MET met proto-oncogene (hepatocyte growth factor receptor)
2001-10-01 Debora Angeloni , Michael L. Nickerson , Laura Schmidt AffiliationLaboratory of Immunobiology, National Cancer Institute, Frederick Cancer Research Facility Bldg. 560 Rm. 12 26 Frederick, MD 21702, USA
Identity
HGNC
LOCATION
7q31.2
LOCUSID
ALIAS
AUTS9,DFNB97,HGFR,RCCP2,c-Met
FUSION GENES
DNA/RNA
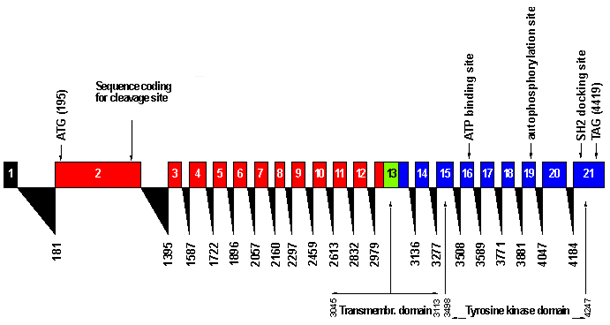
The coding sequence (4173 bp) is represented as red boxes (exons coding for the extracellular domain), blue boxes (sequence coding for the intracellular part) and a green box (coding for the transmembrane domain). Black triangles indicate the splicing sites. Several structural/functional elements are indicated, described in the text.
Description
The human MET gene spans more than 120 kb in length and consists of 21 exons separated by 20 introns . The size of the exons range from 81 bp (exon 16) to about 4kb (exon 21) The size of introns ranges from 0.1 to about 26 kb. The first exon encodes the 5 untranslated region (UTR) (394 bp) of the MET transcript and is separated by 26 kb circa from the second exon. Exon 2 is the largest coding exon, it contains14 bp of 5 UTR and encodes for 400 amino acids. The cleavage site that yields the a- and b-subunits from the precursor polypeptide is encoded by exon 2. Exon 13 contains the hydrophobic transmembrane domain, together with the end of the extracellular domain and the beginning of the intracellular domain. Exons 14 to 21 code for the intracellular part and exons 15 to 21 for the kinase domain. Exon 21 includes the region coding for the carboxy terminus of the protein and the large 3 UTR of the gene. The ATP binding site is encoded for by exon 16; the autophosphorylation site by exon 19; the SH2 docking site by exon 21.
Transcription
Alternative splicing: Multiple MET transcripts of different size were identified in human cell lines and tissues. At least three 8-kb variants were described and presumed to be generated by alternative splicing. A MET isoform was described that lacks 18 amino acids in the extracellular region (exon 10) and is the most abundant form in a variety of tissues and cell lines . Alternative splicing of exon 14 generates another variant that has an in-frame deletion of 47 amino acids in the juxtamembrane cytoplasmic domain of the receptor. A possible mechanism of alternative splicing could be at the origin of a 85 kDa, N-terminally truncated form of MET found in malignant musculo-skeletal tumors, although this short form could also originate from alternative transcription start or proteolitic cleavage.
Proteins
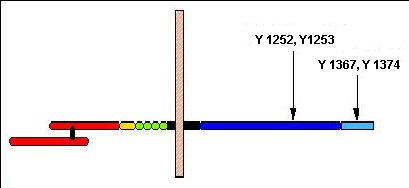
Red: SEMA domain. Yellow: PSI domain. Green: IPT domains. Black: transmembrane domain. Blue: tyrosine kinase domain. Light blue: C-terminal region. Pink: cell membrane. Y1252, Y11253: autophosphorylation site. Y1367, Y1374: docking site.
Description
The MET receptor (1408 amino acids) is initially synthesized as a partially glycosylated 170 kDa single chain precursor. Following further glycosylation, it is cleaved at a basic amino acid site into an a- and b-chain. The mature form is a transmembrane heterodimer composed of a- and b-chain linked together by disulphide bridges. The 50 kDa a-chain (amino acids 1-307) is located entirely extracellularly and contains the ligand binding pocket. The 140 kDa b-chain (amino acids 308-1408) comprises an extracellular part, the membrane spanning region and an intracellular C-terminal region that contains the tyrosine kinase domain. The C-terminal tail contains a conserved two-tyrosine multifunctional docking site that interacts with multiple SH2-containing intracellular signal transducers. The 5 alternative splicing leads to a form with 1390 amino acids.
Expression
MET and its ligand hepatocyte growth factor/scatter factor (HGF/SF) are expressed in numerous tissues although predominantly in cells of epithelial and mesenchymal origin, respectively. MET is amplified and overexpressed in many types of tumors, including tumors of the kidney (see below), thyroid, pancreas and osteosarcoma.
Localisation
MET is a transmembrane tyrosine kinase receptor. Upon ligand binding, the receptor is rapidly internalized, polyubiquitinated and degraded in a proteasome-dependent manner.
Function
The tyrosine kinase MET is the high affinity receptor for HGF/SF, a multifunctional cytokine. Upon ligand binding, MET dimerizes and transphosphorylates tyrosine residues in the C-terminal domain, which then interacts with members of a variety of signaling pathways. These include Grb-2 associated binder 1, phosphoinositide 3 kinase and c-Src. Under physiological conditions, MET-HGF/SF signaling has been shown to affect a wide range of biological activities depending on the cell target. These activities vary from cell proliferation (mitogenesis) to cellular shaping (morphogenesis) and motility (motogenesis). The coordination of these diverse activities constitutes a genetic program of invasive growth that allows branched morphogenesis (the formation of epithelial tubular structures), myoblast migration and neurite branching. MET/HGF cell targets comprise epithelial and mesenchymal cells, hematopoietic cells, myoblasts, spinal motor neurons. MET-HGF/SF signaling is also essential for normal development: mouse embryos carrying null mutations in both HGF alleles die in midgestation and show impaired liver formation.
Homology
MET amino acid sequence shows overall 33% identity with RON (the macrophage stimulating protein (MSP) receptor), which increases to 64% in the kinase domain. MET and RON are grouped in the family of so called scatter factor receptors. However, some structural domains shared by MET and RON are also present in other proteins such as semaphorins and plexins (semaphorins receptors), so that these proteins altogether are thought to originate from a common ancestor. The common domains are: the SEMA domain, a conserved box encompassing about 500 amino acids, with several highly conserved cysteines, and the PSI domain (from Plexin Semaphorins Integrines), of about 50 amino acids with 8 conserved cysteines, found also in integrins. Another structural motif repeated four times in the extracellular region of MET is the IPT domain, an immunoglobulin-like fold shared also by plexins and transcription factors. The function of these domains is at present under investigation but they are thought to mediate protein-protein interactions possibly related to cell dissociations and motility.
Mutations
Germinal
Germline mutations in the MET proto-oncogene are responsible for hereditary papillary renal carcinoma (HPRC) type 1 (see below). 10 known mutations are clustered in exons 16-19 of the tyrosine kinase domain and all are missense mutations which change the amino acid (V1110I, H1112R, H1112Y, M1149T, V1206L, V1238I, D1246N, Y1248C, Y1248D, M1268T). Mutations at four codons (V1110, D1246, Y1248, M1268) are homologous to sites of disease-associated activating mutations in other RTKs ( RET, c-kit, c-erbB. Two unrelated North American families have been identified with the H1112R mutation and shared flanking genotyping data, suggesting a founder effect. Most HPRC-associated MET mutations are constitutively activating, causing phosphorylation on tyrosine in a ligand-independent manner when transfected into NIH3T3 cells, and oncogenic when the mutant MET transfected cell lines are injected into nude mice. Other mutations with only weak transforming potential (Y1248C, L1213V) confer anchorage-independent growth and an invasive phenotype in transfected cells.
Somatic
Somatic missense mutations (H1112Y, H1112L, H1124D, L1213V, D1246H, Y1248H, Y1248C, M1268T) were identified in the TK domain of MET in 13% (17/129) of sporadic papillary renal carcinoma (PRC) type 1; 15 different germline and somatic MET mutations in 10 codons of the TK domain were identified in PRC. Infrequently, MET mutations have been identified in childhood hepatocellular carcinoma (3/10 in TK domain, T1191I, K1262R, M1268I), gastric carcinoma (1/85 in juxtamembrane domain, P1009S), glioma (1/11 in TK domain, G1137V;) and lymph node metastases (4/15 head and neck squamous cell carcinomas, Y1253D, Y1248C).
Implicated in
Entity name
Hereditary papillary renal carcinoma and Sporadic papillary renal carcinoma (HPRC, SPRC)
Disease
An inherited autosomal dominant form of renal carcinoma with reduced penetrance comprising 10% of all carcinomas of the kidney. Affected family members develop, in the fourth or fifth decade of life, bilateral, multifocal renal tumors with a papillary growth pattern which can be subdivided by histology into type 1 and type 2. Multiple tumors were shown to arise from independent clones. Sporadic PRCs are usually solitary tumors with microscopic papillary lesions in the surrounding renal parenchyma. It is important to note that patients with germline mutations in the MET gene are predisposed to develop papillary renal carcinoma type 1 specifically. These mutations are not known to predispose to any other type of malignant disease.
Prognosis
HPRC is a rare disease with an estimate incidence of 1 in 1 million. The age of development of PRCs is best studied in patients with H1112R mutation, because an elevated number of them has been described. By age 55, one half of H1112R mutation carriers have developed a detectable disease. Also, in H1112R mutation carriers has been observed that 26 % of patients develop distant metastases. The complete expression of the PRC phenotype may be affected by modifier genes. In fact, not only the age of onset varies among individuals but also the severity of the disease: in one patient were found at least 100 renal tumors, whereas his two brothers had a total of 1.
Cytogenetics
Papillary renal tumors are characterized by trisomy of chromosome 7 and 17, loss of Y in males. Only type 1 sporadic and hereditary PRCs harbor MET mutations with nonrandom duplication of the chromosome 7 bearing the mutant MET allele.
Hybrid gene
MET was first identified as the product of a human oncogene, tpr-MET, which derives from the fusion of two distinct genetic loci: tpr (translocated promoter region), which contributes two leucine zipper motifs (a protein-protein dimerization domain), and MET, which contributes the intracellular kinase domain of the MET receptor. The tpr gene sequence involved in the tpr-MET fusion is found on 1q25, though this gene is located at 1q31 in a database
Fusion protein
The tpr-MET chimeric protein is dimerized and activated in ligand-independent manner and therefore possesses constitutive kinase activity and transforming ability.
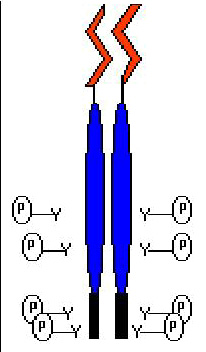
The hybrid protein is made of Tpr leucine zippers (red) that mediate constitutive dimerization - and MET kinase domains (blue), that drive transphosphorylation and constitutive activation.
Oncogenesis
MET/HGF signaling has been implicated in the generation and progression of a variety of tumors. The coexpression of wild type MET and HGF in the same cell (which generates an autocrine stimulatory loop) induces oncogenic transformation.
All investigated naturally occurring MET mutants show increased tyrosine phosphorylation level and enhanced kinase activity. In general, somatic mutations cause higher level of enzymatic activity compared to germline mutants. The kinase domain mutations cause ligand-independent activation and increase the tyrosine kinase activity. The juxtamembrane P1009I mutation does not cause ligand-independent activation but results in prolonged HGF-activated MET response.
The M1268T mutation identified in PRC homologous to the M918T mutation of RET in multiple endocrine neoplasia type 2b results not only in increased level of catalytic activity but also in a change of substrate specificity. Whereas c-Src is transiently activated by wild type MET, M1268T mutant MET is stably associated with c-Src, which is thereby constitutively phosphorylated and activated. Also, wild type MET does not phosphorylate substrate for cytosolic kinase c-Abl, whereas M1268T MET does. Moreover, expression of M1268T MET in NIH3T3 causes activation of the b-catenin pathway.
Different mutations act through distinct mechanisms and result in variable transforming activity. D1246H/N and M1268 mutants (that activate the Ras pathway) have high transforming ability. Y1248C and L1213V have less transforming ability but promote cell migration and invasion via activation of the PI3K/AKT pathway
3D molecular modeling studies using the crystal structure of the insulin receptor tyrosine kinase domain as a model suggest that disease-causing MET mutations interfere with the intrasteric mechanism of tyrosine kinase auto-inhibition to destabilize the inactive form and facilitate transition to the active form. Mutations V1110I, Y1248H/D/C, M1268T can affect contact between residues of the activation loop in its inhibitory conformation. Mutations M1149T and L1213V may increase flexibility of the tertiary structure. D1246N can stabilize the kinase in its active conformation. This structural work gives hints to understanding the switch of substrate specificity by mutant receptors.
All investigated naturally occurring MET mutants show increased tyrosine phosphorylation level and enhanced kinase activity. In general, somatic mutations cause higher level of enzymatic activity compared to germline mutants. The kinase domain mutations cause ligand-independent activation and increase the tyrosine kinase activity. The juxtamembrane P1009I mutation does not cause ligand-independent activation but results in prolonged HGF-activated MET response.
The M1268T mutation identified in PRC homologous to the M918T mutation of RET in multiple endocrine neoplasia type 2b results not only in increased level of catalytic activity but also in a change of substrate specificity. Whereas c-Src is transiently activated by wild type MET, M1268T mutant MET is stably associated with c-Src, which is thereby constitutively phosphorylated and activated. Also, wild type MET does not phosphorylate substrate for cytosolic kinase c-Abl, whereas M1268T MET does. Moreover, expression of M1268T MET in NIH3T3 causes activation of the b-catenin pathway.
Different mutations act through distinct mechanisms and result in variable transforming activity. D1246H/N and M1268 mutants (that activate the Ras pathway) have high transforming ability. Y1248C and L1213V have less transforming ability but promote cell migration and invasion via activation of the PI3K/AKT pathway
3D molecular modeling studies using the crystal structure of the insulin receptor tyrosine kinase domain as a model suggest that disease-causing MET mutations interfere with the intrasteric mechanism of tyrosine kinase auto-inhibition to destabilize the inactive form and facilitate transition to the active form. Mutations V1110I, Y1248H/D/C, M1268T can affect contact between residues of the activation loop in its inhibitory conformation. Mutations M1149T and L1213V may increase flexibility of the tertiary structure. D1246N can stabilize the kinase in its active conformation. This structural work gives hints to understanding the switch of substrate specificity by mutant receptors.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 10637762 | 1999 | Plexins, semaphorins, and scatter factor receptors: a common root for cell guidance signals? | Artigiani S et al |
| 9426201 | 1997 | "Invasive-growth" signaling by the Met/HGF receptor: the hereditary renal carcinoma connection. | Bardelli A et al |
| 10390613 | 1999 | Domains in plexins: links to integrins and transcription factors. | Bork P et al |
| 1846706 | 1991 | Identification of the hepatocyte growth factor receptor as the c-met proto-oncogene product. | Bottaro DP et al |
| 11433311 | 2001 | Pathway specificity for Met signalling. | Comoglio PM et al |
| 10579914 | 1999 | Plasminogen-related growth factor and semaphorin receptors: a gene superfamily controlling invasive growth. | Comoglio PM et al |
| 6590967 | 1984 | Molecular cloning of a new transforming gene from a chemically transformed human cell line. | Cooper CS et al |
| 11486025 | 2001 | Oncogenic mutants of RON and MET receptor tyrosine kinases cause activation of the beta-catenin pathway. | Danilkovitch-Miagkova A et al |
| 9195569 | 1997 | Papillary renal cell carcinoma: a clinicopathologic and immunohistochemical study of 105 tumors. | Delahunt B et al |
| 10734314 | 2000 | Somatic mutations of the MET oncogene are selected during metastatic spread of human HNSC carcinomas. | Di Renzo MF et al |
| 9380410 | 1997 | Gene structure of the human MET proto-oncogene. | Duh FM et al |
| 9715275 | 1998 | Duplication and overexpression of the mutant allele of the MET proto-oncogene in multiple hereditary papillary renal cell tumours. | Fischer J et al |
| 7691885 | 1993 | Identification of mutations in the coding sequence of the proto-oncogene c-kit in a human mast cell leukemia cell line causing ligand-independent activation of c-kit product. | Furitsu T et al |
| 10657996 | 2000 | Different point mutations in the met oncogene elicit distinct biological properties. | Giordano S et al |
| 8892055 | 1996 | Hepatocyte growth factor/scatter factor-Met signaling in tumorigenicity and invasion/metastasis. | Jeffers M et al |
| 9326629 | 1997 | Activating mutations for the met tyrosine kinase receptor in human cancer. | Jeffers M et al |
| 9001234 | 1997 | Degradation of the Met tyrosine kinase receptor by the ubiquitin-proteasome pathway. | Jeffers M et al |
| 10779338 | 2000 | Enhanced transformation by a plasma membrane-associated met oncoprotein: activation of a phosphoinositide 3'-kinase-dependent autocrine loop involving hyaluronic acid and CD44. | Kamikura DM et al |
| 8109322 | 1993 | Molecular cytogenetics of renal cell tumors. | Kovacs G et al |
| 7518457 | 1994 | Identification of a novel type of alternative splicing of a tyrosine kinase receptor. Juxtamembrane deletion of the c-met protein kinase C serine phosphorylation regulatory site. | Lee CC et al |
| 11042681 | 2000 | A novel germ line juxtamembrane Met mutation in human gastric cancer. | Lee JH et al |
| 9666114 | 1998 | The human hepatocyte growth factor receptor gene: complete structural organization and promoter characterization. | Liu Y et al |
| 10433944 | 1999 | Hereditary and sporadic papillary renal carcinomas with c-met mutations share a distinct morphological phenotype. | Lubensky IA et al |
| 10022866 | 1999 | The Gab1 PH domain is required for localization of Gab1 at sites of cell-cell contact and epithelial morphogenesis downstream from the met receptor tyrosine kinase. | Maroun CR et al |
| 11354004 | 2001 | Structural basis of oncogenic activation caused by point mutations in the kinase domain of the MET proto-oncogene: modeling studies. | Miller M et al |
| 7851906 | 1994 | Refined localization of the human TPR gene to chromosome 1q25 by in situ hybridization. | Miranda C et al |
| 11007037 | 2000 | Missense mutation of the MET gene detected in human glioma. | Moon YW et al |
| 10871851 | 2000 | Tumorigenesis mediated by MET mutant M1268T is inhibited by dominant-negative Src. | Nakaigawa N et al |
| 10685632 | 2000 | Expression of hepatocyte growth factor (HGF)/scatter factor and its receptor c-MET correlates with poor prognosis in synovial sarcoma. | Oda Y et al |
| 10417759 | 1999 | Novel mutation in the ATP-binding site of the MET oncogene tyrosine kinase in a HPRCC family. | Olivero M et al |
| 2423252 | 1986 | Mechanism of met oncogene activation. | Park M et al |
| 9927037 | 1999 | Somatic mutations in the kinase domain of the Met/hepatocyte growth factor receptor gene in childhood hepatocellular carcinomas. | Park WS et al |
| 7513258 | 1994 | A multifunctional docking site mediates signaling and transformation by the hepatocyte growth factor/scatter factor receptor family. | Ponzetto C et al |
| 8413267 | 1993 | Dimerization mediated through a leucine zipper activates the oncogenic potential of the met receptor tyrosine kinase. | Rodrigues GA et al |
| 8268192 | 1993 | Hepatocyte growth factor/scatter factor and its receptor, the c-met proto-oncogene product. | Rubin JS et al |
| 8958803 | 1996 | Expression of Met protein in thyroid tumours. | Ruco LP et al |
| 7824936 | 1995 | Activation of RET as a dominant transforming gene by germline mutations of MEN2A and MEN2B. | Santoro M et al |
| 10629560 | 1999 | Hepatocyte growth factor (HGF) stimulates tumour invasiveness in papillary carcinoma of the thyroid. | Scarpino S et al |
| 9140397 | 1997 | Germline and somatic mutations in the tyrosine kinase domain of the MET proto-oncogene in papillary renal carcinomas. | Schmidt L et al |
| 10327054 | 1999 | Novel mutations of the MET proto-oncogene in papillary renal carcinomas. | Schmidt L et al |
| 7854453 | 1995 | Placental defect and embryonic lethality in mice lacking hepatocyte growth factor/scatter factor. | Uehara Y et al |
| 10702398 | 2000 | Overexpression of the hepatocyte growth factor (HGF) receptor (Met) and presence of a truncated and activated intracellular HGF receptor fragment in locally aggressive/malignant human musculoskeletal tumors. | Wallenius V et al |
| 10027283 | 1999 | Semaphorin signaling: a little less per-plexin. | Yu HH et al |
| 10966853 | 2000 | Inherited epithelial tumors of the kidney: old and new diseases. | Zbar B et al |
| 7853572 | 1995 | Hereditary papillary renal cell carcinoma: clinical studies in 10 families. | Zbar B et al |
| 9709810 | 1998 | Inherited carcinomas of the kidney. | Zbar B et al |
| 8308957 | 1994 | Hereditary papillary renal cell carcinoma. | Zbar B et al |
| 9731534 | 1998 | Trisomy 7-harbouring non-random duplication of the mutant MET allele in hereditary papillary renal carcinomas. | Zhuang Z et al |
| 10818677 | 2000 | The hepatocyte growth factor/Met pathway in development, tumorigenesis, and B-cell differentiation. | van der Voort R et al |
Other Information
Locus ID:
NCBI: 4233
MIM: 164860
HGNC: 7029
Ensembl: ENSG00000105976
Variants:
dbSNP: 4233
ClinVar: 4233
TCGA: ENSG00000105976
COSMIC: MET
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA165946122 | crizotinib | Chemical | MultilinkAnnotation | associated | 22162641 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37429463 | 2024 | MET Fusions in NSCLC: Clinicopathologic Features and Response to MET Inhibition. | 5 |
| 37914903 | 2024 | Receptor tyrosine kinase gene expression profiling of orbital rhabdomyosarcoma unveils MET as a potential biomarker and therapeutic target. | 0 |
| 38430418 | 2024 | MET overexpression correlated with prognosis of EGFR-mutant treatment‑naïve advanced lung adenocarcinoma: a real‑world retrospective study. | 0 |
| 38480027 | 2024 | MET alterations in advanced non-small cell lung cancer. | 1 |
| 38650040 | 2024 | Clinical, pathologic, and genomic characteristics of two pediatric glioneuronal tumors with a CLIP2::MET fusion. | 0 |
| 38745318 | 2024 | PAX6 promotes neuroendocrine phenotypes of prostate cancer via enhancing MET/STAT5A-mediated chromatin accessibility. | 2 |
| 38836326 | 2024 | Spatiotemporal regulation of the hepatocyte growth factor receptor MET activity by sorting nexins 1/2 in HCT116 colorectal cancer cells. | 0 |
| 38909206 | 2024 | Deregulated immune cell recruitment orchestrated by c-MET impairs pulmonary inflammation and fibrosis. | 0 |
| 39000441 | 2024 | Differential Immune Checkpoint Protein Expression in HNSCC: The Role of HGF/MET Signaling. | 0 |
| 37429463 | 2024 | MET Fusions in NSCLC: Clinicopathologic Features and Response to MET Inhibition. | 5 |
| 37914903 | 2024 | Receptor tyrosine kinase gene expression profiling of orbital rhabdomyosarcoma unveils MET as a potential biomarker and therapeutic target. | 0 |
| 38430418 | 2024 | MET overexpression correlated with prognosis of EGFR-mutant treatment‑naïve advanced lung adenocarcinoma: a real‑world retrospective study. | 0 |
| 38480027 | 2024 | MET alterations in advanced non-small cell lung cancer. | 1 |
| 38650040 | 2024 | Clinical, pathologic, and genomic characteristics of two pediatric glioneuronal tumors with a CLIP2::MET fusion. | 0 |
| 38745318 | 2024 | PAX6 promotes neuroendocrine phenotypes of prostate cancer via enhancing MET/STAT5A-mediated chromatin accessibility. | 2 |
Citation
Debora Angeloni ; Michael L. Nickerson ; Laura Schmidt
MET met proto-oncogene (hepatocyte growth factor receptor)
Atlas Genet Cytogenet Oncol Haematol. 2001-10-01
Online version: http://atlasgeneticsoncology.org/gene/131/met-met-proto-oncogene-(hepatocyte-growth-factor-receptor)
