TBX3 (T-box 3)
2013-01-01 Reyna Deeya Ballim , Sharon Prince AffiliationDepartment of Human Biology, Faculty of Health Sciences, University of Cape Town, Cape Town, South Africa, 7925
Identity
HGNC
LOCATION
12q24.1
LOCUSID
ALIAS
TBX3-ISO,UMS,XHL
FUSION GENES
DNA/RNA
Note
Tbx2 and Tbx3 both belong to the Tbx2 subfamily of T-box factors (Agulnik et al., 1996). Phylogenetic analysis suggests that this subfamily originated from a single ancestral gene which was duplicated by unequal crossing over to form a two-gene cluster (Tbx2/3 and Tbx4/5) and at some point in the vertebrate lineage, duplicated again to form four separate genes with Tbx2 and Tbx4 linked on chromosome 11, and Tbx3 and Tbx5 on chromosome 5 (chromosomes 17q23 and 12q24 respectively in humans; Figure 1A) (Campbell et al., 1995; Agulnik et al., 1996; Bamshad et al., 1997). Due to the original duplication event, Tbx2 and Tbx3 form a closely related gene pair while Tbx4 and Tbx5 are more closely related to each other (Agulnik et al., 1996).
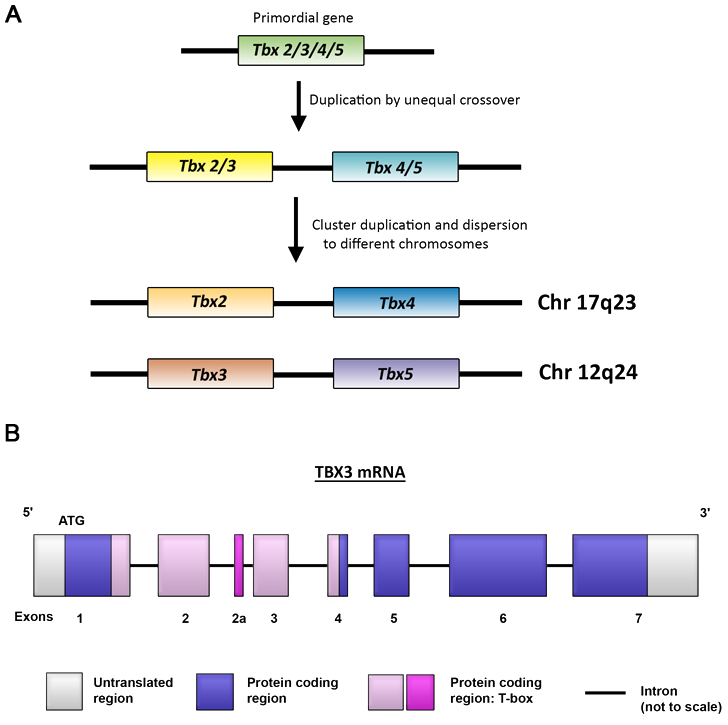
Figure 1: A) Drawing of gene duplication. B) Drawing of TBX3 mRNA. (A) Model describing evolution of Tbx 2/3/4/5 gene family (based on Agulnick et al, 1996). (B) Schematic representation of human TBX3 mRNA depicting relative size and position of exons. In the TBX3 + 2a isoform, exon +2a is included in the mRNA through alternative splicing of the second intron.
Description
The human TBX3 gene is found on the reverse strand of chromosome 12 and spans 13.9 kb.
Transcription
Four TBX3 transcripts have been identified but only two encode for full length functional proteins viz TBX3 and TBX3 + 2a (Figure 1B). The TBX3 mRNA is 4.7 kb and contains 7 exons encoding a protein of 723 amino acids. Alternative splicing of the intronic region between exons 2 and 3 gives rise to the TBX3 + 2a transcript which contains an extra 60 bp sequence designated exon 2a leading to the production of a 743 amino acid protein (Bamshad et al., 1999). TBX3 and TBX3 + 2a isoforms are widely expressed in mouse and human tissues with TBX3 generally observed to be the dominant isoform and the ratio between the two being both tissue and species dependent (Fan et al., 2004).
Pseudogene
No pseudogenes have been identified for TBX3 to date.
Proteins
Note
The TBX3 + 2a transcript yields a protein with an extra 20 amino acids in the middle of the T-box DNA binding domain, giving rise to speculation that it may affect the DNA-binding ability of the protein (Bamshad et al., 1999). While the work of Fan et al. (2004) has provided evidence supporting this hypothesis, subsequent studies have shown no functional difference between the TBX3 and TBX3 + 2a proteins (Hoogaars et al., 2008; Rodriguez et al., 2008).
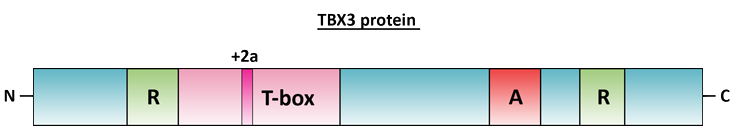
Figure 2. Schematic representation of the human TBX3 protein. The domains depicted above are: T-box, DNA binding domain (pink); R, repression domain; A, putative activation domain. The amino and carboxy termini of the protein are labelled N and C respectively.
Description
The T-box DNA binding domain is found in the N-terminal half of the TBX3 protein extending from amino acids 104 - 285 and in the TBX3 + 2a isoform, the additional 20 amino acids are inserted into the middle of the T-box at position 219 (Figure 2). TBX3 has two repression domains, one in the N-terminus and one in the C-terminus of the protein, as well as a putative activation domain located in the C-terminal end of the protein (Figure 2) (Carlson et al., 2001).
Expression
In humans, TBX3 is expressed in a number of organs, including foetal heart, liver, spleen, lung and kidney, and in adult prostate, lung, placenta, ovary, spleen, heart, kidney, testis, small intestine, adrenal gland, thyroid, breast, bladder, uterus, liver and salivary gland (Bamshad et al., 1999).
Localisation
The TBX3 protein is predominantly nuclear.
Function
TBX3 has been described to function as a transcriptional repressor and to date was shown to directly repress p14, p21, E-cadherin and phosphatase and TENsin homolog (PTEN) (Lingbeek et al., 2002; Hoogaars et al., 2008; Rodriguez et al., 2008; Burgucu et al., 2012). While in vitro assays have suggested that TBX3 is capable of transcriptional activation, it has not yet been shown to activate any physiologically relevant target genes.
Homology
Human TBX3 shares 98% amino acid identity with mouse Tbx3 and homologs have been identified in mammals, reptiles, fish and amphibians, as well as invertebrates such as tunicates (Bamshad et al., 1997).
Mutations
Germinal
Mutations in human TBX3 have been linked with ulnar-mammary syndrome and those described to date include frame shift, premature termination and missense mutations (Bamshad et al., 1997; Bamshad et al.,1999).
Implicated in
Entity name
Ulnar-mammary syndrome
Note
Results from congenital mutations in a single copy of the human TBX3 gene.
Disease
Characterised by posterior limb abnormalities, such as malformed ulna and posterior digits, hypoplasia and/or dysfunction of the mammary and apocrine glands, absent axillary hair, abnormal dentition, delayed puberty in males and genital anomalies (Bamshad et al., 1996; Bamshad et al., 1997).
Entity name
Breast cancer
Note
Breast cancer lines screened by real time PCR analysis displayed increased expression of TBX3 in 15 of 28 cell lines tested (Fan et al., 2004). When TBX3 expression was examined by immunohistochemistry in breast tumour tissues, the results showed that levels of TBX3 protein were higher in tumour tissue compared to adjacent normal tissue, with increased cytoplasmic localisation.
Entity name
Pancreatic cancer
Note
Microarray analyses comparing non-metastatic and metastatic pancreatic endocrine neoplasms revealed that TBX3 expression is upregulated in the latter tumour type (Hansel et al., 2004).
Entity name
Ovarian cancer
Note
Using 2D gel electrophoresis and matrix-assisted laser desorption/ionization (MALDI) time of flight (TOF) mass spectrometry, a truncated form of TBX3 was detected in blood plasma from ovarian cancer patients (Lomnytska et al., 2006).
Entity name
Liver cancer
Note
Increased TBX3 expression was shown to correlate with a mutant active form of β-catenin in both human and mouse hepatocellular carcinomas (HCCs) and human hepatoblastomas. Renard et al. (2007) demonstrated TBX3 to be involved in β-catenins activation of cell proliferation in the human hepatoma cell line HepG2. Using in vitro assays they showed that expression of mutant β-catenin upregulated TBX3 expression and that this was due to direct binding of active β-catenin together with its co-activator T-cell factor (Tcf) to a Tcf-binding element in the TBX3 promoter (Renard et al., 2007).
Entity name
Glioblastoma
Note
Genome-wide DNA methylation profiling of 55 glioblastoma tissue samples compared to non-neoplastic brains revealed that methylation of TBX3 correlated with decreased overall survival, identifying it as a potential independent prognostic marker (Etcheverry et al., 2010).
Entity name
Gastric cancer
Note
Genome-wide screening identified TBX3 to be epigenetically silenced in the gastric cancer cell line AGS and the TBX3 gene was shown to be methylated in 7 out of 10 primary gastric cancers (Yamashita et al., 2006).
Entity name
Uterine cervical cancer
Note
TBX3 expression was shown to be downregulated in microarray analyses comparing lymph node positive to lymph node negative cervical tumours. A significant correlation was observed between low TBX3 expression and the metastatic phenotype. In addition, multivariate analysis identified TBX3 as a potential independent prognostic marker for this cancer (Lyng et al., 2006).
Entity name
Melanoma
Note
In 2004, Hoek et al. showed that compared to normal melanocytes TBX3 expression was upregulated in at least 4 of 6 melanoma cell lines tested and Rodriguez et al. (2008) later demonstrated increased TBX3 protein levels in 6 out of 12 melanoma cell lines. Furthermore, TBX3 was shown to contribute to melanoma formation, migration and invasion by a process involving its ability to repress the cell adhesion molecule E-cadherin (Rodriguez et al., 2008; Peres et al., 2010).
Entity name
Head and neck squamous cell cancer
Note
TBX3 is upregulated in head and neck squamous cell carcinoma (HNSCC) cell lines and tissues (Humtsoe et al., 2011; Burgucu et al., 2012). The study by Humtsoe et al. shows that TBX3 expression is specifically upregulated in HNSCC cells which display characteristics of epithelial to mesenchymal transition (EMT). Interestingly, however they show that TBX3 promotes cell survival and to a lesser extent, cell invasion in these cells (Humtsoe et al., 2011). This is consistent with the results of Burgucu et al. (2012) which demonstrate that TBX3 represses the tumour suppressor, phosphatase and TENsin homolog (PTEN).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 8878690 | 1996 | Evolution of mouse T-box genes by tandem duplication and cluster dispersion. | Agulnik SI et al |
| 10330342 | 1999 | The spectrum of mutations in TBX3: Genotype/Phenotype relationship in ulnar-mammary syndrome. | Bamshad M et al |
| 8923944 | 1996 | Clinical analysis of a large kindred with the Pallister ulnar-mammary syndrome. | Bamshad M et al |
| 7920656 | 1994 | An ancient family of embryonically expressed mouse genes sharing a conserved protein motif with the T locus. | Bollag RJ et al |
| 11748239 | 2002 | TBX-3, the gene mutated in Ulnar-Mammary Syndrome, is a negative regulator of p19ARF and inhibits senescence. | Brummelkamp TR et al |
| 23082988 | 2012 | Tbx3 represses PTEN and is over-expressed in head and neck squamous cell carcinoma. | Burgucu D et al |
| 8530034 | 1995 | Cloning and mapping of a human gene (TBX2) sharing a highly conserved protein motif with the Drosophila omb gene. | Campbell C et al |
| 11689487 | 2001 | A dominant repression domain in Tbx3 mediates transcriptional repression and cell immortalization: relevance to mutations in Tbx3 that cause ulnar-mammary syndrome. | Carlson H et al |
| 12668638 | 2003 | Mammary gland, limb and yolk sac defects in mice lacking Tbx3, the gene mutated in human ulnar mammary syndrome. | Davenport TG et al |
| 21156036 | 2010 | DNA methylation in glioblastoma: impact on gene expression and clinical outcome. | Etcheverry A et al |
| 15289316 | 2004 | TBX3 and its isoform TBX3+2a are functionally distinctive in inhibition of senescence and are overexpressed in a subset of breast cancer cell lines. | Fan W et al |
| 15448002 | 2004 | Met proto-oncogene and insulin-like growth factor binding protein 3 overexpression correlates with metastatic ability in well-differentiated pancreatic endocrine neoplasms. | Hansel DE et al |
| 15289333 | 2004 | Expression profiling reveals novel pathways in the transformation of melanocytes to melanomas. | Hoek K et al |
| 18444963 | 2008 | TBX3 and its splice variant TBX3 + exon 2a are functionally similar. | Hoogaars WM et al |
| 22154512 | 2012 | Transcriptional profiling identifies upregulated genes following induction of epithelial-mesenchymal transition in squamous carcinoma cells. | Humtsoe JO et al |
| 15694670 | 2005 | Tbx3 expression is related to apoptosis and cell proliferation in rat bladder both hyperplastic epithelial cells and carcinoma cells. | Ito A et al |
| 8344258 | 1993 | The Brachyury gene encodes a novel DNA binding protein. | Kispert A et al |
| 12000749 | 2002 | The T-box repressors TBX2 and TBX3 specifically regulate the tumor suppressor gene p14ARF via a variant T-site in the initiator. | Lingbeek ME et al |
| 16049973 | 2006 | Increased expression of cSHMT, Tbx3 and utrophin in plasma of ovarian and breast cancer patients. | Lomnytska M et al |
| 17054779 | 2006 | Gene expressions and copy numbers associated with metastatic phenotypes of uterine cervical cancer. | Lyng H et al |
| 12378529 | 2002 | Frequent amplification of 8q24, 11q, 17q, and 20q-specific genes in pancreatic cancer. | Mahlamäki EH et al |
| 16285859 | 2005 | T-box genes in vertebrate development. | Naiche LA et al |
| 21779450 | 2010 | The Highly Homologous T-Box Transcription Factors, TBX2 and TBX3, Have Distinct Roles in the Oncogenic Process. | Peres J et al |
| 17283120 | 2007 | Tbx3 is a downstream target of the Wnt/beta-catenin pathway and a critical mediator of beta-catenin survival functions in liver cancer. | Renard CA et al |
| 18829543 | 2008 | Tbx3 represses E-cadherin expression and enhances melanoma invasiveness. | Rodriguez M et al |
| 14699590 | 2004 | T-box genes in early embryogenesis. | Showell C et al |
| 16367923 | 2006 | Chemical genomic screening for methylation-silenced genes in gastric cancer cell lines using 5-aza-2'-deoxycytidine treatment and oligonucleotide microarray. | Yamashita S et al |
Other Information
Locus ID:
NCBI: 6926
MIM: 601621
HGNC: 11602
Ensembl: ENSG00000135111
Variants:
dbSNP: 6926
ClinVar: 6926
TCGA: ENSG00000135111
COSMIC: TBX3
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000135111 | ENST00000257566 | O15119 |
| ENSG00000135111 | ENST00000257566 | A0A024RBL6 |
| ENSG00000135111 | ENST00000349155 | O15119 |
| ENSG00000135111 | ENST00000349155 | A0A024RBQ4 |
Expression (GTEx)
Pathways
| Pathway | Source | External ID |
|---|---|---|
| Signaling pathways regulating pluripotency of stem cells | KEGG | hsa04550 |
| Signaling pathways regulating pluripotency of stem cells | KEGG | ko04550 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38909034 | 2024 | Overexpression of TBX3 suppresses tumorigenesis in experimental and human cholangiocarcinoma. | 0 |
| 38909034 | 2024 | Overexpression of TBX3 suppresses tumorigenesis in experimental and human cholangiocarcinoma. | 0 |
| 35856500 | 2023 | TBX3 stimulates proliferation and stem cell self-renewal in bladder carcinoma. | 1 |
| 36622795 | 2023 | TBX3 Promotes Cervical Cancer Proliferation and Migration via HPV E6 and E7 Signaling. | 2 |
| 36941669 | 2023 | TBX3 is dynamically expressed in pancreatic organogenesis and fine-tunes regeneration. | 0 |
| 37172754 | 2023 | Intracellular mechanics and TBX3 expression jointly dictate the spreading mode of melanoma cells in 3D environments. | 0 |
| 35856500 | 2023 | TBX3 stimulates proliferation and stem cell self-renewal in bladder carcinoma. | 1 |
| 36622795 | 2023 | TBX3 Promotes Cervical Cancer Proliferation and Migration via HPV E6 and E7 Signaling. | 2 |
| 36941669 | 2023 | TBX3 is dynamically expressed in pancreatic organogenesis and fine-tunes regeneration. | 0 |
| 37172754 | 2023 | Intracellular mechanics and TBX3 expression jointly dictate the spreading mode of melanoma cells in 3D environments. | 0 |
| 35846885 | 2022 | TBX3 regulates the transcription of VEGFA to promote osteoblasts proliferation and microvascular regeneration. | 2 |
| 35971932 | 2022 | TBX3 activating PVT1 accelerates proliferation, migration, and invasion by modulating the miR-30a/LOX axis in anaplastic thyroid carcinoma. | 1 |
| 36383654 | 2022 | Hierarchical deployment of Tbx3 dictates the identity of hypothalamic KNDy neurons to control puberty onset. | 1 |
| 36923312 | 2022 | Suppressed Cellular Senescence Mediated by T-box3 in Aged Gastric Epithelial Cells may Contribute to Aging-related Carcinogenesis. | 2 |
| 35846885 | 2022 | TBX3 regulates the transcription of VEGFA to promote osteoblasts proliferation and microvascular regeneration. | 2 |
Citation
Reyna Deeya Ballim ; Sharon Prince
TBX3 (T-box 3)
Atlas Genet Cytogenet Oncol Haematol. 2013-01-01
Online version: http://atlasgeneticsoncology.org/gene/42486/tbx3
