Glycogen storage disease type I (GSD I)
2012-06-01 Alix Mollet Boudjemline , François Petit , Aurélie Hubert Buron , Pascale Trioche Eberschweiler , Vincent Gajdos , Philippe Labrune AffiliationAPHP, Centre de References Maladies Hereditaires du Metabolisme Hepatique, Service de pediatrie, Hopital Antoine Beclere, BP 405, 92141 Clamart cedex,, Univ Paris Sud (VG, PL), France
Identity
Name
Glycogen storage disease type I (GSD I)
Alias
von Gierke disease
Note
The disease comprises two sub-types: type Ia (glucose-6-phosphatase deficiency), type Ib (glucose-6-phosphate translocase deficiency).
Inheritance
Autosomal recessive. Incidence around 1\/100 000 births.
Omim
232200 , 232220 , 232240 , 232300 , 232400 , 232500 , 232600 , 232700 , 232800 , 240600 , 261750 , 306000 , 611556 , 613027
Mesh
D005953
Orphanet
364 Glycogen storage disease due to glucose-6-phosphatase deficiency
Umls
C0017920
Clinics
Note
Most of clinical manifestations are common to both sub-types of GSD I. Patients with type Ib have neutropenia.
Phenotype and clinics
Patients have poor tolerance to fasting (with hypoglycemia and hyperlactacidemia after 3 to 4 hours of fasting), marked hepatomegaly, full-cheeked round face, growth retardation (small stature and delayed puberty), generally improved by an appropriate diet, osteopenia and sometimes osteoporosis, enlarged kidneys and platelet dysfunctions leading to frequent epistaxis (Chen, 2000; Matern et al., 2002; Rake et al., 2002). In addition, in GSDIb, neutropenia and neutrophil dysfunction are responsible for tendency towards infections, relapsing aphthous gingivostomatitis, parodontitis, and enterocolitis. Late complications are hepatic (adenomas with rare but possible transformation into hepatocarcinoma (Talente et al., 1994; Labrune et al., 1997; Franco et al., 2005; Reddy et al., 2007) and renal (glomerular hyperfiltration leading to proteinuria and sometimes to renal insufficiency, stones) (Talente et al., 1994; Rake et al., 2002; Scales et al., 2010) rare cases of pulmonary hypertension have been reported (Humbert et al., 2002).
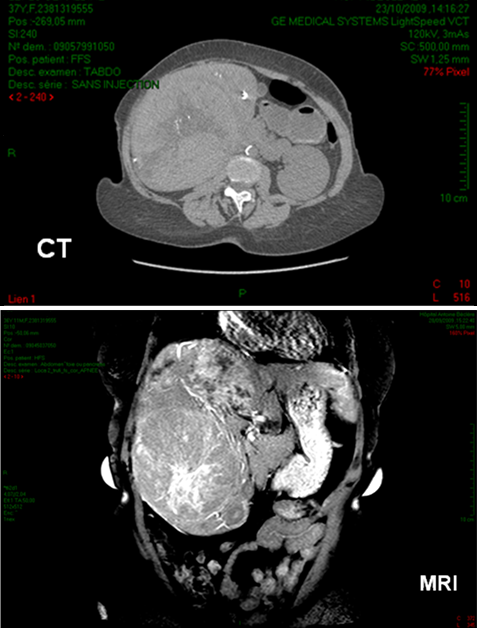
CT: liver tomodensitometry showing an important heterogeneous tumor (white arrows) - MRI: liver MRI illustrating the same hepatic tumor.
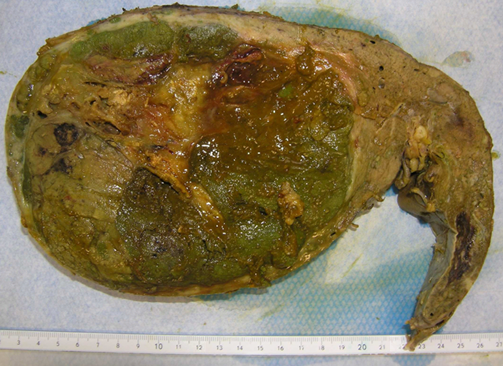
Liver after hepatectomy: picture of the liver after liver transplantation, showing hepatocarcinoma.
Neoplastic risk
Hepatocellular adenomas are at risk of malignant transformation, even though this risk is weak. Patients with such adenomas must be regularly followed-up, with clinical, biological, and MRI evaluations. Several studies have been performed to understand the physiopathology of adenomas development in GSD patients, and the transformation into hepatocellular carcinomas, but the precise mechanisms remain unknown (Kishnani et al., 2009).
Treatment
Diet is the basis of the treatment (Rake et al., 2002). It aims at avoiding hypoglycemia, combining, in infants, frequent meals and quite often nocturnal enteral feeding. Later, oral uncooked starch is introduced. Fructose and galactose intakes are restricted. Many patients are given allopurinol (hyperuricemia frequently occurs), fibrates and\/or statins (hypertriglyceridemia may have to be treated), converting enzyme inhibitors (should increased glomerular filtration rate and\/or microalbuminuria be detected) (Melis et al., 2005). G-CSF may be used in type Ib patients, to correct neutropenia. Liver transplantation, performed on the basis of poor metabolic control and\/or hepatocarcinoma, corrects hypoglycemia, but renal involvement may continue to progress and neutropenia is not always corrected in type Ib (Rake et al., 2002; Rake et al., 2002). Kidney transplantation can be performed in case of severe renal insufficiency. Combined liver-kidney grafts have been performed in a few cases.
Prognosis
Prognosis is usually good: late hepatic and renal complications may occur, however, with adapted management, patients have almost normal life span.
Genes involved and Proteins
Note
GSD type Ia. Not imprinted, maternally and paternally expressed.
Description
The human G6PC gene is 12,6 kb long and includes 5 coding exons (1071 bp for the coding region) (Lei et al., 1993).
Expression
The G6PC protein is constituted by 357 aminoacids and is expressed in liver, kidney and intestine. This protein is not expressed in neutrophils, explaining the absence of neutropenia in GSD type Ia.
Localisation
The protein is located in the endoplasmic reticulum membrane with its catalytic site on the internal side.
Function
G6PC hydrolyses glucose-6-phosphate to glucose in the endoplasmic reticulum. The enzyme forms with the glucose-6-phosphate transporter (SLC37A4) the complex responsible for glucose production and homeostatic regulation of blood glucose levels.
Somatic
The G6PC mutations are responsible for the glycogen storage disease type Ia (Von Gierke disease). More than 90 mutations have been described affecting the whole coding sequence (Matern et al., 2002; Shieh et al., 2002; Chou and Mansfield, 2008).
Note
GSD type Ib. Not imprinted, maternally and paternally expressed.
Description
The human SLC37A4 gene is 6,4 kb long and includes 9 coding exons (3870 bp for the coding region, exon 7bis being exclusively expressed in neutrophils (Hiraiwa et al., 1999; Veiga-da-Cunha et al., 1999).
Expression
The SLC37A4 protein is constituted of 1290 aminoacids and is co-expressed with G6PC gene in liver, kidney and intestine, and with G6PC3 in neutrophils (probably required for normal neutrophil function).
Localisation
The protein is located in the endoplasmic reticulum membrane.
Function
SLC37A4 transfers glucose-6-phosphate from cytoplasm to internal endoplasmic reticulum. This activity associated with glucose-6-phosphatase activity (G6PC or G6PC3) regulates glucose production from glycogenolysis and gluconeogenesis (Van Schaftingen and Gerin, 2002).
Somatic
The SLC37A4 mutations are responsible for the glycogen storage disease type Ib (Von Gierke disease). More than 80 mutations have been described affecting the whole coding sequence (Veiga-da-Cunha et al., 1999).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 9463334 | 1998 | The gene for glycogen-storage disease type 1b maps to chromosome 11q23. | Annabi B et al |
| 18449899 | 2008 | Mutations in the glucose-6-phosphatase-alpha (G6PC) gene that cause type Ia glycogen storage disease. | Chou JY et al |
| 15877204 | 2005 | Hepatocellular carcinoma in glycogen storage disease type Ia: a case series. | Franco LM et al |
| 10026167 | 1999 | Inactivation of the glucose 6-phosphate transporter causes glycogen storage disease type 1b. | Hiraiwa H et al |
| 12166582 | 2002 | Pulmonary arterial hypertension and type-I glycogen-storage disease: the serotonin hypothesis. | Humbert M et al |
| 19762333 | 2009 | Chromosomal and genetic alterations in human hepatocellular adenomas associated with type Ia glycogen storage disease. | Kishnani PS et al |
| 9138172 | 1997 | Hepatocellular adenomas in glycogen storage disease type I and III: a series of 43 patients and review of the literature. | Labrune P et al |
| 8211187 | 1993 | Mutations in the glucose-6-phosphatase gene that cause glycogen storage disease type 1a. | Lei KJ et al |
| 12373566 | 2002 | Glycogen storage disease type I: diagnosis and phenotype/genotype correlation. | Matern D et al |
| 15963056 | 2005 | Efficacy of ACE-inhibitor therapy on renal disease in glycogen storage disease type 1: a multicentre retrospective study. | Melis D et al |
| 12373584 | 2002 | Guidelines for management of glycogen storage disease type I - European Study on Glycogen Storage Disease Type I (ESGSD I). | Rake JP et al |
| 17637480 | 2007 | Resection of hepatocellular adenoma in patients with glycogen storage disease type Ia. | Reddy SK et al |
| 20092831 | 2010 | Stone forming risk factors in patients with type Ia glycogen storage disease. | Scales CD Jr et al |
| 11739393 | 2002 | The molecular basis of glycogen storage disease type 1a: structure and function analysis of mutations in glucose-6-phosphatase. | Shieh JJ et al |
| 8273986 | 1994 | Glycogen storage disease in adults. | Talente GM et al |
| 10482962 | 1999 | The putative glucose 6-phosphate translocase gene is mutated in essentially all cases of glycogen storage disease type I non-a. | Veiga-da-Cunha M et al |
| 11879177 | 2002 | The glucose-6-phosphatase system. | van Schaftingen E et al |
External Links
Citation
Alix Mollet Boudjemline ; François Petit ; Aurélie Hubert Buron ; Pascale Trioche Eberschweiler ; Vincent Gajdos ; Philippe Labrune
Glycogen storage disease type I (GSD I)
Atlas Genet Cytogenet Oncol Haematol. 2012-06-01
Online version: http://atlasgeneticsoncology.org/cancer-prone-disease/10071/glycogen-storage-disease-type-i-(gsd-i)/
