renal disease is usually the major cause of morbidity in FS patients. The focal and segmental glomerulosclerosis progresses slowly (often for more than 10 years) and leads to terminal renal failure, requiring dialysis therapy and renal transplantation which can result in complications and increased morbidity. There are limited data regarding the clinical outcome after renal transplantation in these patients. The occurrence of germ cell neoplasia in FS patients can affect their prognosis. However, there is no evidence that FS-associated germ cell tumors have a different clinical outcome in comparison with sporadic tumors.
Genes involved and Proteins
Description
10 exons; spans approximately 50 kb. Encodes 4 zinc finger domains.
Transcription
Alternative splicing in two different sites (exons 5 and 9) leads to variable insertion of exon 5 and\/or insertion of 9 nucleotides in exon 9, resulting in transcription of four different isoforms.
Description
Transcription factor: contains 4 zinc finger domains.
Four different isoforms, ranging from 52 to 54 kDa (429-449 aminoacids).
Alternative splicing in exon 9: variable insertion of aminoacids lysine (K), threonine (T) and serine (S) between 3rd and 4th zinc fingers results in either +KTS or -KTS isoforms.
Four different isoforms, ranging from 52 to 54 kDa (429-449 aminoacids).
Alternative splicing in exon 9: variable insertion of aminoacids lysine (K), threonine (T) and serine (S) between 3rd and 4th zinc fingers results in either +KTS or -KTS isoforms.
Expression
During embryonal life, the WT1 protein is mainly expressed in the metanephros and developing kidney, gonadal ridges, coelomic surfaces, heart, spleen, liver, thymus, uterus and muscles of the abdominal wall.
An adequate ratio of +KTS\/-KTS expression is essential for the wild type function of WT1.
An adequate ratio of +KTS\/-KTS expression is essential for the wild type function of WT1.
Localisation
Nuclear (transcription factor function).
Function
WT1 functions mainly as a transcription factor, with many different downstream target genes; a post-transcriptional regulatory function of some target mRNAs has been also proposed.
In mammalian embryos, expression of the -KTS isoform induces gonadal ridge formation through proliferation of the coelomic epithelium, resulting in the bipotential gonad.
In XY individuals, expression of the +KTS isoform will activate the transcription of the SRY gene located on Y chromosome, which induces the expression of anti-mYllerian hormone by the developing Sertoli cells. Expression of the anti-mYllerian hormone in the developing testis results in formation of seminiferous cords, allowing sex-specific gonadal development, and regression of mYllerian structures (which give rise to the female genitalia).
During early kidney development in mammal embryos, the -KTS isoform promotes proliferation of the primordial mesenchyme, epithelial-mesenchymal interactions and ureteric bud branching. In later phases of kidney development, expression of +KTS leads to differentiation of podocytes and glomerular capillaries.
In mammalian embryos, expression of the -KTS isoform induces gonadal ridge formation through proliferation of the coelomic epithelium, resulting in the bipotential gonad.
In XY individuals, expression of the +KTS isoform will activate the transcription of the SRY gene located on Y chromosome, which induces the expression of anti-mYllerian hormone by the developing Sertoli cells. Expression of the anti-mYllerian hormone in the developing testis results in formation of seminiferous cords, allowing sex-specific gonadal development, and regression of mYllerian structures (which give rise to the female genitalia).
During early kidney development in mammal embryos, the -KTS isoform promotes proliferation of the primordial mesenchyme, epithelial-mesenchymal interactions and ureteric bud branching. In later phases of kidney development, expression of +KTS leads to differentiation of podocytes and glomerular capillaries.
Note
Most of the WT1 gene mutations in FS are located in positions 2, 4, 5 or 6 of the second splice donor site in intron 9.
These mutations lead to a decrease in the +KTS isoform, affecting the zinc fingers DNA binding affinity.
A decrease in +KTS is in keeping with the phenotype observed in FS, in which there seems to be a defect in antimüllerian hormone expression resulting in abnormal genital development in XY individuals. Also, defective expression of this isoform could explain the glomerular lesion observed in FS.
These mutations lead to a decrease in the +KTS isoform, affecting the zinc fingers DNA binding affinity.
A decrease in +KTS is in keeping with the phenotype observed in FS, in which there seems to be a defect in antimüllerian hormone expression resulting in abnormal genital development in XY individuals. Also, defective expression of this isoform could explain the glomerular lesion observed in FS.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12592634 | 2003 | Surgical management and genotype/phenotype correlations in WT1 gene-related diseases (Drash, Frasier syndromes). | Auber F et al |
| 9398852 | 1997 | Donor splice-site mutations in WT1 are responsible for Frasier syndrome. | Barbaux S et al |
| 15211353 | 2004 | One tissue, two fates: molecular genetic events that underlie testis versus ovary development. | Brennan J et al |
| 16009184 | 2005 | Successful pregnancy in a gonadectomized woman with 46,XY gonadal dysgenesis and gonadoblastoma. | Chen MJ et al |
| 10505699 | 1999 | Frasier syndrome: a cause of focal segmental glomerulosclerosis in a 46,XX female. | Demmer L et al |
| 14149008 | 1964 | GONADOBLASTOMA ASSOCIATED WITH PURE GONADAL DYSGENESIS IN MONOZYGOUS TWINS. | FRASIER SD et al |
| 17378674 | 2008 | Expanding the clinical spectrum of Frasier syndrome. | Gwin K et al |
| 1658787 | 1991 | Alternative splicing and genomic structure of the Wilms tumor gene WT1. | Haber DA et al |
| 9499425 | 1998 | Frasier syndrome is caused by defective alternative splicing of WT1 leading to an altered ratio of WT1 +/-KTS splice isoforms. | Klamt B et al |
| 3130865 | 1987 | Chronic renal failure and XY gonadal dysgenesis: "Frasier" syndrome--a commentary on reported cases. | Moorthy AV et al |
| 11065340 | 2000 | Wilms' tumor suppressor gene WT1: from structure to renal pathophysiologic features. | Mrowka C et al |
| 16110318 | 2005 | Wilms' tumour: connecting tumorigenesis and organ development in the kidney. | Rivera MN et al |
| 11595161 | 2001 | WT1 proteins: functions in growth and differentiation. | Scharnhorst V et al |
| 15973330 | 2005 | Frasier syndrome comes full circle: genetic studies performed in an original patient. | Wang NJ et al |
External Links
Citation
Mariana M Cajaiba ; Miguel Reyes-Múgica
Frasier syndrome (FS)
Atlas Genet Cytogenet Oncol Haematol. 2007-05-01
Online version: http://atlasgeneticsoncology.org/cancer-prone-disease/10035/frasier-syndrome-(fs)
