Soft Tissues: Embryonal rhabdomyosarcoma
2009-01-01 Frederic G Barr Affiliation1.Department of Pathology, Laboratory Medicine, University of Pennsylvania School of Medicine, Philadelphia, PA 19104, USA
Summary
Note
Embryonal rhabdomyosarcoma (ERMS) refers to one subtype of the rhabdomyosarcoma family of soft tissue tumors. These are mesenchymal tumours related to the skeletal muscle lineage.
Classification
Classification
ERMS is one of two subtypes of rhabdomyosarcoma. The other major subtype is alveolar rhabdosarcoma (ARMS). Within the ERMS subtype, there are two histopathologic variants with superior outcome, botryoid and spindle cell. Another proposed histologic variant is anaplastic ERMS. There are no genetic subtypes of ERMS with clinical significance.
Clinics and Pathology
Embryonic origin
In many cases, ERMS occurs in regions without abundant or in some cases, regions without any detectable skeletal muscle. Therefore, the relationship of ERMS to skeletal muscle is not clear. However, it is postulated that ERMS is derived from mesenchymal precursors of mesodermal origin.
Epidemiology
ERMS accounts for 70-80% of all RMS tumors, and usually occurs in young children (median age of 6.5 years). ERMS represents ~4% of all malignancies among children and adolescents, and has an annual incidence of ~4 per million.
Clinics
ERMS often occurs in the head and neck region, genitourinary tract, and retroperitoneum. This tumor often presents as a painless mass, but in other cases, may be discovered from symptoms produced by compression of structures at the primary site. A small fraction of ERMS (10%) will have metastatic disease at the time of diagnosis, with the most frequent site of metastasis being the lungs. The standard treatment for ERMS is a combination of surgery, radiation, and intensive chemotherapy.
Pathology
The term embryonal RMS was coined to indicate the microcopic similarity of the tumor cells to developing skeletal myocytes. The tumors cells show variable myogenic differentiation, from small round cells to larger oblong cells with eosinophilic cytoplasm. In the most differentiated cells, there is a strap-like appearance, occasionally with cross striations and multinucleation. From an architectural standpoint, these tumors classically have variable cellularity, with hypercellular areas alternating with hypocellular areas containing a loose myxoid stroma.
Spindle cell RMS has dense whorls or bundles of spindle-shaped cells resembling smooth muscle. These tumors often occur in the paratesticular region of children and the head and neck region of adults. Botryoid RMS usually occurs in the lumen of a hollow internal organ, such as the urinary bladder or vagina. This form of RMS has the gross appearance of multiple polypoid nodules, and the microscopic appearance of a dense cambium layer of tumor cells under an intact epithelial surface.
Anaplasia is recognized by the presence of large, lobated hyperchromatic nuclei and atypical mitoses. Anaplastic cells can be found in a focal or diffuse distribution.
Spindle cell RMS has dense whorls or bundles of spindle-shaped cells resembling smooth muscle. These tumors often occur in the paratesticular region of children and the head and neck region of adults. Botryoid RMS usually occurs in the lumen of a hollow internal organ, such as the urinary bladder or vagina. This form of RMS has the gross appearance of multiple polypoid nodules, and the microscopic appearance of a dense cambium layer of tumor cells under an intact epithelial surface.
Anaplasia is recognized by the presence of large, lobated hyperchromatic nuclei and atypical mitoses. Anaplastic cells can be found in a focal or diffuse distribution.
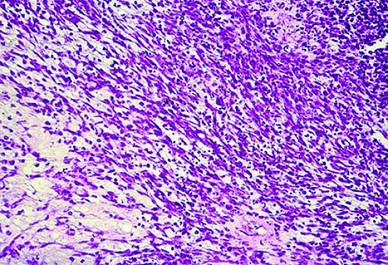
Histopathology of ERMS (hematoxylin-eosin, original magnification: 100X; courtesy of Dr. Linda Ernst).
Prognosis
Patients with ERMS tumors have a better outcome than patients with ARMS tumors. The 4-year failure free survival rates for patients with localized and metastastic ARMS are 85% and 35%, respectively. Other risk factors that influence outcome of ERMS include age, primary site, size of primary tumor, extent of local spread, and the presence of nodal and distal metastases. Based on these other factors, patients with ERMS tumors with limited extent of disease or in a favorable site (orbit, superficial head and neck, biliary tree, vagina, and paratestis) have a 85-95% probability of long-term survival. It should be also noted that though metastastic disease is a poor prognostic sign, patients with metastatic ERMS under 10 years of age have a survival rate of 40-50%, thus far exceeding the survival rate for metastatic ARMS.
Cytogenetics
Cytogenetics morphological
In contrast to the recurrent chromosomal translocations found in ARMS, ERMS does not have recurrent structural chromosome rearrangements, but rather has frequent chromosome gains. The most notable gains in ERMS were chromosomes 2, 8, 11, 12, 13, and 20.
Comparative genomic hybridization studies indicated that usual ERMS tumors had a low frequency of amplification (6-10%) but anaplastic ERMS tumors had a higher frequency of amplification (67%).
Comparative genomic hybridization studies indicated that usual ERMS tumors had a low frequency of amplification (6-10%) but anaplastic ERMS tumors had a higher frequency of amplification (67%).
Cytogenetics molecular
There is frequently a loss of one of the two parental alleles at one or more contiguous chromosomal 11 loci in the tumor cells. The smallest chromosomal region of consistent allelic loss is 11p15.5. Multiple genes are present in this region and demonstrate expression that is epigenetically regulated in a parent-of-origin-specific fashion by genomic imprinting. It is postulated that one allele of a tumor suppressor gene in this region is physiologically inactivated by imprinting and the second allele is removed by the allelic loss event, and thus both alleles of the tumor suppressor are inactivated to promote an oncogenic effect.
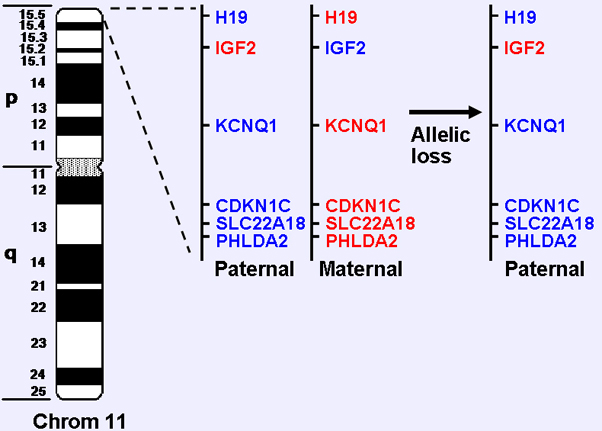
Allelic loss of imprinted region at 11p15.5 in ERMS. In the 11p15.5 chromosomal region, there is parent-of-origin-specific expression (imprinting) of multiple genes, such that red indicates an expressed allele and blue indicates an unexpressed allele. In many cases of ERMS and other tumors, the maternal alleles in the 11p15.5 region (and variable amounts of contiguous regions) are lost by one of a variety of genomic mechanisms in a process termed allelic loss, loss of heterozygosity, or conversion to homozygosity.
Genetics
Note
Though most cases of ERMS occur sporadically without an apparent genetic predisposition, a subset of ERMS occurs in association with the several genetic syndromes, including:Neurofibromatosis type I (NF1) Costello syndrome (HRAS)
Genes Involved and Proteins
Gene name
IGF2 (insulin like growth factor 2)
Location
11p15.5
Protein description
Growth factor.
Gene name
H19 (H19, imprinted maternally expressed transcript (non-protein coding))
Location
11p15.5
Protein description
Non-coding RNA.
Gene name
CDKN1C (cyclin dependent kinase inhibitor 1C)
Location
11p15.4
Protein description
Kinase inhibitor.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16170316 | 2005 | Germline mutations in HRAS proto-oncogene cause Costello syndrome. | Aoki Y et al |
| 12506174 | 2003 | Prognostic factors and clinical outcomes in children and adolescents with metastatic rhabdomyosarcoma--a report from the Intergroup Rhabdomyosarcoma Study IV. | Breneman JC et al |
| 11807989 | 2002 | Genomic gains and losses are similar in genetic and histologic subsets of rhabdomyosarcoma, whereas amplification predominates in embryonal with anaplasia and alveolar subtypes. | Bridge JA et al |
| 11408506 | 2001 | Intergroup rhabdomyosarcoma study-IV: results for patients with nonmetastatic disease. | Crist WM et al |
| 16010679 | 2005 | Tumor predisposition in Costello syndrome. | Gripp KW et al |
| 102756 | 1978 | Rhabdomyosarcoma complicating multiple neurofibromatosis. | McKeen EA et al |
| 17090187 | 2006 | Rhabdomyosarcomas in adults and children: an update. | Parham DM et al |
| 18985676 | 2008 | Prevalence and clinical impact of anaplasia in childhood rhabdomyosarcoma : a report from the Soft Tissue Sarcoma Committee of the Children's Oncology Group. | Qualman S et al |
| 3657988 | 1987 | Chromosomal localization of the human rhabdomyosarcoma locus by mitotic recombination mapping. | Scrable HJ et al |
| 12783965 | 2003 | Myogenin and MyoD1 expression in paediatric rhabdomyosarcomas. | Sebire NJ et al |
| 15127010 | 2004 | Neurofibromatosis in children with Rhabdomyosarcoma: a report from the Intergroup Rhabdomyosarcoma study IV. | Sung L et al |
| 9315099 | 1997 | Allelotype of pediatric rhabdomyosarcoma. | Visser M et al |
| 3338090 | 1988 | Chromosomal analysis of sixteen human rhabdomyosarcomas. | Wang-Wuu S et al |
| 8764111 | 1996 | Gains, losses, and amplification of genomic material in rhabdomyosarcoma analyzed by comparative genomic hybridization. | Weber-Hall S et al |
| 12170781 | 2002 | Molecular pathogenesis of rhabdomyosarcoma. | Xia SJ et al |
Citation
Frederic G Barr
Soft Tissues: Embryonal rhabdomyosarcoma
Atlas Genet Cytogenet Oncol Haematol. 2009-01-01
Online version: http://atlasgeneticsoncology.org/solid-tumor/5193/soft-tissues-embryonal-rhabdomyosarcoma
