CYR61 (cysteine-rich, angiogenic inducer, 61)
2013-01-01 Chih-Chiun Chen , Lester F Lau AffiliationDepartment of Biochemistry, Molecular Genetics, University of Illinois at Chicago, 900 South Ashland Avenue, Chicago, IL 60607, USA
Identity
HGNC
LOCATION
1p22.3
LOCUSID
ALIAS
CYR61,GIG1,IGFBP10
FUSION GENES
DNA/RNA
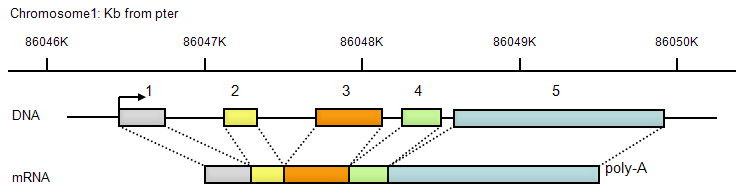
CYR61 is located on chromosome 1, 86046 kbp ~ 86049 kbp from the p-arm telomere (pter). Each exon is depicted as a box with a unique color, with the numbers of exons shown above. Mature mRNA is ~2,3 kbp in length plus a poly-A tail.
Description
Gene structure
The transcribed region of the CYR61 genomic locus spans ~3,2 kb, with 5 exons interspaced by 4 introns. The first exon encodes the 5 untranslated region of the mRNA and followed by sequences encoding the protein secretory signal peptide. The remaining 4 exons each encode a discrete CYR61 domain, and the last exon also contains the mRNA 3 untranslated sequence (OBrien et al., 1990). The 2 kb DNA upstream of the transcription start site contains sufficient information as the Cyr61 promoter to drive accurate expression in transgenic mice both during embryonic development and in adult wound healing (Latinkic et al., 2001). Since each CYR61 domain is encoded by a discrete exon, it was hypothesized that CYR61 arose through an exon-shuffling mechanism (Brigstock, 1999; Lau and Lam, 1999).
The transcribed region of the CYR61 genomic locus spans ~3,2 kb, with 5 exons interspaced by 4 introns. The first exon encodes the 5 untranslated region of the mRNA and followed by sequences encoding the protein secretory signal peptide. The remaining 4 exons each encode a discrete CYR61 domain, and the last exon also contains the mRNA 3 untranslated sequence (OBrien et al., 1990). The 2 kb DNA upstream of the transcription start site contains sufficient information as the Cyr61 promoter to drive accurate expression in transgenic mice both during embryonic development and in adult wound healing (Latinkic et al., 2001). Since each CYR61 domain is encoded by a discrete exon, it was hypothesized that CYR61 arose through an exon-shuffling mechanism (Brigstock, 1999; Lau and Lam, 1999).
SNP
rs3753794 and rs3753793 (r2= 0.77) were associated plasma HDL-cholesterol levels (p= 0.016 and p= 0.008) (Bouchard et al., 2007).
rs954353, located 1,8 kb upstream of CYR61, is linked to susceptibility to colorectal cancer (Fernández-Rozadilla et al., 2010).
Transcription
The CYR61 promoter is a TATA-box promoter containing binding sites for many transcription factors including AP1, ATF, E2F, HNF3b, NF1, NFkB, SP1, and SRF, and 2 poly(CA) stretches that may form Z-DNA structure. CYR61 transcription is highly responsive to induction by serum growth factors, cytokines, hormones (angiotensin II, estrogen, vitamin D3), and environmental perturbations including UV irradiation, hypoxia, and mechanical stress. During mouse embryo development Cyr61 is highly expressed in cardiovascular, neuronal, and skeletal systems. In adulthood CYR61 is induced at sites of inflammation and wound repair, and de-regulated in many forms of cancer.
Internal ribosome entry site: CYR61 mRNA contains a highly conserved, guanine-cytosine (GC)-rich region of approximately 200 bp near the 5-end of the open reading frame which serves as an internal ribosome entry site (Mukudai et al., 2010). It may facilitate CYR61 production in stressful conditions such as during viral infection (Johannes et al., 1999).
Alternative splicing: hypoxia during breast carcinogenesis may promote a shift of CYR61 alternative splicing to promote CYR61 production. In normal breast tissue most CYR61 RNA transcript contains intron-3, which does not produce CYR61 protein. In cancerous tissues that often suffer hypoxia, intron-3 is spliced out, resulting in mature CYR61 mRNA and a higher level of CYR61 production (Hirschfeld et al., 2009; Hirschfeld et al., 2011).
3-UTR: CYR61 3UTR contains 5 copies of AU-rich element (ARE) in the 3-UTR, which causes RNA instability (Leng et al., 2002). In addition, CYR61 mRNA 3-UTR contains a microRNA-155 target site. In preeclampsia where placental miR-155 level is high, CYR61 mRNA is reduced due to targeted degradation by miR-155 (Zhang et al., 2010).
Internal ribosome entry site: CYR61 mRNA contains a highly conserved, guanine-cytosine (GC)-rich region of approximately 200 bp near the 5-end of the open reading frame which serves as an internal ribosome entry site (Mukudai et al., 2010). It may facilitate CYR61 production in stressful conditions such as during viral infection (Johannes et al., 1999).
Alternative splicing: hypoxia during breast carcinogenesis may promote a shift of CYR61 alternative splicing to promote CYR61 production. In normal breast tissue most CYR61 RNA transcript contains intron-3, which does not produce CYR61 protein. In cancerous tissues that often suffer hypoxia, intron-3 is spliced out, resulting in mature CYR61 mRNA and a higher level of CYR61 production (Hirschfeld et al., 2009; Hirschfeld et al., 2011).
3-UTR: CYR61 3UTR contains 5 copies of AU-rich element (ARE) in the 3-UTR, which causes RNA instability (Leng et al., 2002). In addition, CYR61 mRNA 3-UTR contains a microRNA-155 target site. In preeclampsia where placental miR-155 level is high, CYR61 mRNA is reduced due to targeted degradation by miR-155 (Zhang et al., 2010).
Pseudogene
None identified yet.
Proteins
Note
CYR61 is a secreted protein located in the extracellular matrix and on cell surface (Yang and Lau, 1991), and regulates diverse cellular activity including cell adhesion, migration, proliferation, apoptosis, senescence, and differentiation (Lau, 2011). It binds to and acts through cell surface integrin receptors, with heparan sulfate proteoglycans (HSPGs) as coreceptors in some contexts (Chen and Lau, 2009; Lau, 2011). Cell adhesion to CYR61 as a substrate induces activation of various signaling molecules including Rac1, FAK, the MAP kinases ERK1/ERK2, and transcription factors such as NFκB (Chen et al., 2001a; Bai et al., 2010). CYR61 is a strong inducer of intracellular reactive oxygen species (ROS) through multiple cellular sources including NADPH Oxidase, 5-lipoxygenase, sphingomyelinase, and the mitochondria; accumulation of ROS mediates Cyr61-promoted apoptosis and cellular senescence (Chen CC et al., 2007; Jun and Lau, 2010b; Juric et al., 2012).
Targeted Cyr61 deletion in mice is embryonic lethal due to cardiac atrioventricular septal defects, impaired placentation, and compromised blood vessel integrity (Mo et al., 2002; Mo and Lau, 2006). Consistently, CYR61 induces pro-angiogenic activity in endothelial cells in culture and neovascularization in vivo (Babic et al., 1998; Fataccioli et al., 2002; Leu et al., 2002).
In adulthood Cyr61 expression is strongly associated with inflammation and wound repair (Lau, 2011). In particular, Cyr61 plays a critical role in the resolution of fibrosis during wound healing by inducing cellular senescence in myofibroblasts (Jun and Lau, 2010b). In inflammation, CYR61 is critical for TNFα-mediated apoptosis in vivo, and can enhance the cytotoxicity of other TNF family cytokines including LTα, FasL, and TRAIL (Chen CC et al., 2007; Franzen et al., 2009; Juric et al., 2009). Finally, CYR61 expression is de-regulated in most human chronic inflammatory diseases including rheumatoid arthritis, diabetic retinopathy and nephropathy, atherosclerosis, and many cancers (Lau, 2011).
Targeted Cyr61 deletion in mice is embryonic lethal due to cardiac atrioventricular septal defects, impaired placentation, and compromised blood vessel integrity (Mo et al., 2002; Mo and Lau, 2006). Consistently, CYR61 induces pro-angiogenic activity in endothelial cells in culture and neovascularization in vivo (Babic et al., 1998; Fataccioli et al., 2002; Leu et al., 2002).
In adulthood Cyr61 expression is strongly associated with inflammation and wound repair (Lau, 2011). In particular, Cyr61 plays a critical role in the resolution of fibrosis during wound healing by inducing cellular senescence in myofibroblasts (Jun and Lau, 2010b). In inflammation, CYR61 is critical for TNFα-mediated apoptosis in vivo, and can enhance the cytotoxicity of other TNF family cytokines including LTα, FasL, and TRAIL (Chen CC et al., 2007; Franzen et al., 2009; Juric et al., 2009). Finally, CYR61 expression is de-regulated in most human chronic inflammatory diseases including rheumatoid arthritis, diabetic retinopathy and nephropathy, atherosclerosis, and many cancers (Lau, 2011).
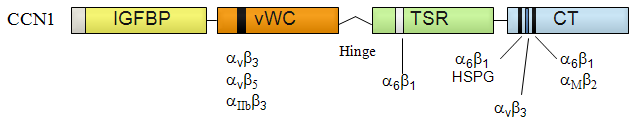
Each domain is depicted as a box with a unique color, with the domain name shown in the box. The grey box represents the secretory signal peptide. Integrin/HSPGs binding sites are marked as stripes.
Description
Domain structure, receptor binding sites, and interacting proteins
Full-length CYR61 precursor is 381 amino acids in length, with aa 1-24 being the secretory signal peptide. The calculated molecular size for mature CYR61 is 39,4 kDa, and on SDS-PAGE it migrates as a ~40 kDa protein band. CYR61 contains unusually high cysteine content (37 cysteines in the processed protein), in addition, all cysteines are completely conserved among CCN1, CCN2, CCN3, and CCN4.
CYR61 is a direct ligand of several integrin receptors, including αvβ3, αvβ5, αIIbβ3, αMβ2, and α6β1, and has strong binding for cell surface HSPGs (Kireeva et al., 1998; Jedsadayanmata et al., 1999; Chen et al., 2000; Grzeszkiewicz et al., 2002; Schober et al., 2002). Cyr61 binds preferentially to particular integrins in a cell-type specific manner, and this interaction mediates many activities of CYR61.
Proteins of the CCN protein family consists of four structural domains (Holbourn et al., 2008). The first CYR61 domain has sequence homologies to insulin-like growth factor binding protein (IGFBP), although CYR61 does not bind IGFs. The second domain is homologous to the von Willebrand Factor type C repeat (vWC). It has a functional integrin αvβ3 binding site with the sequence NCKHQCTCIDGAVGCIPLCP from aa 116 to 135 (Chen et al., 2004). A point mutation substituting the center aspartic acid residue to alanine (D125A mutation) eliminates the binding activity. Consistent with the notion that CYR61 induces angiogenesis through direct binding to integrin αvβ3, the D125A mutation in Cyr61 effectively abrogates its angiogenic activity (Chen et al., 2004).
Following the vWC-domain is a hinge-region that does not seem to have a particular 3d structure, and its aa sequence is not conserved among the CCN-family members.
The third domain has homology to thrombospondin type 1 repeat (TSR). It contains an integrin α6β1 binding site called T1 with the sequence TTSWSQCSKS from aa 231-240 (Leu et al., 2003).
The fourth domain (CT) has homology to the cysteine-knot motif, and has two binding sites to integrin α6β1 and HSPGs called H1 (aa 278-295, YSSLKKGKKCSKTKKSPE) and H2 (aa 302-314, SSVKKYRPKYCGS), each contributes to heparin binding and integrin α6β1/HSPGs-mediated fibroblast cell adhesion (Chen et al., 2000). The H2 site also overlaps a binding site for integrin αMβ2 and can mediate monocyte cell adhesion (Schober et al., 2002).
The domains of CYR61 may function independently. A mutant CYR61 protein with the CT-domain deleted is unable to induce fibroblast cell adhesion, but still able to activate αv integrin-mediated activities, including cell migration, and potentiate bFGF-induced cell proliferation (Grzeszkiewicz et al., 2001). Similarly, a CYR61 mutant protein (DM) with disrupted integrin α6β1/HSPGs-binding sites is defective for CYR61 supported fibroblast functions, but can still promoting αv integrin-mediated angiogenic activities (Chen et al., 2004). The N-terminal half of CYR61 alone, which contains domains 1 and 2, can bind purified integrin αvβ3 and mediate endothelial cell adhesion (Chen et al., 2004). Mixing CYR61-D125A (mutated in the αv integrin binding site), with CYR61-DM (mutated in the α6β1/HSPG binding sites) can reconstitute wild-type CYR61 function to synergize with TNFα and induce fibroblast apoptosis (Chen CC et al., 2007).
Full-length CYR61 precursor is 381 amino acids in length, with aa 1-24 being the secretory signal peptide. The calculated molecular size for mature CYR61 is 39,4 kDa, and on SDS-PAGE it migrates as a ~40 kDa protein band. CYR61 contains unusually high cysteine content (37 cysteines in the processed protein), in addition, all cysteines are completely conserved among CCN1, CCN2, CCN3, and CCN4.
CYR61 is a direct ligand of several integrin receptors, including αvβ3, αvβ5, αIIbβ3, αMβ2, and α6β1, and has strong binding for cell surface HSPGs (Kireeva et al., 1998; Jedsadayanmata et al., 1999; Chen et al., 2000; Grzeszkiewicz et al., 2002; Schober et al., 2002). Cyr61 binds preferentially to particular integrins in a cell-type specific manner, and this interaction mediates many activities of CYR61.
Proteins of the CCN protein family consists of four structural domains (Holbourn et al., 2008). The first CYR61 domain has sequence homologies to insulin-like growth factor binding protein (IGFBP), although CYR61 does not bind IGFs. The second domain is homologous to the von Willebrand Factor type C repeat (vWC). It has a functional integrin αvβ3 binding site with the sequence NCKHQCTCIDGAVGCIPLCP from aa 116 to 135 (Chen et al., 2004). A point mutation substituting the center aspartic acid residue to alanine (D125A mutation) eliminates the binding activity. Consistent with the notion that CYR61 induces angiogenesis through direct binding to integrin αvβ3, the D125A mutation in Cyr61 effectively abrogates its angiogenic activity (Chen et al., 2004).
Following the vWC-domain is a hinge-region that does not seem to have a particular 3d structure, and its aa sequence is not conserved among the CCN-family members.
The third domain has homology to thrombospondin type 1 repeat (TSR). It contains an integrin α6β1 binding site called T1 with the sequence TTSWSQCSKS from aa 231-240 (Leu et al., 2003).
The fourth domain (CT) has homology to the cysteine-knot motif, and has two binding sites to integrin α6β1 and HSPGs called H1 (aa 278-295, YSSLKKGKKCSKTKKSPE) and H2 (aa 302-314, SSVKKYRPKYCGS), each contributes to heparin binding and integrin α6β1/HSPGs-mediated fibroblast cell adhesion (Chen et al., 2000). The H2 site also overlaps a binding site for integrin αMβ2 and can mediate monocyte cell adhesion (Schober et al., 2002).
The domains of CYR61 may function independently. A mutant CYR61 protein with the CT-domain deleted is unable to induce fibroblast cell adhesion, but still able to activate αv integrin-mediated activities, including cell migration, and potentiate bFGF-induced cell proliferation (Grzeszkiewicz et al., 2001). Similarly, a CYR61 mutant protein (DM) with disrupted integrin α6β1/HSPGs-binding sites is defective for CYR61 supported fibroblast functions, but can still promoting αv integrin-mediated angiogenic activities (Chen et al., 2004). The N-terminal half of CYR61 alone, which contains domains 1 and 2, can bind purified integrin αvβ3 and mediate endothelial cell adhesion (Chen et al., 2004). Mixing CYR61-D125A (mutated in the αv integrin binding site), with CYR61-DM (mutated in the α6β1/HSPG binding sites) can reconstitute wild-type CYR61 function to synergize with TNFα and induce fibroblast apoptosis (Chen CC et al., 2007).
Mutations
Germinal
The CYR61 gene is located in human chromosome 1p22.3, within the region of a susceptibility gene locus for atrioventricular septal defects (AVSD; 1p21-1p31) (Sheffield et al., 1997). Targeted disruption of Cyr61 in mice results in severe atrioventricular septal defects (AVSD). Furthermore, Cyr61 haploinsufficiency results in delayed formation of the ventricular septa in embryos and persistent ostium primum atrial septal defects (ASD) in ≈20% of adults (Mo and Lau, 2006).
Implicated in
Entity name
Cancer
Note
CYR61 expression is regulated by a wide array of growth factors, inflammatory cytokines, and hormones, which are often de-regulated in cancerous tissues. CYR61 protein is a potent context-dependent regulator of many cellular functions important in tumorigenesis, including cell proliferation, survival/apoptosis, senescence, motility/chemotaxis, invasion in extracellular matrix, gene expression, differentiation, and angiogenesis. Perhaps as a consequence CYR61 is aberrantly expressed in most forms of solid tumors where, depending on cancer types, it may have either promoting or suppressing roles. Many research works are ongoing to understand CYR61 function in the context of each specific type of cancer, which may in the future lead to better prognostic tools and cancer treatment.
Entity name
Breast cancer
Note
CYR61 may have a role in breast cancer progression and the development of resistance to anti-cancer drugs. CYR61 expression is associated with more advanced cancer features, including tumor stage, size, lymph node infiltration, age, and estrogen receptor expression (Xie et al., 2001). Over-expressing CYR61 in the breast cancer cell line MCF-7 can induce estrogen-independent cell growth, and promote tumorigenesity in nude mice (Tsai et al., 2002). CYR61 over-expression confers cellular resistance to apoptosis induction by anti-cancer drugs including Taxol, Adriamycin, and β-lapachone (Lin et al., 2004; Menendez et al., 2005). In addition, CYR61 induction by the Hippo-LATS signaling pathway enables Taxol resistance in breast cancer cells (Lai et al., 2011). These results suggest CYR61 may be a novel therapeutic target in treating drug-resistant breast cancers.
Entity name
Endometrial cancer
Note
CYR61 expression in endometrial cancers is down-regulated when compared to matched normal endometrial tissue, suggesting it may have a tumor-suppressing role (Chien et al., 2004). Consistent with the idea, over-expressing CYR61 in the endometrial cancer cell line AN3CA decreased cell growth in vitro, and abrogated tumorigenicity in nude mice (Chien et al., 2004). Moderate to high CYR61 levels are detectable by immunohistochemical analysis in ~50% of endometrioid adenocarcinoma, the most common form of endometrial cancer. However its expression is not associated with pathological features including age, cancer stage, invasion to surrounding tissues, or lymph node metastasis (Watari et al., 2009).
Entity name
Ovarian cancer
Note
CYR61 mRNA in ovarian cancer tissues is up-regulated in ~60% patients with a range from 2 to 10 fold higher than that found in normal ovary samples. Its over-expression is also associated with higher disease stages (Gery et al., 2005). In ovarian cancer cell lines, CYR61 promotes cell proliferation, enhances tumor growth in nude mice, and confers resistance to cytotoxicity by the anti-cancer drug carboplatin (Gery et al., 2005). These results suggest that CYR61 may have a promoting role in ovarian cancer.
Entity name
Prostate cancer
Note
CYR61 may promote primary prostate cancer in situ but repress the spreading of prostate cancer cells after surgical removal of the primary tumor. Its expression is up-regulated in benign prostate hyperplasia compared to normal prostate tissues (Sakamoto et al., 2004), and higher in prostate cancer than in normal (DAntonio et al., 2010b) and benign hyperplasia tissues (Lv et al., 2009). In addition, CYR61 supports cell adhesion and proliferation in prostate epithelial and cancer cells (Franzen et al., 2009; Sakamoto et al., 2004). Together these results show CYR61 may have a promoting role in prostate cancer. However, higher CYR61 expression is associated with >50% reduced cancer recurrence rates in patients after radical prostatectomy for clinically localized prostate cancer (DAntonio et al., 2010a). In addition CYR61 can cooperate with TRAIL to induce prostate cancer cell apoptosis (Franzen et al., 2009). It is hypothesized that CYR61 may function in a TRAIL-dependent immune surveillance mechanism to repress potential prostate cancer cell spreading in the hosts leading to reduced cancer recurrence.
Entity name
Lung cancer
Note
CYR61 acts as a tumor-suppressor in lung cancers. Its mRNA level in lung tumor is decreased compared to adjacent non-cancerous tissues, and more reduced expression is associated with more advanced disease and poorer patient survival rates (Chen PP et al., 2007; Mori et al., 2007). Stable ectopic expression of CYR61 in non-small cell lung carcinoma cell lines caused cell cycle arrests in G1, elevated p53 and p21 expression, and reduced tumorigenecity in nude mice (Tong et al., 2001).
Entity name
Note
Immunohistochemical analysis of osteosarcoma tissue microarrays using anti-CYR61 antibodies shows that higher CYR61 levels are associated with higher tumor grades and shorter patient survival (Sabile et al., 2012; Fromigue et al., 2011). Over-expressing CYR61 in osteosarcoma cell lines enhanced tumorigenecity and lung metastasis in nude mice (Sabile et al., 2012), while silencing CYR61 abrogated cell Matrigel invasion in vitro, and lung metastasis in vivo (Fromigue et al., 2011). These results suggest that CYR61 may promote osteosarcoma tumorigenesis and metastasis.
Entity name
Pancreatic cancer
Note
Elevated CYR61 in pancreatic ductal adenocarcinoma compared to adjacent normal tissue was detected in patient tissue samples by in situ hybridization and immunihistochemical analysis (Haque et al., 2012; Haque et al., 2011). In a human pancreatic cancer cell line Panc-1, CYR61 expression is necessary for cellular expression of stem cell-like phenotype and cell migration, and tumorigenesis in nude mice (Haque et al., 2011). CYR61 may act by binding integrin alphavbeta3 to suppress cellular Notch-1 proteosome degradation, leading to the activation of Sonic Hedgehog signaling and cell migration (Haque et al., 2012).
Entity name
Glioma
Note
CYR61 has promoting roles in glioma tumorigenesis. Its mRNA and protein levels are elevated compared to normal brain tissues, and in addition, its aberrant expression is associated with higher cancer grades and poor prognosis (Xie et al., 2004a; Goodwin et al., 2010). Over-expressing CYR61 in glioma cells in vitro activates ILK, Akt, and GSK3/b-catenin signaling, and promotes pro-tumorigenic cellular activities including proliferation, colony-formation in soft agar, and cell migration. Forced CYR61 expression in glioma cells enhanced tumorigenicity when xenografted in nude mice (Xie et al., 2004a), while injecting CYR61 siRNA in pre-established tumor xenografts reduced cell proliferation and tumor growth (Goodwin et al., 2010).
Entity name
Colorectal cancer
Note
CYR61 mRNA levels are ~10-fold higher in colorectal cancers compared to that in normal colon tissues. However expression is reduced in more advanced cancers and there is no statistically significant association between CYR61 and pathological parameters (Ladwa et al., 2011).
Entity name
Wound healing and fibrogenesis
Note
CYR61 expression is highly induced during injury/repair of many tissue and organs, including in the granulation tissue of skin wounds (Chen et al., 2001b; Rittié et al., 2011), liver after partial hepatectomy (Su et al., 2002), the callus in the healing bone (Hadjiargyrou et al., 2000; Lienau et al., 2006), and the heart after myocardial infarction (Hilfiker-Kleiner et al., 2004).
Tissue wound repair in mammals occurs in three distinct but overlapping phases: inflammation, followed by formation of granulation tissues (fibroblasts activation, proliferation, fibrogenesis, and angiogenesis), and finally remodeling and resolution of the granulation tissue to form the mature tissue (Gurtner et al., 2008). Resident fibroblasts and recruited fibrocytes are activated in wounds to proliferate and become myofibroblasts, which rapidly synthesize extracellular matrix. The matrix is important for maintaining tissue integrity and may help parenchymal cell regeneration. CYR61 is highly expressed in the granulation tissue during the resolution phase in skin wounds, where CYR61 drives fibrogenic myofibroblasts to become senescent cells. Senescent myofibroblasts cease to proliferate and express an anti-fibrotic phenotype, including the enhanced expression of matrix degradation enzymes to help resolution of fibrosis (Rodier and Campisi, 2011; Jun and Lau, 2010b; Jun and Lau, 2010a).
Tissue wound repair in mammals occurs in three distinct but overlapping phases: inflammation, followed by formation of granulation tissues (fibroblasts activation, proliferation, fibrogenesis, and angiogenesis), and finally remodeling and resolution of the granulation tissue to form the mature tissue (Gurtner et al., 2008). Resident fibroblasts and recruited fibrocytes are activated in wounds to proliferate and become myofibroblasts, which rapidly synthesize extracellular matrix. The matrix is important for maintaining tissue integrity and may help parenchymal cell regeneration. CYR61 is highly expressed in the granulation tissue during the resolution phase in skin wounds, where CYR61 drives fibrogenic myofibroblasts to become senescent cells. Senescent myofibroblasts cease to proliferate and express an anti-fibrotic phenotype, including the enhanced expression of matrix degradation enzymes to help resolution of fibrosis (Rodier and Campisi, 2011; Jun and Lau, 2010b; Jun and Lau, 2010a).
Entity name
Diabetic retinopathy and nephropathy
Note
Cyr61 may contribute to diabetic retinopathy. Its expression is induced by advanced glycation end products (Hughes et al., 2007). In proliferative diabetic retinal membrane CYR61 is induced in ganglion cells, endothelial cells, and smooth muscle cells, where it may contribute to the loss of normal capillary pericytes by apoptosis (Liu et al., 2008) and enhancement of aberrant neovascularization (Zhang et al., 2012). Elevated CYR61 in vitreous is detected in patients with proliferative diabetic retinopathy (You et al., 2012). In the normal adult kidney CYR61 is expressed by podocytes and tubular epithelial cells, where CYR61 may function to maintain normal podocyte differentiation, but suppress mesangial cell adhesion and migration (Sawai et al., 2007). Podocyte expression of Cyr61 is diminished in patients with diabetic nephropathy, and this may be associated with mesangial expansion in glomerulopathies (Sawai et al., 2007).
Entity name
Vascular disease
Note
CYR61 is induced in human arteriosclerotic lesions (Hilfiker et al., 2002) and inflammatory cardiomyopathy (Rother et al., 2010).
Entity name
Inflammatory diseases
Note
CYR61 is highly expressed in rheumatoid arthritis (Zhang et al., 2009) as well as in patients with Crohns disease or ulcerative colitis (Koon et al., 2008).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 9600969 | 1998 | CYR61, a product of a growth factor-inducible immediate early gene, promotes angiogenesis and tumor growth. | Babic AM et al |
| 20164416 | 2010 | Matricellular protein CCN1 activates a proinflammatory genetic program in murine macrophages. | Bai T et al |
| 17718860 | 2007 | CYR61 polymorphisms are associated with plasma HDL-cholesterol levels in obese individuals. | Bouchard L et al |
| 12665631 | 2003 | Proposal for a unified CCN nomenclature. | Brigstock DR et al |
| 11120741 | 2001 | The angiogenic factors Cyr61 and connective tissue growth factor induce adhesive signaling in primary human skin fibroblasts. | Chen CC et al |
| 18775791 | 2009 | Functions and mechanisms of action of CCN matricellular proteins. | Chen CC et al |
| 11584015 | 2001 | The angiogenic factor Cyr61 activates a genetic program for wound healing in human skin fibroblasts. | Chen CC et al |
| 17318182 | 2007 | Cytotoxicity of TNFalpha is regulated by integrin-mediated matrix signaling. | Chen CC et al |
| 10821835 | 2000 | Adhesion of human skin fibroblasts to Cyr61 is mediated through integrin alpha 6beta 1 and cell surface heparan sulfate proteoglycans. | Chen N et al |
| 15308622 | 2004 | Identification of a novel integrin alphavbeta3 binding site in CCN1 (CYR61) critical for pro-angiogenic activities in vascular endothelial cells. | Chen N et al |
| 17579708 | 2007 | Expression of Cyr61, CTGF, and WISP-1 correlates with clinical features of lung cancer. | Chen PP et al |
| 15471875 | 2004 | Cyr61 suppresses growth of human endometrial cancer cells. | Chien W et al |
| 20172544 | 2010 | Extracellular matrix associated protein CYR61 is linked to prostate cancer development. | D'Antonio KB et al |
| 12215267 | 2002 | Stimulation of angiogenesis by Cyr61 gene: a new therapeutic candidate. | Fataccioli V et al |
| 20142256 | 2010 | Colorectal cancer susceptibility quantitative trait loci in mice as a novel approach to detect low-penetrance variants in humans: a two-stage case-control study. | Fernández-Rozadilla C et al |
| 19584265 | 2009 | Matrix protein CCN1 is critical for prostate carcinoma cell proliferation and TRAIL-induced apoptosis. | Franzen CA et al |
| 21312266 | 2011 | CYR61 downregulation reduces osteosarcoma cell invasion, migration, and metastasis. | Fromigue O et al |
| 16243794 | 2005 | Ovarian carcinomas: CCN genes are aberrantly expressed and CCN1 promotes proliferation of these cells. | Gery S et al |
| 20233866 | 2010 | Cyr61 mediates hepatocyte growth factor-dependent tumor cell growth, migration, and Akt activation. | Goodwin CR et al |
| 11897702 | 2002 | The angiogenic factor cysteine-rich 61 (CYR61, CCN1) supports vascular smooth muscle cell adhesion and stimulates chemotaxis through integrin alpha(6)beta(1) and cell surface heparan sulfate proteoglycans. | Grzeszkiewicz TM et al |
| 18480812 | 2008 | Wound repair and regeneration. | Gurtner GC et al |
| 10841170 | 2000 | Temporal expression of the chondrogenic and angiogenic growth factor CYR61 during fracture repair. | Hadjiargyrou M et al |
| 23027863 | 2012 | The matricellular protein CCN1/Cyr61 is a critical regulator of Sonic Hedgehog in pancreatic carcinogenesis. | Haque I et al |
| 21232118 | 2011 | Cyr61/CCN1 signaling is critical for epithelial-mesenchymal transition and stemness and promotes pancreatic carcinogenesis. | Haque I et al |
| 12105167 | 2002 | Expression of CYR61, an angiogenic immediate early gene, in arteriosclerosis and its regulation by angiotensin II. | Hilfiker A et al |
| 15117851 | 2004 | Regulation of proangiogenic factor CCN1 in cardiac muscle: impact of ischemia, pressure overload, and neurohumoral activation. | Hilfiker-Kleiner D et al |
| 21447598 | 2011 | Expression of tumor-promoting Cyr61 is regulated by hTRA2-β1 and acidosis. | Hirschfeld M et al |
| 18789696 | 2008 | The CCN family of proteins: structure-function relationships. | Holbourn KP et al |
| 17333105 | 2007 | Advanced glycation end products cause increased CCN family and extracellular matrix gene expression in the diabetic rodent retina. | Hughes JM et al |
| 10446209 | 1999 | Activation-dependent adhesion of human platelets to Cyr61 and Fisp12/mouse connective tissue growth factor is mediated through integrin alpha(IIb)beta(3). | Jedsadayanmata A et al |
| 10557283 | 1999 | Identification of eukaryotic mRNAs that are translated at reduced cap binding complex eIF4F concentrations using a cDNA microarray. | Johannes G et al |
| 22129992 | 2011 | Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. | Jun JI et al |
| 22363611 | 2012 | TNFα-induced apoptosis enabled by CCN1/CYR61: pathways of reactive oxygen species generation and cytochrome c release. | Juric V et al |
| 9446626 | 1998 | Adhesion of human umbilical vein endothelial cells to the immediate-early gene product Cyr61 is mediated through integrin alphavbeta3. | Kireeva ML et al |
| 18599605 | 2008 | Substance P-mediated expression of the pro-angiogenic factor CCN1 modulates the course of colitis. | Koon HW et al |
| 21081514 | 2011 | Expression of CTGF and Cyr61 in colorectal cancer. | Ladwa R et al |
| 21349946 | 2011 | Taxol resistance in breast cancer cells is mediated by the hippo pathway component TAZ and its downstream transcriptional targets Cyr61 and CTGF. | Lai D et al |
| 11356704 | 2001 | Promoter function of the angiogenic inducer Cyr61gene in transgenic mice: tissue specificity, inducibility during wound healing, and role of the serum response element. | Latinkic BV et al |
| 10094812 | 1999 | The CCN family of angiogenic regulators: the integrin connection. | Lau LF et al |
| 21805345 | 2011 | CCN1/CYR61: the very model of a modern matricellular protein. | Lau LF et al |
| 11810026 | 2002 | Organization and expression of the Cyr61 gene in normal human fibroblasts. | Leng E et al |
| 12364323 | 2002 | Pro-angiogenic activities of CYR61 (CCN1) mediated through integrins alphavbeta3 and alpha6beta1 in human umbilical vein endothelial cells. | Leu SJ et al |
| 12826661 | 2003 | Identification of a novel integrin alpha 6 beta 1 binding site in the angiogenic inducer CCN1 (CYR61). | Leu SJ et al |
| 16435358 | 2006 | CYR61 (CCN1) protein expression during fracture healing in an ovine tibial model and its relation to the mechanical fixation stability. | Lienau J et al |
| 15044484 | 2004 | Cyr61 expression confers resistance to apoptosis in breast cancer MCF-7 cells by a mechanism of NF-kappaB-dependent XIAP up-regulation. | Lin MT et al |
| 18187544 | 2008 | Cysteine-rich protein 61 and connective tissue growth factor induce deadhesion and anoikis of retinal pericytes. | Liu H et al |
| 19180570 | 2009 | Cyr61 is up-regulated in prostate cancer and associated with the p53 gene status. | Lv H et al |
| 15592521 | 2005 | A novel CYR61-triggered 'CYR61-alphavbeta3 integrin loop' regulates breast cancer cell survival and chemosensitivity through activation of ERK1/ERK2 MAPK signaling pathway. | Menendez JA et al |
| 17023674 | 2006 | The matricellular protein CCN1 is essential for cardiac development. | Mo FE et al |
| 12446788 | 2002 | CYR61 (CCN1) is essential for placental development and vascular integrity. | Mo FE et al |
| 18058471 | 2007 | CYR61: a new measure of lung cancer outcome. | Mori A et al |
| 21053272 | 2010 | A coding RNA segment that enhances the ribosomal recruitment of chicken ccn1 mRNA. | Mukudai Y et al |
| 2355916 | 1990 | Expression of cyr61, a growth factor-inducible immediate-early gene. | O'Brien TP et al |
| 21484592 | 2011 | Spatial-temporal modulation of CCN proteins during wound healing in human skin in vivo. | Rittié L et al |
| 21321098 | 2011 | Four faces of cellular senescence. | Rodier F et al |
| 21135363 | 2010 | Matricellular signaling molecule CCN1 attenuates experimental autoimmune myocarditis by acting as a novel immune cell migration modulator. | Rother M et al |
| 21976359 | 2012 | Cyr61 expression in osteosarcoma indicates poor prognosis and promotes intratibial growth and lung metastasis in mice. | Sabile AA et al |
| 14988385 | 2004 | Increased expression of CYR61, an extracellular matrix signaling protein, in human benign prostatic hyperplasia and its regulation by lysophosphatidic acid. | Sakamoto S et al |
| 17699553 | 2007 | Expression of CCN1 (CYR61) in developing, normal, and diseased human kidney. | Sawai K et al |
| 12036876 | 2002 | Identification of integrin alpha(M)beta(2) as an adhesion receptor on peripheral blood monocytes for Cyr61 (CCN1) and connective tissue growth factor (CCN2): immediate-early gene products expressed in atherosclerotic lesions. | Schober JM et al |
| 9002679 | 1997 | Identification of a complex congenital heart defect susceptibility locus by using DNA pooling and shared segment analysis. | Sheffield VC et al |
| 12177410 | 2002 | Gene expression during the priming phase of liver regeneration after partial hepatectomy in mice. | Su AI et al |
| 11598125 | 2001 | Cyr61, a member of CCN family, is a tumor suppressor in non-small cell lung cancer. | Tong X et al |
| 12444554 | 2002 | Cyr61 promotes breast tumorigenesis and cancer progression. | Tsai MS et al |
| 19007976 | 2009 | Cyr61, a member of ccn (connective tissue growth factor/cysteine-rich 61/nephroblastoma overexpressed) family, predicts survival of patients with endometrial cancer of endometrioid subtype. | Watari H et al |
| 11751417 | 2001 | Elevated levels of connective tissue growth factor, WISP-1, and CYR61 in primary breast cancers associated with more advanced features. | Xie D et al |
| 15041728 | 2004 | Levels of expression of CYR61 and CTGF are prognostic for tumor progression and survival of individuals with gliomas. | Xie D et al |
| 1782153 | 1991 | Cyr61, product of a growth factor-inducible immediate early gene, is associated with the extracellular matrix and the cell surface. | Yang GP et al |
| 21822163 | 2012 | Elevation of angiogenic factor Cysteine-rich 61 levels in vitreous of patients with proliferative diabetic retinopathy. | You JJ et al |
| 19950293 | 2009 | A critical role of Cyr61 in interleukin-17-dependent proliferation of fibroblast-like synoviocytes in rheumatoid arthritis. | Zhang Q et al |
| 22160564 | 2012 | Cysteine-rich 61 (CYR61) is up-regulated in proliferative diabetic retinopathy. | Zhang X et al |
| 20452491 | 2010 | MicroRNA-155 contributes to preeclampsia by down-regulating CYR61. | Zhang Y et al |
Citation
Chih-Chiun Chen ; Lester F Lau
CYR61 (cysteine-rich, angiogenic inducer, 61)
Atlas Genet Cytogenet Oncol Haematol. 2013-01-01
Online version: http://atlasgeneticsoncology.org/gene/40256/cyr61
