Schinzel-Giedion midface retraction syndrome
2006-04-01 Elizabeth McPherson AffiliationMedical Genetics Services, Marshfield Clinic, Marshfield, WI 54449, USA
Identity
Name
Schinzel-Giedion midface retraction syndrome
Alias
Schinzel-Giedion syndrome
Note
The use of the long form of the name, Schinzel-Giedion midface retraction syndrome, is preferred to prevent confusion with Schinzel ulnar-mammary syndrome, a completely unrelated and clinically non-overlapping condition also described by Dr Schinzel.
Inheritance
Schinzel-Giedion midface retraction syndrome is presumed to be inherited as autosomal recessive on the basis of several pairs of affected sibs with normal parents. No specific gene or chromosome region has been identified. Among 44 reported families there are only 2 well-documented affected sib pairs and 2 more pairs in which a previous deceased sib was reported to have had similar anomalies. Therefore alternative hypotheses such as an autosomal dominant mutation, microdeletion, or microduplication with the few recurrence being explained by parental gonadal mosaicism must also be considered.
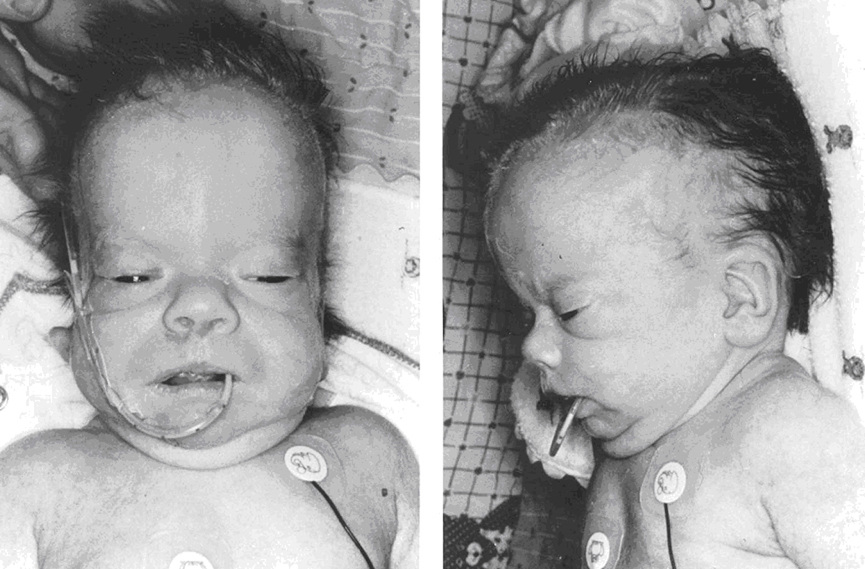
Frontal view of an infant with Schinzel-Giedion midface retraction Syndrome.
Note the rather coarse face with tall forehead, widely patent metopic suture, hypertelorism, shallow orbits, infraorbital folds and anteverted nares. Ear lobes are prominent.
Lateral views of an infant with Schinzel-Giedion midface retraction Syndrome.
Note the midface hypoplasia and lowest, posteriorly rotated ear with prominent lobules.
Omim
269150
Mesh
C536632
Orphanet
798 Schinzel-Giedion syndrome
Umls
C0265227
Clinics
Phenotype and clinics
The craniofacial appearance is essential to the diagnosis. Hydronephrosis is present in over 90% of cases. Other features, particularly skeletal findings, are helpful, but less specific. No generally accepted diagnostic guidelines exist.
Craniofacies: In addition to the pathognomonic \"midface retraction\" consisting of shallow orbits and midface hypoplasia with resulting prominence of the forehead, all reported patients have high forehead, large fontanelles and widely patent cranial sutures, particularly the metopic suture. The facial appearance is often described as coarse. Hypertelorism, flat nasal bridge, anteverted nares and lowset ears with protruding lobules are frequent. Nearly half the patients have choanal stenosis.
Central nervous system: Mental deficiency is usually profound. Both hypotonia and spasticity are common and may occur at different times in the same patient. Seizures of varying types, often including infantile spasms, are reported in most patients. Central nervous system malformations, particularly agenesis of the corpus callosum are reported occasionally, but the major anomaly on cranial MRI is cerebral atrophy which has been documented to progress over time in many of the longer surviving patients.
Skeletal: The skull base is steep, short, and often sclerotic with wide occipital synchondrosis, and multiple wormian bones. The ribs are broad and the clavicles are long. There is frequent hypoplasia of the first ribs, pubis, and distal phalanges. There is mild mesomelic shortening with widening of distal femur and increased density and broad cortex of the long bones. Additional limb anomalies such as polydactyly and talipes are found in a significant minority of patients.
Urogenital: Hydronephrosis on prenatal ultrasound is often the first abnormality noted. A few patients have died neonatally from lung hypoplasia related to either oligohydramnios sequence or compression from massive dilated kidneys. Virtually all patients have renal anomalies, primarily hydronephrosis, but only a few require treatment for obstructive lesions. Most patients have varying degrees of genital hypoplasia with hypospadias, micropenis and occasional ambiguous genitalia in males and labial hypoplasia, hymenal atresia and occasional bicornuate uterus in females.
Cardiac: About 1\/3 of the patients have congenital heart disease, most commonly atrial septal defect. Ventricular septal defect, pulmonic stenosis, AV canal, and carctation have also been reported.
Dermatologic: Most patients have hypertrichosis, redundant nuchal skin, hypoplastic nipples, hyperconvex nails and hypoplastic dermal ridges. Facial hemangiomas are common.
Other: Alacrima, visual impairment, hearing loss, macroglossia. Gingival hyperplasia occurs in long-term survivors.
Craniofacies: In addition to the pathognomonic \"midface retraction\" consisting of shallow orbits and midface hypoplasia with resulting prominence of the forehead, all reported patients have high forehead, large fontanelles and widely patent cranial sutures, particularly the metopic suture. The facial appearance is often described as coarse. Hypertelorism, flat nasal bridge, anteverted nares and lowset ears with protruding lobules are frequent. Nearly half the patients have choanal stenosis.
Central nervous system: Mental deficiency is usually profound. Both hypotonia and spasticity are common and may occur at different times in the same patient. Seizures of varying types, often including infantile spasms, are reported in most patients. Central nervous system malformations, particularly agenesis of the corpus callosum are reported occasionally, but the major anomaly on cranial MRI is cerebral atrophy which has been documented to progress over time in many of the longer surviving patients.
Skeletal: The skull base is steep, short, and often sclerotic with wide occipital synchondrosis, and multiple wormian bones. The ribs are broad and the clavicles are long. There is frequent hypoplasia of the first ribs, pubis, and distal phalanges. There is mild mesomelic shortening with widening of distal femur and increased density and broad cortex of the long bones. Additional limb anomalies such as polydactyly and talipes are found in a significant minority of patients.
Urogenital: Hydronephrosis on prenatal ultrasound is often the first abnormality noted. A few patients have died neonatally from lung hypoplasia related to either oligohydramnios sequence or compression from massive dilated kidneys. Virtually all patients have renal anomalies, primarily hydronephrosis, but only a few require treatment for obstructive lesions. Most patients have varying degrees of genital hypoplasia with hypospadias, micropenis and occasional ambiguous genitalia in males and labial hypoplasia, hymenal atresia and occasional bicornuate uterus in females.
Cardiac: About 1\/3 of the patients have congenital heart disease, most commonly atrial septal defect. Ventricular septal defect, pulmonic stenosis, AV canal, and carctation have also been reported.
Dermatologic: Most patients have hypertrichosis, redundant nuchal skin, hypoplastic nipples, hyperconvex nails and hypoplastic dermal ridges. Facial hemangiomas are common.
Other: Alacrima, visual impairment, hearing loss, macroglossia. Gingival hyperplasia occurs in long-term survivors.
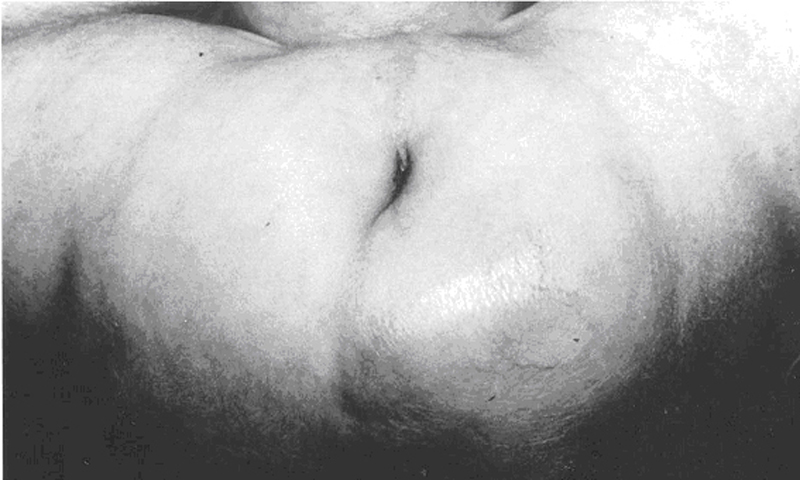
Gluteal area of same patient supine. Sacral asymmetry is due to a mass, which proved to be a primitive neurectodermal tumor.
Neoplastic risk
Among 46 reported patients, 7 (15%) have had childhood tumors including 3 sacrococcygeal teratomas, 2 primitive neurectodermal tumors arising in the sacral area, 1 hepatoblastoma, and 1 Wilms tumor arising in a multicystic dysplastic kidney. Both of the latter tumors arose in older children (above age 2), raising concern that long term survivors need to be followed for possible tumor development. Because of early deaths and lack of follow-up reports for many patients, the reported incidence of tumors must be considered a minimal estimate. Among 15 patients known to have survived beyond age 2, three (20%) have had tumors.
Treatment
Treatment is entirely symptomatic and directed at complications. The profound central nervous system involvement is the major cause of morbidity and mortality. Seizures are frequent and difficult to control with many patients failing to respond to anticonvulsants or ketogenic diet. Several patients with hearing loss have had some benefit from hearing aids. Hydronephrosis is usually stable, and only a few patients have required surgery for obstruction. Patients with tumors have responded to standard care including surgery and\/or chemotherapy.
Evolution
Facial features may become less characteristic in long-term survivors. Hydronephrosis rarely progresses to renal failure. Progressive gingival hyperplasia in older patients raises concern about a storage disease, but patients coming to autopsy do not have evidence of a lysosomal disorder. Unfortunately the neurologic status tends to deteriorate over time. Developmental delay is usually profound with most patients remaining non-ambulatory and non-verbal throughout their lives. Seizures can develop at any age and tend to become increasingly difficult to control. Patients who were hypotonic as neonates may develop severe spasticity. Follow-up MRI studies often show worsening cerebral atrophy and increasing ventriculomegaly without obstructive hydrocephalus.
Prognosis
Half of all reported patients were deceased at the time of the initial report. 20% died in the first year, another 17% in the second year and 13% between 2 and 10 years of age. Only 4 patients (
Cytogenetics
Note
Chromosome studies are normal in Schinzel-Giedion midface retraction syndrome. A single patient coincidentally had Klinefelter syndrome, which was not thought to contribute to the observed phenotype.
Cancer cytog
Tumor cytogenetic data are available in only one case, an infant with Schinzel-Giedion midface retraction and a neuroepithelial tumor in the sacral area. The infant had a normal 46, XY constitutional karyotype but the tumor contained a clone with duplication of the 17q22 region and a marker consisting of extra material from 17q {47,XY,dup?(17)(q22),+mar[5]-ishdup?(17)(q22)(wcp17+),der(17)(wcp17+)\/46,XY[35]}.
Other Findings
Note
None known. The Schinzel-Giedion midface retraction phenotype is suggestive of biochemical or storage disease but metabolic studies and organ histology at autopsy are not indicative of any unusual material being stored.
To be noted
Associations
http:\/\/www.schinzel-giedion.org Schinzel-Giedion Support Grouphttp:\/\/members.aol.com\/wjcufm\/patrick\/index2.htm Patricks Promise 14398 East Carroll Boulevard, Cleveland, Ohio USA 44118
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 7883373 | 1994 | Schinzel-Giedion syndrome. | Alavi S et al |
| 8849020 | 1995 | Schinzel-Giedion syndrome: report of two sibs. | Antich J et al |
| 8723563 | 1996 | Schinzel-Giedion syndrome: further delineation of the phenotype. | Elliott AM et al |
| 7086596 | 1982 | Congenital hydronephrosis, skeletal dysplasia, and severe developmental retardation: the Schinzel-Giedion syndrome. | Kelley RI et al |
| 11285076 | 2001 | A case of Schinzel-Giedion syndrome complicated with progressive severe gingival hyperplasia and progressive brain atrophy. | Kondoh T et al |
| 16409612 | 2005 | Malignant retroperitoneal tumor arising in a multicystic dysplastic kidney of a girl with Schinzel-Giedion syndrome. | Matsumoto F et al |
| 9738870 | 1998 | Sacral tumors in Schinzel-Giedion syndrome. | McPherson E et al |
| 8946113 | 1996 | Agenesis of the corpus callosum in Schinzel-Giedion syndrome associated with 47,XXY karyotype. | Ozkinay FF et al |
| 2184969 | 1990 | The Schinzel-Giedion syndrome. A case report and review of the literature. | Pul M et al |
| 7506484 | 1993 | New finding of Schinzel-Giedion syndrome: a case with a malignant sacrococcygeal teratoma. | Robin NH et al |
| 7864048 | 1994 | Schinzel-Giedion syndrome: autopsy report and additional clinical manifestations. | Rodríguez JI et al |
| 12847324 | 2003 | Schinzel-Giedion syndrome with sacrococcygeal teratoma. | Sandri A et al |
| 665725 | 1978 | A syndrome of severe midface retraction, multiple skull anomalies, clubfeet, and cardiac and renal malformations in sibs. | Schinzel A et al |
| 10051170 | 1999 | Schinzel-Giedion syndrome: evidence for a neurodegenerative process. | Shah AM et al |
External Links
Citation
Elizabeth McPherson
Schinzel-Giedion midface retraction syndrome
Atlas Genet Cytogenet Oncol Haematol. 2006-04-01
Online version: http://atlasgeneticsoncology.org/cancer-prone-disease/10129/schinzel-giedion-midface-retraction-syndrome
