MEN1 (multiple endocrine neoplasia I)
1999-05-01 Alain Calender AffiliationService de genetique moleculaire et medicale, hopital Edouard-Herriot, batiment B7, 5, place dArsonval, 69437 Lyon 03, France
DNA/RNA
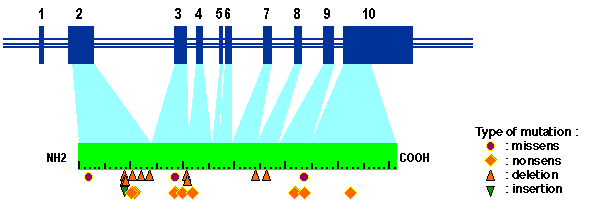
structure of the MEN1 gene (The European Consortium on MEN1, 1997)
Description
the MEN1 gene spans 9 Kb of the genome and is characterized by 10 exons; exon 1 and the 3 832 bp of exon 10 are untranslated. The figure shows the general structure of the gene and some of germline mutations in patients affected by inherited MEN1 disease
Transcription
a major 2,8 Kb transcript is detected in all tissues tested; a large 4,0 Kb mRNA has been characterized in the pancreas and in the thymus but the 5 structure of the MEN1 gene and the promoter region remain to date unknown; the 2,8 Kb major mRNA could be initiated inside exon 1
Proteins
Description
the MEN1 protein, menin, contains 610 amino-acids (67 Kda); contains two nuclear localization signals (NLS-1 and NLS-2) at the C-terminal end of the protein (exon 10), between amino-acids 479-497 for NLS-1 and 588-608 for NLS-2; this has been shown in vitro by deletion mutants construction with GFP-coexpressing vectors
Expression
menin is widely expressed and mainly in testis and central nervous system; murine equivalent to MEN1 has been cloned and most of the expression data have been confirmed in murine tissues, either in adults and during embryogenesis by RNA in situ experiments
Localisation
primarily localized in the nucleus and could translate in the cytoplasm during specific steps of the cell cycle
Function
the MEN1 gene is a growth-suppressor gene, as shown by allelic deletion (LOH) in tumoral DNA from MEN1 patients; menin has been showed to interact with the AP1 transcrition factor through his JunD component; this interaction involves mainly the first 40 amino-acids at the N-terminal end of menin and some specifics amino-acids in the central domain of the protein; Menin interacts specifically with JunD but with none of the other AP1 proteins, such as JunB, c-Jun, c-Fos and Fra1/2; among 11 missense mutations described in MEN1 patients, the authors reported that four of them decreased or abolished binding to JunD suggesting a separate domain between amino-acids residues 139 and 142 could have a critical role in menin-JunD interaction; using mammalian two-hybrid assays, menin has been shown to repress JunD-mediated transcriptionnal activation but most of menin mutatnts with impaired JunD-binding properties lossed this inhibitory activity; strikingly, overexpression of normal or mutant menin in similar experimental assays led to the absence of repressional activity suggesting that unknown factors could be involved in the menin-JunD interaction; new partners binding menin will be probably characterized in a near future and help us to understand the MEN1-related pathways
Homology
no homology has been found to date either by comparison of primary sequence and secondary/tertiary structure of this protein with all known proteins involved in cellular physiology
Mutations
Germinal
germline mutations in the MEN1 gene cause familial and sporadic multiple endocrine neoplasia type 1 (MEN1) and the majority of mutations described predict premature protein truncation either by nonsenses and frameshifts in coding sequences; missense mutations have been identified in ˜ 30% of cases and when characterized in sporadic cases, most of them need analysis of a large (> 50) number of control individuals in order to exclude frequent polymorphisms; interestingly, all truncating mutations affect one or both NLSs and no missense mutations were observed inside NLS-1 and NLS-2; mutations are spread over the gene and most of them occur once in a single family; some mutations were observed in more than one family and when a common ancestor was excluded by haplotyping, these recurrent mutations might be accounted for hot-spots in the MEN1 sequence; most recurrent mutations are nonsenses and frameshifts in exons 2 and 10; for example, single base deletion occurs frequently at nucleotide 1650 in exon 10 and has been related to the presence of an highly repetitive motif (CCCCCCCG) in this region inducing replication errors by slipped-strand mispairing; between 10 and 15% of sporadic MEN1 could be explained by de novo mutations, but this must be confirmed by an exhautive analysis of affected individuals and both parents
Implicated in
Entity name
Multiple Endocrine Neoplasia type 1 or Wermer Syndrome
Disease
an inherited autosomal dominant predisposition to endocrine tumors, including parathyroids, endocrine pancreas, pituitary, adrenal glands, and the diffuse neuroendocrine tissues deriving from foregut; non-endocrine tumors have been observed in some MEN1 patients, including ependymoma, meningioma, cutaneous angiofibroma and lipoma, melanoma and rare visceral lesions such as rhabdomyosarcoma and leiomyoma; MEN1 is highly penetrant and more than 90% of gene-carriers will present biological and/or clinical signs of the disease affter the fifth decade; around 5-10% of patients have an agressive disease before age 20
no genotype-phenotype correlation were found to date in MEN1; nevertheless, most families with agressive NET have truncating mutations either in exons 2, 3, 9 or 10 but no studies have been able to find statistical evidence of this putative correlation; recent investigations suggested that some MEN1 families could express only primary hyperparathyroidism, so called familial primary hyperparathyroidism (FIHPT), an allelic variant of MEN1; MEN1 related FIHPT appears as a benign disease but hyperplasia and/or adenoma occur in all parathyroid glands; recent data suggest that this variant could be associated to missense mutations in exons 4 to 7 of the MEN1 sequence; nevertheless, such correlations remain uncertain an do not have clinical implications in medical practice; the identification of germline missense mutations in exons 4 to 7 must lead to an extensive biological and clinical screening of patients in order to exclude the occurrence of pancreatic and pituitary disease, as recently shown in a typical MEN1 family carrying a Leu264Pro in exon 5; approximately 10-15% of MEN1 families do not show any mutation in the known part of MEN1 sequence; clinical profile in these families do not differ from that of families with identified mutations and it is therefore possible that MEN1 mutations occur outside the coding sequence; deletion of part or full MEN1 sequence has been also suggested as a rare mechanism of germline mutation
Prognosis
it is mainly related to metabolic and organic complications of hormonal hypersecretion by tumoral cells (Zollinger-Ellison syndrome induced by gastrinoma, hyperinsulinism, hyperparathyroidism, hyeperaldoseronism, Cushing syndrome, hyperprolactinemia, acromegaly; more than 30-50% of digestive neuroendocrine tumors and those localized in thymus and bronchi have a metastatic potential
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 9989505 | 1999 | Menin interacts with the AP1 transcription factor JunD and represses JunD-activated transcription. | Agarwal SK et al |
| 9463336 | 1998 | Characterization of mutations in patients with multiple endocrine neoplasia type 1. | Bassett JH et al |
| 1968641 | 1990 | Localization of the MEN1 gene to a small region within chromosome 11q13 by deletion mapping in tumors. | Byström C et al |
| 9666051 | 1998 | Genetic testing in multiple endocrine neoplasia and related syndromes. | Calender A et al |
| 10189986 | 1998 | [Clinicogenetic study of MEN1: recent physiopathological data and clinical applications. Study Group of Multiple Endocrine Neoplasia (GENEM)]. | Calender A et al |
| 9103196 | 1997 | Positional cloning of the gene for multiple endocrine neoplasia-type 1. | Chandrasekharappa SC et al |
| 9843042 | 1998 | Novel V184E MEN1 germline mutation in a Japanese kindred with familial hyperparathyroidism. | Fujimori M et al |
| 9683585 | 1998 | Germ-line mutation analysis in patients with multiple endocrine neoplasia type 1 and related disorders. | Giraud S et al |
| 9465067 | 1998 | Menin, the product of the MEN1 gene, is a nuclear protein. | Guru SC et al |
| 10092066 | 1999 | Nuclear/cytoplasmic localization of the multiple endocrine neoplasia type 1 gene product, menin. | Huang SC et al |
| 9510467 | 1998 | A large germline deletion of the MEN1 gene in a family with multiple endocrine neoplasia type 1. | Kishi M et al |
| 2894610 | 1988 | Multiple endocrine neoplasia type 1 gene maps to chromosome 11 and is lost in insulinoma. | Larsson C et al |
| 9215690 | 1997 | Identification of the multiple endocrine neoplasia type 1 (MEN1) gene. The European Consortium on MEN1. | Lemmens I et al |
| 9437237 | 1997 | Menin mutations in patients with multiple endocrine neoplasia type 1. | Mayr B et al |
| 9888389 | 1999 | Mutation analysis of the MEN1 gene in Belgian patients with multiple endocrine neoplasia type 1 and related diseases. | Poncin J et al |
| 9439676 | 1997 | Germline mutations of the MEN1 gene in Japanese kindred with multiple endocrine neoplasia type 1. | Shimizu S et al |
| 9824159 | 1998 | Characterization of the mouse Men1 gene and its expression during development. | Stewart C et al |
| 9792884 | 1998 | A family with isolated hyperparathyroidism segregating a missense MEN1 mutation and showing loss of the wild-type alleles in the parathyroid tumors. | Teh BT et al |
| 9709921 | 1998 | Mutation analysis of the MEN1 gene in multiple endocrine neoplasia type 1, familial acromegaly and familial isolated hyperparathyroidism. | Teh BT et al |
| 13138607 | 1954 | Genetic aspects of adenomatosis of endocrine glands. | WERMER P et al |
Other Information
Locus ID:
NCBI: 4221
MIM: 613733
HGNC: 7010
Ensembl: ENSG00000133895
Variants:
dbSNP: 4221
ClinVar: 4221
TCGA: ENSG00000133895
COSMIC: MEN1
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA446760 | Multiple Endocrine Neoplasia Type 1 | Disease | DataAnnotation, Literature, MultilinkAnnotation | associated | 23788249 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38609433 | 2024 | Combined deletion of MEN1, ATRX and PTEN triggers development of high-grade pancreatic neuroendocrine tumors in mice. | 0 |
| 38609433 | 2024 | Combined deletion of MEN1, ATRX and PTEN triggers development of high-grade pancreatic neuroendocrine tumors in mice. | 0 |
| 36795828 | 2023 | Menin "reads" H3K79me2 mark in a nucleosomal context. | 6 |
| 36842074 | 2023 | [Casuistic cases of parathyroid carcinoma with a verified mutation in the MEN1 gene]. | 0 |
| 36922589 | 2023 | MEN1 mutations mediate clinical resistance to menin inhibition. | 24 |
| 36925188 | 2023 | Multiple Endocrine Neoplasia Type 1 Syndrome Pancreatic Neuroendocrine Tumor Genotype/Phenotype: Is There Any Advance on Predicting or Preventing? | 2 |
| 36939378 | 2023 | MEN1 Degradation Induced by Neddylation and the CUL4B-DCAF7 Axis Promotes Pancreatic Neuroendocrine Tumor Progression. | 4 |
| 37078545 | 2023 | MENIN-mediated regulation of gastrin gene expression and its role in gastrinoma development. | 1 |
| 37307230 | 2023 | Blood-based Proteomic Signatures Associated With MEN1-related Duodenopancreatic Neuroendocrine Tumor Progression. | 2 |
| 37395406 | 2023 | MEN1 is a regulator of alternative splicing and prevents R-loop-induced genome instability through suppression of RNA polymerase II elongation. | 2 |
| 37437063 | 2023 | Evaluating the role of MEN1 gene expression and its clinical significance in breast cancer patients. | 2 |
| 37492626 | 2023 | Clinically Defined Mutations in MEN1 Alter Its Tumor-suppressive Function Through Increased Menin Turnover. | 0 |
| 37867522 | 2023 | Multiple endocrine neoplasia type 1 with Zollinger-Ellison syndrome: clinicopathological analysis of a Japanese family with focus on menin immunohistochemistry. | 0 |
| 38003662 | 2023 | Dependency of B-Cell Acute Lymphoblastic Leukemia and Multiple Myeloma Cell Lines on MEN1 Extends beyond MEN1-KMT2A Interaction. | 1 |
| 36795828 | 2023 | Menin "reads" H3K79me2 mark in a nucleosomal context. | 6 |
Citation
Alain Calender
MEN1 (multiple endocrine neoplasia I)
Atlas Genet Cytogenet Oncol Haematol. 1999-05-01
Online version: http://atlasgeneticsoncology.org/gene/148/men1-(multiple-endocrine-neoplasia-i)
