SPINK1 (Serine Peptidase Inhibitor, Kazal Type 1)
2014-12-01 Hannu Koistinen , Outi Itkonen , Ulf-Hakan Stenman AffiliationDepartment of Clinical Chemistry, University of Helsinki (HK,OI, UHS),, Laboratory Division HUSLAB, Helsinki University Central Hospital (OI), Helsinki, Finland [email protected]; [email protected]; [email protected]
Identity
Abstract
Review on SPINK1, with data on DNA, on the protein encoded, and where the gene is implicated.
DNA/RNA
Note
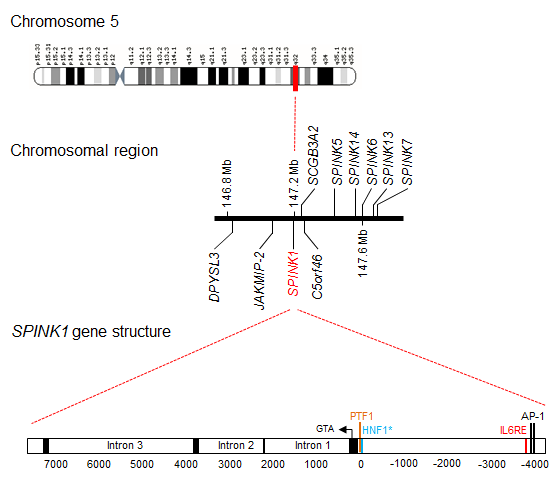
Description
Transcription
Proteins
Note
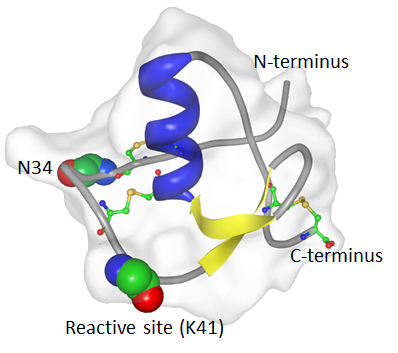
Description
Expression
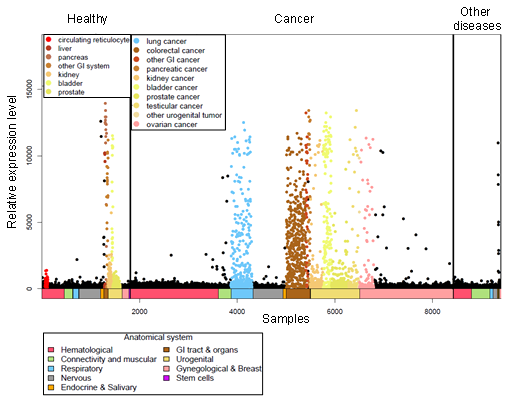
Localisation
Function
Homology
Mutations
Note
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 21368222 | 2011 | Therapeutic targeting of SPINK1-positive prostate cancer. | Ateeq B et al |
| 12360463 | 2002 | Tropical calcific pancreatitis: strong association with SPINK1 trypsin inhibitor mutations. | Bhatia E et al |
| 22343981 | 2012 | Functional analysis of eight missense mutations in the SPINK1 gene. | Boulling A et al |
| 19453252 | 2009 | Chronic pancreatitis: genetics and pathogenesis. | Chen JM et al |
| 4231655 | 1967 | [On protease inhibitors, V. On the chemistry and physiology of the specific trypsin inhibitors from the ox, dog, pig and human pancreas]. | Fritz H et al |
| 19384300 | 2009 | High expression of tumour-associated trypsin inhibitor correlates with liver metastasis and poor prognosis in colorectal cancer. | Gaber A et al |
| 20849596 | 2010 | Increased serum levels of tumour-associated trypsin inhibitor independently predict a poor prognosis in colorectal cancer patients. | Gaber A et al |
| 18514770 | 2008 | Evaluation of urine tumor-associated trypsin inhibitor, CYFRA 21-1, and urinary bladder cancer antigen for detection of high-grade bladder carcinoma. | Gkialas I et al |
| 5928200 | 1966 | Trypsin inhibitor from bovine pancreatic juice. | Greene LJ et al |
| 23843146 | 2013 | SPINK1 expression is tightly linked to 6q15- and 5q21-deleted ERG-fusion negative prostate cancers but unrelated to PSA recurrence. | Grupp K et al |
| 1870127 | 1991 | Three-dimensional structure of the complexes between bovine chymotrypsinogen A and two recombinant variants of human pancreatic secretory trypsin inhibitor (Kazal-type). | Hecht HJ et al |
| 3501289 | 1987 | Primary structure of human pancreatic secretory trypsin inhibitor (PSTI) gene. | Horii A et al |
| 16327984 | 2006 | Differential expression of trypsinogen and tumor-associated trypsin inhibitor (TATI) in bladder cancer. | Hotakainen K et al |
| 6384012 | 1984 | Demonstration of a new acrosin inhibitor in human seminal plasma. | Huhtala ML et al |
| 6190763 | 1983 | Excretion of a tumor-associated trypsin inhibitor (TATI) in urine of patients with gynecological malignancy. | Huhtala ML et al |
| 4429557 | 1974 | Epidermal growth factor: internal duplication and probable relationship to pancreatic secretory trypsin inhibitor. | Hunt LT et al |
| 24583226 | 2014 | TATI as a biomarker. | Itkonen O et al |
| 21296855 | 2011 | Global cancer statistics. | Jemal A et al |
| 18882536 | 1948 | Isolation of a crystalline trypsin inhibitor-anticoagulant protein from pancreas. | KAZAL LA et al |
| 12893328 | 2003 | Tumor-associated trypsin inhibitor as a prognostic factor during follow-up of bladder cancer. | Kelloniemi E et al |
| 18803840 | 2008 | Systematic bioinformatic analysis of expression levels of 17,330 human genes across 9,783 samples from 175 types of healthy and pathological tissues. | Kilpinen S et al |
| 17525091 | 2007 | Missense mutations in pancreatic secretory trypsin inhibitor (SPINK1) cause intracellular retention and degradation. | Király O et al |
| 22526274 | 2012 | Do genetic variants in the SPINK1 gene affect the level of serum PSTI? | Kume K et al |
| 19864383 | 2010 | Hepatitis B and hepatitis C virus replication upregulates serine protease inhibitor Kazal, resulting in cellular resistance to serine protease-dependent apoptosis. | Lamontagne J et al |
| 17267202 | 2007 | Overexpression of tumour-associated trypsin inhibitor (TATI) enhances tumour growth and is associated with portal vein invasion, early recurrence and a stage-independent prognostic factor of hepatocellular carcinoma. | Lee YC et al |
| 24083995 | 2013 | Loss of PTEN is associated with aggressive behavior in ERG-positive prostate cancer. | Leinonen KA et al |
| 23459095 | 2013 | A high-density tissue microarray from patients with clinically localized prostate cancer reveals ERG and TATI exclusivity in tumor cells. | Lippolis G et al |
| 22043941 | 2011 | Role of the inflammatory protein serine protease inhibitor Kazal in preventing cytolytic granule granzyme A-mediated apoptosis. | Lu F et al |
| 23889187 | 2013 | Prognostic significance of tumor-associated trypsin inhibitor (TATI) and human chorionic gonadotropin-β (hCGβ) in patients with hepatocellular carcinoma. | Lyytinen I et al |
| 8774127 | 1996 | Distribution and expression of pancreatic secretory trypsin inhibitor and its possible role in epithelial restitution. | Marchbank T et al |
| 23527199 | 2013 | Global gene expression profiling reveals SPINK1 as a potential hepatocellular carcinoma marker. | Marshall A et al |
| 7601186 | 1995 | Tumour-associated trypsin inhibitor and renal cell carcinoma. | Meria P et al |
| 3428272 | 1987 | A secretory protease inhibitor requires androgens for its expression in male sex accessory tissues but is expressed constitutively in pancreas. | Mills JS et al |
| 15694009 | 2005 | The Molecular Biology Toolkit (MBT): a modular platform for developing molecular visualization applications. | Moreland JL et al |
| 2170560 | 1990 | Identification and characterization of receptors specific for human pancreatic secretory trypsin inhibitor. | Niinobu T et al |
| 2449986 | 1988 | Pancreatic secretory trypsin inhibitor as an acute phase reactant. | Ogawa M et al |
| 8244568 | 1993 | Specific expression of the pancreatic-secretory-trypsin-inhibitor (PSTI) gene in hepatocellular carcinoma. | Ohmachi Y et al |
| 16083722 | 2005 | Autophagic cell death of pancreatic acinar cells in serine protease inhibitor Kazal type 3-deficient mice. | Ohmuraya M et al |
| 19737965 | 2009 | Serine protease inhibitor Kazal type 1 promotes proliferation of pancreatic cancer cells through the epidermal growth factor receptor. | Ozaki N et al |
| 17306443 | 2007 | Increased expression of tumor-associated trypsin inhibitor, TATI, in prostate cancer and in androgen-independent 22Rv1 cells. | Paju A et al |
| 11176522 | 2001 | Tumor associated trypsin inhibitor as a prognostic factor in renal cell carcinoma. | Paju A et al |
| 16517420 | 2006 | Biochemistry and clinical role of trypsinogens and pancreatic secretory trypsin inhibitor. | Paju A et al |
| 15269150 | 2004 | Expression of trypsinogen-1, trypsinogen-2, and tumor-associated trypsin inhibitor in ovarian cancer: prognostic study on tissue and serum. | Paju A et al |
| 7667604 | 1995 | Tumour-associated trypsin inhibitor (TATI) in patients with colorectal cancer: a comparison with CEA, CA 50 and CA 242. | Pasanen P et al |
| 21739120 | 2012 | Association of tumor-associated trypsin inhibitor (TATI) expression with molecular markers, pathologic features and clinical outcomes of urothelial carcinoma of the urinary bladder. | Patschan O et al |
| 10982753 | 2000 | SPINK1/PSTI polymorphisms act as disease modifiers in familial and idiopathic chronic pancreatitis. | Pfützer RH et al |
| 4818832 | 1974 | Trypsin inhibitor from human pancreas and pancreatic juice. | Pubols MH et al |
| 16029158 | 2005 | Human mesotrypsin defies natural trypsin inhibitors: from passive resistance to active destruction. | Sahin-Tóth M et al |
| 15963628 | 2005 | Urinary levels of tumor-associated trypsin inhibitor (TATI) in the detection of transitional cell carcinoma of the urinary bladder. | Shariat SF et al |
| 14675563 | 2004 | Tumour-associated trypsin inhibitor, carcinoembryonic antigen and acute-phase reactant proteins CRP and alpha1-antitrypsin in patients with gastrointestinal malignancies. | Solakidi S et al |
| 21656687 | 2011 | Combined genomic and phenotype screening reveals secretory factor SPINK1 as an invasion and survival factor associated with patient prognosis in breast cancer. | Soon WW et al |
| 3202976 | 1988 | Characterization of a tumor-associated serine protease. | Stenman UH et al |
| 1780686 | 1991 | Evaluation of TATI and other markers in solid tumors. | Taccone W et al |
| 18538735 | 2008 | The role of SPINK1 in ETS rearrangement-negative prostate cancers. | Tomlins SA et al |
| 16863567 | 2006 | Relationship between pancreatic secretory trypsin inhibitor and early recurrence of intrahepatic cholangiocarcinoma following surgical resection. | Tonouchi A et al |
| 12590650 | 2003 | Purification and identification of a binding protein for pancreatic secretory trypsin inhibitor: a novel role of the inhibitor as an anti-granzyme A. | Tsuzuki S et al |
| 2461702 | 1988 | Reaction of a tumour-associated trypsin inhibitor with serine proteinases associated with coagulation and tumour invasion. | Turpeinen U et al |
| 9637119 | 1998 | Pre-operative serum level of tumour-associated trypsin inhibitor and residual tumour size as prognostic indicators in Stage III epithelial ovarian cancer. | Venesmaa P et al |
| 24600409 | 2014 | Pancreatic cancer risk in hereditary pancreatitis. | Weiss FU et al |
| 18751407 | 2008 | Comparison of the prognostic value of a panel of tissue tumor markers and established clinicopathological factors in patients with gastric cancer. | Wiksten JP et al |
| 10835640 | 2000 | Mutations in the gene encoding the serine protease inhibitor, Kazal type 1 are associated with chronic pancreatitis. | Witt H et al |
| 19956577 | 2009 | Generating triangulated macromolecular surfaces by Euclidean Distance Transform. | Xu D et al |
| 3877508 | 1985 | Molecular cloning and nucleotide sequence of human pancreatic secretory trypsin inhibitor (PSTI) cDNA. | Yamamoto T et al |
| 7691687 | 1993 | Identification of the IL-6-responsive element in an acute-phase-responsive human pancreatic secretory trypsin inhibitor-encoding gene. | Yasuda T et al |
| 9852108 | 1998 | Identification of novel pancreas-specific regulatory sequences in the promoter region of human pancreatic secretory trypsin inhibitor gene. | Yasuda T et al |
Other Information
Locus ID:
NCBI: 6690
MIM: 167790
HGNC: 11244
Ensembl: ENSG00000164266
Variants:
dbSNP: 6690
ClinVar: 6690
TCGA: ENSG00000164266
COSMIC: SPINK1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000164266 | ENST00000296695 | P00995 |
| ENSG00000164266 | ENST00000510027 | D6RIU5 |
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38781741 | 2024 | High expression of serine protease inhibitor kazal type 1 predicts poor prognosis and promotes the progression and invasion of oral tongue squamous cell carcinoma. | 0 |
| 38781741 | 2024 | High expression of serine protease inhibitor kazal type 1 predicts poor prognosis and promotes the progression and invasion of oral tongue squamous cell carcinoma. | 0 |
| 34807350 | 2023 | Hereditary Pancreatitis Related to SPINK-1 Mutation. Is There an Increased Risk of Developing Pancreatic Cancer? | 2 |
| 36883543 | 2023 | [Influence of environmental factors and polymorphic loci rs6580502 of the SPINK1 gene, rs10273639 of the PRSS1 gene, rs213950 of the CFTR gene on the risk of developing acute alcoholic-alimentary pancreatitis]. | 0 |
| 34807350 | 2023 | Hereditary Pancreatitis Related to SPINK-1 Mutation. Is There an Increased Risk of Developing Pancreatic Cancer? | 2 |
| 36883543 | 2023 | [Influence of environmental factors and polymorphic loci rs6580502 of the SPINK1 gene, rs10273639 of the PRSS1 gene, rs213950 of the CFTR gene on the risk of developing acute alcoholic-alimentary pancreatitis]. | 0 |
| 34794144 | 2022 | TATI, TAT-2, and CRP as Prognostic Factors in Colorectal Cancer. | 2 |
| 35050902 | 2022 | Collision tumors revealed by prospectively assessing subtype-defining molecular alterations in 904 individual prostate cancer foci. | 4 |
| 35102613 | 2022 | Nuclear magnetic resonance spectroscopy reveals dysregulation of monounsaturated fatty acid metabolism upon SPINK1 attenuation in colorectal cancer. | 0 |
| 35194087 | 2022 | Raised SPINK1 levels play a role in angiogenesis and the transendothelial migration of ALL cells. | 1 |
| 35408828 | 2022 | Structural and Biophysical Insights into SPINK1 Bound to Human Cationic Trypsin. | 3 |
| 35924241 | 2022 | Serine Protease Inhibitor Kazal Type 1, A Potential Biomarker for the Early Detection, Targeting, and Prediction of Response to Immune Checkpoint Blockade Therapies in Hepatocellular Carcinoma. | 5 |
| 36053457 | 2022 | Serine protease inhibitor Kazal type 1 (SPINK1) promotes proliferation, migration, invasion and radiation resistance in rectal cancer patients receiving concurrent chemoradiotherapy: a potential target for precision medicine. | 3 |
| 36112498 | 2022 | The roles of mutated SPINK1 gene in prostate cancer cells. | 0 |
| 36194314 | 2022 | Identification and Exploration of Serine Peptidase Inhibitor Kazal Type I (SPINK1) as a Potential Biomarker Correlated with the Progression of Non-Small Cell Lung Cancer. | 1 |
Citation
Hannu Koistinen ; Outi Itkonen ; Ulf-Hakan Stenman
SPINK1 (Serine Peptidase Inhibitor, Kazal Type 1)
Atlas Genet Cytogenet Oncol Haematol. 2014-12-01
Online version: http://atlasgeneticsoncology.org/gene/42375/spink1-(serine-peptidase-inhibitor-kazal-type-1)
