CTSL (cathepsin L1)
2009-01-01 Brigitte Goulet , Alain Nepveu AffiliationCancer research Laboratory Program, London Regional Cancer Program at London Health Science Centre, the University of Western Ontario, London, Canada (BG); Department of Biochemistry, Goodman Cancer Centre, McGill University, Montreal, Canada (AN)
Identity
HGNC
LOCATION
9q21.33
LOCUSID
ALIAS
CATL,CTSL1,MEP
FUSION GENES
DNA/RNA

Shown are the eight exons and seven intron of the human Cathepsin L1 gene. The black boxes correspond to protein coding.
Description
The human Cathepsin L1 gene comprises eight exons and seven introns, and span 5411 bases. The first AUG translation initiation site is located within exon 2. Three spliced variants of hCATL-A have been identified; hCATL-AI, hCATL-AII, hCATL-AIII (Rescheleit et al., 1996; Bakhshi et al., 2001; Aurora et al., 2002; Jean et al., 2002). These spliced forms lack 27 nucleotide (nt), 90 nt and 144 nt from the 3 end of exon 1 respectively and lead to mRNA species that differ in the 5 untranslated regions. However, they are translated into identical proteins. The shorter 5UTR lack secondary loops and are translated more effectively than hCATL-A (Aurora et al., 2002).
Transcription
One major transcription initiation site is situated at -290 from the starting AUG on the human cDNA sequence (Joseph et al., 1988; Chatham et al., 1993; Bakhshi et al., 2001; Aurora et al., 2002; Jean et al., 2002). This mRNA of 1.7 Kb corresponds to hCATL-A. Another mRNA, hCATL-B, is transcribed from another TATA-less promoter localized within the first intron of hCATL-A and encodes the same cathepsin L protein (Joseph et al., 1988; Bakhshi et al., 2001; Seth, gene 2003). The hCATL-B therefore differs from hCATL-A in the 5 untranslated region (Bakhshi et al., 2001; Jean et al., 2002). The transcription factors NF-Y, SP1 and SP3 have been shown to be responsible for more than 85% of Cathepsin L expression in melanoma cells (Jean et al., 2002). In melanoma and in lymphoma cells, Cathepsin L expression is also regulated by CpG methylation, and gene amplification has been observed in one melanoma cell line (Jean et al., 2006). In tissue culture models, phorbol esters, certain oncogenes such as ras, v-src, SV-40 Large T and raf, cytokines such as IL-1, IL-6 and TNF-alpha, and hypoxia have all been shown to induce cathepsin L expression (Troen et al., 1991; Lemaire et al., 1994; Kakegawa et al., 1995; Lemaire et al., 1997; Heinrich et al., 2000; Gerber et al., 2001; Jean et al., 2008).
Pseudogene
Using the program TBLASTN three pseudogenes, closely related to cathepsin L were identified on chromosome 10 (Rossi et al., 2004).
Proteins
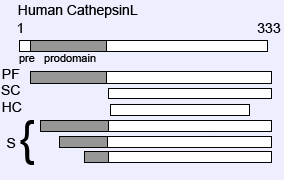
Shown is a schematic representation of the various human cathepsin L isoforms. The full-length protein is composed of a signal peptide (pre), a pro-domain and the mature polypeptide. PF stands for proform, SC for single chain, HC for heavy chain and S correspond to the various short isoforms that are devoid of signal peptide and whose translation is initiated from internal, in-frame, AUGs located within the prodomain coding sequence.
Description
CSTL1 codes for a protein of 333 amino-acids. Like all lysosomal enzymes, cathepsin L is translated as an inactive pre-pro-enzyme. The pre-region, located at the amino terminus of the protein, is a 17 amino-acid signal peptide (or signal sequence). Human cathepsin L is N-glycosylated at Asn 204. This glycosylation event is not required for proper folding of the protein or for its enzymatic activity or stability, but is important for lysosomal targeting (Smith et al., 1989; Kane, 1993). In certain cases, especially in transformed cells, secretion of procathepsin L (MEP) is observed (Gottesman, 1978). The crystal structure of procathepsin L has been resolved (Coulombe et al., 1996). Structurally, like most enzymes from the papain family, the mature cathepsin L consists of two globular regions, the R-domain (right) and L- domain (left) (Turk et al., 1997). These two domains are organized to form an open V-shaped active site cleft. The propiece of cathepsin L can also be separated into two regions. The amino-terminal part, which consists of the first 60 amino acids, is important for proper folding and glycosylation of procathepsin L (Chapman et al., 1997). Expression of the carboxy-terminal part of the prodomain is responsible for the inhibitory role of the propiece, by preventing the entry of the substrate into the active cleft. In order to do so, when the amino-terminus binds to the prodomain binding loop, the carboxy-terminus of the proregion bends over the groove of the active site in the opposite direction that the substrate would have been (Coulombe et al., 1996). The removal of the propiece occurs via an intra and/or inter molecular mechanism as the zymogen reaches the acidic environment of late endosomes or lysosomes (Nishimura et al., 1989; Salminen et al., 1990; Nomura et al., 1997; Ishidoh, et al., 2002). In order for the enzyme to become active, the proregion must be removed (Mason et al., 1992; Ishidoh et al., 1995). Studies have shown that other lysosomal enzymes, such as cathepsin D can also process procathepsin L and be involved in the initial steps of activation (Nishimura et al., 1989). In some cell types, the mature single chain form of cathepsin L is further processed into a two chains, heavy and light, by cleavage of the carboxy-terminus (Mason et al., 1985; Gal et al., 1986; Erickson, 1989). The optimal activity of mature cathepsin L requires a slightly acidic pH and a reducing environment which permits the active cysteine to be oxidized. The maximal activity of cathepsin L, using small synthetic peptides as substrates is at pH 5.5. The enzyme is most stable between pH 4.5 and 5.5. Like all cysteine proteases, the active site of cathepsin L is composed of a reactive cysteine (Cys 25), and a histidine (His 159). In the active form, both residues are charged, forming a thiolate-imidazolium ion pair (McGrath, 1999). Cathepsin L prefers a hydrophobic residue (mainly L/I) in the P2 position (cleavage occurs between residues P1 and P1) (Chapman et al., 1997). In addition, a shorter isoform of Cathepsin L has been detected. Translation at downstream, in-frame, AUGs is responsible for generating a protein that is devoid of a signal peptide which cannot be routed to the endoplasmic reticulum (Goulet et al., 2004; Goulet et al., 2007).
Localisation
Cathepsin L is ubiquitously expressed. It is generally localized to the endosomes/lysosomes or secreted. Recently, various groups have reported the presence of Cathepsin L in the nucleus and the cytoplasm in various cell types (Goulet et al., 2004; Bulynko et al., 2006; Varanou et al., 2006; Sever et al., 2007; Duncan et al., 2008). Moreover, in neuroendocrine chromaffin cell types, Cathepsin L is detected in regulatory secretory vesicules (Yasothornsrikul et al., 2003).
Function
Cathepsin L is a lysosomal enzyme originally thought to be involved in terminal protein degradation only. However, knockout mice showed that terminal protein degradation was not the work of a single cathepsin, as none of these mice had defects in protein degradation. The various phenotypes of these mice rather suggested that this protease has other specific biological roles. One knockout was generated, and two mice with natural mutations within the cathepsin L gene were also identified, Furless and Nackt. The furless mice possess a G-to-A mutation, which substitute an arginine for a glycine at position 149 of the cathepsin L protein sequence, resulting in an inactive enzyme (Roth et al., 2000). The nackt mice display a deletion in the cathepsin L gene, preventing the generation of any functional protein (Benavides et al., 2002). These three animal phenotypes have revealed that Cathepsin L plays a role in various physiological events in different tissues.
For example, Cathepsin L is important for epidermal homeostasis, regulation of the hair cycle, control of keratinocyte proliferation, MHC class-II mediated antigen presentation and selection of CD4+ T cells in cortical epithelial cells of the thymus (Nakagawa et al., 1998; Roth et al., 2000; Benavides et al., 2002; Reinheckel et al., 2005). Cathepsin L expression in thymocytes has been shown to be essential for natural killer cell development (Honey et al., 2002). Knockout mice models have demonstrated that Cathepsin L directly participates in atherosclerosis and in neovascularisation induced by endothelial progenitor cells (Maehr et al., 2005; Urbich et al., 2005; Kitamoto et al., 2007). These knockout mice have been observed to develop heart disease similar to human dilated cardiomyopathy (Stypmann et al., 2002). In general, pups lacking cathepsin L also have a slightly higher mortality upon weaning than their littermates (Reinheckel et al., 2001). Cathepsin L is responsible for the processing of viral proteins (Chandran et al., 2005; Pager et al., 2005; Kaletsky et al., 2007; Bosch et al., 2008) and the generation of neuropeptides and thyroid hormone (Funkelstein et al., 2008; Funkelstein et al., 2008). Cathepsin L activity is responsible for adipogenesis and glucose tolerance by degrading matrix fibronectin and processing the insulin receptor and IGF-1R beta (Yang et al., 2007). It is involved in intestinal epithelial cell polarization and differentiation (Boudreau et al., 2007) and in proteinuric kidney disease (Sever et al., 2007). Nuclear Cathepsin L was shown to proteolytically process a transcription factor during the G1/S progression of the cell cycle (Goulet et al., 2004) and histone H3 during mouse embryonic stem cell differentiation (Duncan et al., 2008). The landscape modifications of the histone on the Y chromosome and pericentromeric heterochromatine are stabilized by Cathepsin L (Bulynko et al., 2006). The role of Cathepsin L in cancer has been studied extensively. Secreted Cathepsin L degrades basal membrane and extracellular matrix therefore could increase the development of metastases. Intracellular cathepsin L activity can lead to activation of oncogenes or inactivation of tumor suppressors (Goulet et al., 2007). A recent paper also indicate that Cathepsin L plays a role in drug resistance (Zheng et al., 2008).
For example, Cathepsin L is important for epidermal homeostasis, regulation of the hair cycle, control of keratinocyte proliferation, MHC class-II mediated antigen presentation and selection of CD4+ T cells in cortical epithelial cells of the thymus (Nakagawa et al., 1998; Roth et al., 2000; Benavides et al., 2002; Reinheckel et al., 2005). Cathepsin L expression in thymocytes has been shown to be essential for natural killer cell development (Honey et al., 2002). Knockout mice models have demonstrated that Cathepsin L directly participates in atherosclerosis and in neovascularisation induced by endothelial progenitor cells (Maehr et al., 2005; Urbich et al., 2005; Kitamoto et al., 2007). These knockout mice have been observed to develop heart disease similar to human dilated cardiomyopathy (Stypmann et al., 2002). In general, pups lacking cathepsin L also have a slightly higher mortality upon weaning than their littermates (Reinheckel et al., 2001). Cathepsin L is responsible for the processing of viral proteins (Chandran et al., 2005; Pager et al., 2005; Kaletsky et al., 2007; Bosch et al., 2008) and the generation of neuropeptides and thyroid hormone (Funkelstein et al., 2008; Funkelstein et al., 2008). Cathepsin L activity is responsible for adipogenesis and glucose tolerance by degrading matrix fibronectin and processing the insulin receptor and IGF-1R beta (Yang et al., 2007). It is involved in intestinal epithelial cell polarization and differentiation (Boudreau et al., 2007) and in proteinuric kidney disease (Sever et al., 2007). Nuclear Cathepsin L was shown to proteolytically process a transcription factor during the G1/S progression of the cell cycle (Goulet et al., 2004) and histone H3 during mouse embryonic stem cell differentiation (Duncan et al., 2008). The landscape modifications of the histone on the Y chromosome and pericentromeric heterochromatine are stabilized by Cathepsin L (Bulynko et al., 2006). The role of Cathepsin L in cancer has been studied extensively. Secreted Cathepsin L degrades basal membrane and extracellular matrix therefore could increase the development of metastases. Intracellular cathepsin L activity can lead to activation of oncogenes or inactivation of tumor suppressors (Goulet et al., 2007). A recent paper also indicate that Cathepsin L plays a role in drug resistance (Zheng et al., 2008).
Homology
Human Cathepsin L1 belongs to the papain superfamily. Cathepsin L2 (formerly called Cathepsin V) originated from ancestral Cathepsin L as they share 77% amino-acids identity. Moreover, both are similar to mouse cathepsin L (72% and 75% respectively) and other mammals (Itoh et al., 1999).
Mutations
Note
Not determined.
Implicated in
Entity name
Various cancers
Oncogenesis
Cathepsin L was initially identified as the major excreted protein (MEP) secreted from transformed fibroblastic cells (Gottesman, 1978; Troen et al., 1987; Troen et al., 1988). Oncogenic signals such as Ras, Raf, v-Src, fos, SV40 Large T as well as tumor promoters like phorbol ester can induce MEP expression and secretion (Joseph et al., 1987; Taniguchi et al., 1990; Troen et al., 1991; Lemaire et al., 1994; Heinrich et al., 2000). Moreover, cathepsin L secretion correlated with the metastatic potential of transformed cell lines (Denhardt et al., 1987; Chambers et al., 1992). Increased cathepsin L activity and secretion has been observed in many human cancers (Watanabe et al., 1987; Sheahan et al., 1989; Chauhan et al., 1991; Heidtmann et al., 1993; Nishida et al., 1995; Plebani et al., 1995; Park et al., 1996; Shuja et al., 1996; Sivaparvathi et al., 1996; Leto et al., 1997; Kim et al., 1998; Dohchin et al., 2000). Various reports also suggested that cathepsin L levels could be used as a potential indicator of tumor aggressiveness and metastasis (Thomssen et al., 1995; Park et al., 1996). Increased nuclear Cathepsin L expression and activity was recently found in various cancer cells, suggesting a different mechanism of cellular transformation (Goulet et al., 2007).
Therefore, although the association of cathepsin L and cancer is well established, its specific roles have not yet been fully elucidated.
Therefore, although the association of cathepsin L and cancer is well established, its specific roles have not yet been fully elucidated.
Entity name
Breast cancer
Prognosis
In breast cancer, numerous studies have indicated that secreted cathepsin L could be a strong and independent prognostic factor, with a strength similar to lymph node status and grading (Castiglioni et al., 1994; Thomssen et al., 1995; Duffy, 1996; Foekens et al., 1998; Thomssen et al., 1998; Harbeck et al., 2000; Harbeck et al., 2001; Levicar et al., 2002). Cathepsin L expression could also predict response to adjuvant chemotherapy (Jagodic et al., 2005).
Entity name
Gastric carcinoma
Prognosis
Cathepsin L expression correlates with an early event in gastric carcinogenesis and with depth of invasion in early stage of gastric carcinoma. Higher expression is associated with worst prognosis (Plebani et al., 1995; Farinati et al., 1996; Dohchin et al., 2000).
Entity name
Skin cancer
Prognosis
Higher concentration of Cathepsin L in early primary melanomas correlates with poor prognosis and indicate possible early metastasis spread (Stabuc et al., 2006). In malignant cells of squamous cell carcinoma, Cathepsin L is mainly overexpressed at the periphery of the tumor. Cathepsin L is also overexpressed in various inflammatory skin diseases such as psoriasis and atopic eczema (Bylaite et al., 2006).
Entity name
Ovarian cancer
Prognosis
Cathepsin L expression is increased in ovarian cancer sample as well as in the serum of patients with ovarian cancer. Serum levels of Cathepsin L could be used in early detection of ovarian cancers (Nishida et al., 1995).
Entity name
Bladder cancer
Prognosis
Urinary Cathepsin L is an independent predictor of bladder urothelial cell cancer and invasiveness (Svatek et al., 2008).
Entity name
Brain cancer (neuroblastoma)
Prognosis
Cathepsin L has no prognostic value in glioma, but its expression is increased in tumor cells (Strojnik et al., 2005). In invasive benign meningioma and pituitary adenomas, Cathepesin L levels are also higher (Strojnik et al., 2001; Strojnik et al., 2005).
Entity name
Pancreatic adenocarcinoma
Prognosis
Cathepsin L is a strong independent prognostic marker in resectable cancers (Niedergethmann et al., 2004).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12137950 | 2002 | Identification and characterization of a novel human cathepsin L splice variant. | Arora S et al |
| 11574156 | 2001 | Cloning and characterization of human cathepsin L promoter. | Bakhshi R et al |
| 12163394 | 2002 | Impaired hair follicle morphogenesis and cycling with abnormal epidermal differentiation in nackt mice, a cathepsin L-deficient mutation. | Benavides F et al |
| 18562523 | 2008 | Cathepsin L functionally cleaves the severe acute respiratory syndrome coronavirus class I fusion protein upstream of rather than adjacent to the fusion peptide. | Bosch BJ et al |
| 17622569 | 2007 | Loss of cathepsin L activity promotes claudin-1 overexpression and intestinal neoplasia. | Boudreau F et al |
| 16705169 | 2006 | Cathepsin L stabilizes the histone modification landscape on the Y chromosome and pericentromeric heterochromatin. | Bulynko YA et al |
| 16433682 | 2006 | Expression of cathepsin L and its inhibitor hurpin in inflammatory and neoplastic skin diseases. | Bylaite M et al |
| 8088759 | 1994 | Immunohistochemical analysis of cathepsins D, B, and L in human breast cancer. | Castiglioni T et al |
| 1586450 | 1992 | Increased expression of cathepsins L and B and decreased activity of their inhibitors in metastatic, ras-transformed NIH 3T3 cells. | Chambers AF et al |
| 15831716 | 2005 | Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. | Chandran K et al |
| 9074757 | 1997 | Emerging roles for cysteine proteases in human biology. | Chapman HA et al |
| 8419312 | 1993 | Cloning, genomic organization, and chromosomal localization of human cathepsin L. | Chauhan SS et al |
| 8896443 | 1996 | Structure of human procathepsin L reveals the molecular basis of inhibition by the prosegment. | Coulombe R et al |
| 3438085 | 1987 | Cysteine proteinase cathepsin L expression correlates closely with the metastatic potential of H-ras-transformed murine fibroblasts. | Denhardt DT et al |
| 10931446 | 2000 | Immunostained cathepsins B and L correlate with depth of invasion and different metastatic pathways in early stage gastric carcinoma. | Dohchin A et al |
| 9816210 | 1996 | Proteases as prognostic markers in cancer. | Duffy MJ et al |
| 18957203 | 2008 | Cathepsin L proteolytically processes histone H3 during mouse embryonic stem cell differentiation. | Duncan EM et al |
| 2663888 | 1989 | Biosynthesis of lysosomal endopeptidases. | Erickson AH et al |
| 9006092 | 1996 | Increased levels of cathepsin B and L, urokinase-type plasminogen activator and its inhibitor type-1 as an early event in gastric carcinogenesis. | Farinati F et al |
| 9508185 | 1998 | Prognostic significance of cathepsins B and L in primary human breast cancer. | Foekens JA et al |
| 18849346 | 2008 | Major role of cathepsin L for producing the peptide hormones ACTH, beta-endorphin, and alpha-MSH, illustrated by protease gene knockout and expression. | Funkelstein L et al |
| 3533059 | 1986 | The major excreted protein (MEP) of transformed mouse cells and cathepsin L have similar protease specificity. | Gal S et al |
| 11177576 | 2001 | Interleukin-6 and transforming growth factor-beta 1 control expression of cathepsins B and L in human lung epithelial cells. | Gerber A et al |
| 351614 | 1978 | Transformation-dependent secretion of a low molecular weight protein by murine fibroblasts. | Gottesman MM et al |
| 15099520 | 2004 | A cathepsin L isoform that is devoid of a signal peptide localizes to the nucleus in S phase and processes the CDP/Cux transcription factor. | Goulet B et al |
| 17855659 | 2007 | Increased expression and activity of nuclear cathepsin L in cancer cells suggests a novel mechanism of cell transformation. | Goulet B et al |
| 11555589 | 2001 | Prognostic impact of proteolytic factors (urokinase-type plasminogen activator, plasminogen activator inhibitor 1, and cathepsins B, D, and L) in primary breast cancer reflects effects of adjuvant systemic therapy. | Harbeck N et al |
| 8054705 | 1993 | Secretion of a latent, acid activatable cathepsin L precursor by human non-small cell lung cancer cell lines. | Heidtmann HH et al |
| 11043381 | 2000 | Induction of putative tumor-suppressing genes in Rat-1 fibroblasts by oncogenic Raf-1 as evidenced by robot-assisted complex hybridization. | Heinrich J et al |
| 12021314 | 2002 | Cathepsin L regulates CD4+ T cell selection independently of its effect on invariant chain: a role in the generation of positively selecting peptide ligands. | Honey K et al |
| 12553719 | 2002 | Processing and activation of lysosomal proteinases. | Ishidoh K et al |
| 10382972 | 1999 | Genomic organization and chromosomal localization of the human cathepsin L2 gene. | Itoh R et al |
| 15739019 | 2005 | Prognostic and predictive value of cathepsins D and L in operable breast cancer patients. | Jagodic M et al |
| 11742542 | 2002 | Characterization of human cathepsin L promoter and identification of binding sites for NF-Y, Sp1 and Sp3 that are essential for its activity. | Jean D et al |
| 18366346 | 2008 | Cathepsin L expression is up-regulated by hypoxia in human melanoma cells: role of its 5'-untranslated region. | Jean D et al |
| 3550705 | 1987 | The major ras induced protein in NIH3T3 cells is cathepsin L. | Joseph L et al |
| 2835398 | 1988 | Complete nucleotide and deduced amino acid sequences of human and murine preprocathepsin L. An abundant transcript induced by transformation of fibroblasts. | Joseph LJ et al |
| 7649308 | 1995 | Secretion and processing mechanisms of procathepsin L in bone resorption. | Kakegawa H et al |
| 17928356 | 2007 | Proteolysis of the Ebola virus glycoproteins enhances virus binding and infectivity. | Kaletsky RL et al |
| 8496193 | 1993 | Mouse procathepsin L lacking a functional glycosylation site is properly folded, stable, and secreted by NIH 3T3 cells. | Kane SE et al |
| 9699522 | 1998 | Presence of activated ras correlates with increased cysteine proteinase activities in human colorectal carcinomas. | Kim K et al |
| 17404153 | 2007 | Cathepsin L deficiency reduces diet-induced atherosclerosis in low-density lipoprotein receptor-knockout mice. | Kitamoto S et al |
| 7983639 | 1994 | Synovial fibroblast-like cell transfection with the SV40 large T antigen induces a transformed phenotype and permits transient tumor formation in immunodeficient mice. | Lemaire R et al |
| 9255106 | 1997 | Selective induction of the secretion of cathepsins B and L by cytokines in synovial fibroblast-like cells. | Lemaire R et al |
| 8978598 | 1997 | Lysosomal cathepsins B and L and Stefin A blood levels in patients with hepatocellular carcinoma and/or liver cirrhosis: potential clinical implications. | Leto G et al |
| 12088202 | 2002 | Comparison of potential biological markers cathepsin B, cathepsin L, stefin A and stefin B with urokinase and plasminogen activator inhibitor-1 and clinicopathological data of breast carcinoma patients. | Levicar N et al |
| 16184198 | 2005 | Cathepsin L is essential for onset of autoimmune diabetes in NOD mice. | Maehr R et al |
| 3977867 | 1985 | Human liver cathepsin L. | Mason RW et al |
| 1482371 | 1992 | Surface activation of pro-cathepsin L. | Mason RW et al |
| 10410800 | 1999 | The lysosomal cysteine proteases. | McGrath ME et al |
| 9545226 | 1998 | Cathepsin L: critical role in Ii degradation and CD4 T cell selection in the thymus. | Nakagawa T et al |
| 15367886 | 2004 | Prognostic impact of cysteine proteases cathepsin B and cathepsin L in pancreatic adenocarcinoma. | Niedergethmann M et al |
| 7705668 | 1995 | Increased cathepsin L levels in serum in some patients with ovarian cancer: comparison with CA125 and CA72-4. | Nishida Y et al |
| 2658811 | 1989 | Evidence that aspartic proteinase is involved in the proteolytic processing event of procathepsin L in lysosomes. | Nishimura Y et al |
| 9020032 | 1997 | Processing properties of recombinant human procathepsin L. | Nomura T et al |
| 16188974 | 2005 | Cathepsin L is involved in proteolytic processing of the Hendra virus fusion protein. | Pager CT et al |
| 8835762 | 1996 | Enhanced expression of cathepsin L in metastatic bone tumors. | Park IC et al |
| 8625115 | 1995 | Cysteine and serine proteases in gastric cancer. | Plebani M et al |
| 16079282 | 2005 | The lysosomal cysteine protease cathepsin L regulates keratinocyte proliferation by control of growth factor recycling. | Reinheckel T et al |
| 8830671 | 1996 | Sequence analysis and distribution of two new human cathepsin L splice variants. | Rescheleit DK et al |
| 15195995 | 2004 | Comprehensive search for cysteine cathepsins in the human genome. | Rossi A et al |
| 11023992 | 2000 | Cathepsin L deficiency as molecular defect of furless: hyperproliferation of keratinocytes and pertubation of hair follicle cycling. | Roth W et al |
| 2264836 | 1990 | Inhibitor studies indicate that active cathepsin L is probably essential to its own processing in cultured fibroblasts. | Salminen A et al |
| 17671649 | 2007 | Proteolytic processing of dynamin by cytoplasmic cathepsin L is a mechanism for proteinuric kidney disease. | Sever S et al |
| 2544258 | 1989 | Cysteine protease activities and tumor development in human colorectal carcinoma. | Sheahan K et al |
| 8635854 | 1996 | Marked increases in cathepsin B and L activities distinguish papillary carcinoma of the thyroid from normal thyroid or thyroid with non-neoplastic disease. | Shuja S et al |
| 8521613 | 1996 | Expression and immunohistochemical localization of cathepsin L during the progression of human gliomas. | Sivaparvathi M et al |
| 2480110 | 1989 | Glycosylation of procathepsin L does not account for species molecular-mass differences and is not required for proteolytic activity. | Smith SM et al |
| 16652198 | 2006 | Expression and prognostic significance of Cathepsin L in early cutaneous malignant melanoma. | Stabuc B et al |
| 15816632 | 2005 | Immunohistochemical staining of cathepsins B, L and stefin A in human hypophysis and pituitary adenomas. | Strojnik T et al |
| 11972068 | 2002 | Dilated cardiomyopathy in mice deficient for the lysosomal cysteine peptidase cathepsin L. | Stypmann J et al |
| 18076936 | 2008 | Role of urinary cathepsin B and L in the detection of bladder urothelial cell carcinoma. | Svatek RS et al |
| 2185753 | 1990 | Augmented excretion of procathepsin L of a fos-transferred highly metastatic rat cell line. | Taniguchi S et al |
| 9703780 | 1998 | Identification of low-risk node-negative breast cancer patients by tumor biological factors PAI-1 and cathepsin L. | Thomssen C et al |
| 2826441 | 1988 | Cloning and expression of the gene for the major excreted protein of transformed mouse fibroblasts. A secreted lysosomal protease regulated by transformation. | Troen BR et al |
| 1848774 | 1991 | Downstream sequences mediate induction of the mouse cathepsin L promoter by phorbol esters. | Troen BR et al |
| 3689328 | 1987 | Sequence and expression of the cDNA for MEP (major excreted protein), a transformation-regulated secreted cathepsin. | Troen BR et al |
| 9165064 | 1997 | Structural and functional aspects of papain-like cysteine proteinases and their protein inhibitors. | Turk B et al |
| 15665831 | 2005 | Cathepsin L is required for endothelial progenitor cell-induced neovascularization. | Urbich C et al |
| 16440214 | 2006 | The importance of cysteine cathepsin proteases for placental development. | Varanou A et al |
| 3610018 | 1987 | Elevation of tissue cathepsin B and L activities in gastric cancer. | Watanabe M et al |
| 17643114 | 2007 | Cathepsin L activity controls adipogenesis and glucose tolerance. | Yang M et al |
| 12869695 | 2003 | Cathepsin L in secretory vesicles functions as a prohormone-processing enzyme for production of the enkephalin peptide neurotransmitter. | Yasothornsrikul S et al |
| 18971393 | 2009 | Cathepsin L inhibition suppresses drug resistance in vitro and in vivo: a putative mechanism. | Zheng X et al |
Other Information
Locus ID:
NCBI: 1514
MIM: 116880
HGNC: 2537
Ensembl: ENSG00000135047
Variants:
dbSNP: 1514
ClinVar: 1514
TCGA: ENSG00000135047
COSMIC: CTSL
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 35900152 | 2023 | Reduced Cathepsin L expression and secretion into the extracellular milieu contribute to lung fibrosis in systemic sclerosis. | 5 |
| 36382580 | 2023 | Methyltransferase-like 3 promotes cervical cancer metastasis by enhancing cathepsin L mRNA stability in an N6-methyladenosine-dependent manner. | 6 |
| 37738239 | 2023 | Large-scale whole exome sequencing studies identify two genes,CTSL and APOE, associated with lung cancer. | 0 |
| 35900152 | 2023 | Reduced Cathepsin L expression and secretion into the extracellular milieu contribute to lung fibrosis in systemic sclerosis. | 5 |
| 36382580 | 2023 | Methyltransferase-like 3 promotes cervical cancer metastasis by enhancing cathepsin L mRNA stability in an N6-methyladenosine-dependent manner. | 6 |
| 37738239 | 2023 | Large-scale whole exome sequencing studies identify two genes,CTSL and APOE, associated with lung cancer. | 0 |
| 34807310 | 2022 | Expressions and significances of CTSL, the target of COVID-19 on GBM. | 8 |
| 34883264 | 2022 | XPA is susceptible to proteolytic cleavage by cathepsin L during lysis of quiescent cells. | 2 |
| 35414771 | 2022 | COVID-19 receptor and malignant cancers: Association of CTSL expression with susceptibility to SARS-CoV-2. | 18 |
| 35941174 | 2022 | A novel high-risk subpopulation identified by CTSL and ZBTB7B in gastric cancer. | 6 |
| 34807310 | 2022 | Expressions and significances of CTSL, the target of COVID-19 on GBM. | 8 |
| 34883264 | 2022 | XPA is susceptible to proteolytic cleavage by cathepsin L during lysis of quiescent cells. | 2 |
| 35414771 | 2022 | COVID-19 receptor and malignant cancers: Association of CTSL expression with susceptibility to SARS-CoV-2. | 18 |
| 35941174 | 2022 | A novel high-risk subpopulation identified by CTSL and ZBTB7B in gastric cancer. | 6 |
| 32242285 | 2021 | Mechanisms of L-Serine-Mediated Neuroprotection Include Selective Activation of Lysosomal Cathepsins B and L. | 9 |
Citation
Brigitte Goulet ; Alain Nepveu
CTSL (cathepsin L1)
Atlas Genet Cytogenet Oncol Haematol. 2009-01-01
Online version: http://atlasgeneticsoncology.org/gene/40208/ctsl
