PA2G4 (proliferation-associated 2G4, 38kDa)
2011-09-01 Anne Hamburger , Arundhati Ghosh , Smita Awasthi AffiliationUniversity of Maryland School of Medicine, Department of Pathology, University of Maryland Greenebaum Cancer Center, Baltimore, USA
Identity
HGNC
LOCATION
12q13.2
LOCUSID
ALIAS
EBP1,HG4-1,p38-2G4
FUSION GENES
DNA/RNA
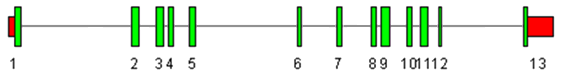
The alignment of PA2G4 mRNA to its genomic sequence.
Description
The PA2G4 gene contains 13 exons. The sizes of the exons 1-13 are 88, 128, 105, 69, 92, 63, 78, 78, 133, 94, 127, 53 and 65 bp (to the stop codon). Exon 1 contains the translation initiation ATG. Exon 13 contains the stop codon.
Transcription
The human PA2G4 promoter contains several putative transcription factor binding sites. The major transcript length is 2643 nt. Two proteins are translated due to alternative splicing (Liu et al., 2006). An alternatively spliced version missing 29 NT between the first and third ATGs has been observed.
The PA2G4 promoter contains two tandem DNA elements that bind E2F1. E2F1 increases endogenous EBP1 mRNA levels in cancer cells, but decreases EBP1 mRNA abundance in non transformed cells (Judah et al., 2010).
The PA2G4 promoter contains two tandem DNA elements that bind E2F1. E2F1 increases endogenous EBP1 mRNA levels in cancer cells, but decreases EBP1 mRNA abundance in non transformed cells (Judah et al., 2010).
Pseudogene
Six pseudogenes, located on chromosomes 3, 6, 9, 18, 20 and X, have been identified.
Proteins

The linear schematic of EBP1. Functional domains, including Nucleolar Localization Signal (NuLS), σ70 RNA binding region (σ70), amphipathic helical domain (AHD), LXXLL nuclear receptor binding motif (LX) and demonstrated in vivo phosphorylation sites (*).
Description
p38-2G4 was initially isolated as a DNA binding protein from mouse Ehrlich ascites cells (Radomski and Jost, 1995). The MW of this protein is predicted to be 38058 D, consisting of 340 amino acids. The human orthologue EBP1 was later identified as an ErbB3 binding protein of the same MW as the mouse protein (Yoo et al., 2000). This form migrates at approximately 42 kD in SDS-PAGE gels. Later, a larger 394 amino acid form (predicted MW 43787 D, migrating at 48 kD) was observed in mammalian cells (Xia et al., 2001). The two forms have been demonstrated to be the result of alternative splicing (Liu et al., 2006) or usage of alternative translation initiation sites (Xia et al., 2001). Amino acids 1-48 are required for nucleolar localization and the C terminal domain (aa 364-394) is required for interactions with nucleic acids (Moonie et al., 2007) and protein (Zhang et al., 2002).
EBP1 is post translationally modified at several phosphorylation sites (Ser 360 (Ahn et al., 2006), Ser 363 (Akinmade et al., 2007) and Thr 261 (Akinmade et al., 2008)) in vivo. The protein stability of the short form is regulated by ubiquitination (Liu et al., 2009). The short form is also sumoylated by the TLF/FUS E3 ligase and this sumoylation is required for the anti-proliferative effects of EBP1 (Oh et al., 2010).
The crystal structure of both murine (Monie et al., 2007) and human (Kowalinski et al., 2007) EBP1 has been solved. There is a core domain that is homologous to methionine aminopeptidases, although no enzymatic activity has been reported. The C terminal domain containing a Lys-rich nuclear hormone receptor binding motif (LKALL) was reported to mediate RNA binding (Monie et al., 2007).
EBP1 is post translationally modified at several phosphorylation sites (Ser 360 (Ahn et al., 2006), Ser 363 (Akinmade et al., 2007) and Thr 261 (Akinmade et al., 2008)) in vivo. The protein stability of the short form is regulated by ubiquitination (Liu et al., 2009). The short form is also sumoylated by the TLF/FUS E3 ligase and this sumoylation is required for the anti-proliferative effects of EBP1 (Oh et al., 2010).
The crystal structure of both murine (Monie et al., 2007) and human (Kowalinski et al., 2007) EBP1 has been solved. There is a core domain that is homologous to methionine aminopeptidases, although no enzymatic activity has been reported. The C terminal domain containing a Lys-rich nuclear hormone receptor binding motif (LKALL) was reported to mediate RNA binding (Monie et al., 2007).
Expression
EBP1 has been found to be ubiquitously expressed with high expression levels in skeletal muscle (Yoo et al., 2000).
Localisation
Under logarithmically growing conditions in cell culture, EBP1 localizes to the nucleolus and the cytoplasm (Xia et al., 2001; Squatrito et al., 2004). Upon stimulation with the ErbB3 ligand heregulin, the short form of EBP1 is recruited to the nucleus in AU565 breast cancer cells (Yoo et al., 2000). Sumoylation is required for nuclear translocation (Oh et al., 2010). In primary normal epithelial cells, EBP1 is confined to the cytoplasm (Zhang et al., 2008b).
Function
EBP1 was initially isolated as a cell cycle-regulated DNA binding protein (Radomski and Jost, 1995) and has been shown to induce cell cycle arrest in the G2/M phase of the cell cycle (Zhang et al., 2005). EBP1 acts as a corepressor for several proliferation-associated genes including Cyclin D1, E2F1 (Zhang and Hamburger, 2004) and the androgen receptor (Zhang et al., 2005). EBP1 inhibits transcription of these genes by recruiting HDAC2 via Sin3A to the E2F1 and AR-regulated promoters (Zhang et al., 2005). EBP1 interacts with RB1 and the interaction is enhanced upon EBP1 dephosphorylation (Xia et al., 2001).
EBP1 was isolated as an ErbB3 binding protein using a yeast-two hybrid screen (Yoo et al., 2000). The interactions of EBP1 with ErbB3 is disrupted by the ErbB3 ligand heregulin, leading to EBP1 nuclear translocation. This leads to the eventual inhibition of heregulin-stimulated proliferation, presumably due to the repression of proliferation associated genes (Zhang et al., 2008a).
EBP1 also binds RNA and associates with 28S, 18S and 5.8S mature rRNAs, several rRNA precursors and probably U3 small nucleolar RNA. It has been implicated in the regulation of intermediate and late steps of rRNA processing (Squatrito et al., 2004; Squatrito et al., 2006). EBP1 also mediates cap-independent translation of specific viral IRES (internal ribosome entry site) (Pilipenko et al., 2000). EBP1 regulates translation of AR mRNA (Zhou et al., 2010).
EBP1 has also been implicated in protein stability via its interaction with the proteasome. Overexpression of EBP1 results in decreased stability of ErbB2 protein in breast cancer cells via a proteasome-mediated pathway (Lu et al., 2011). The long form of EBP1 binds to the p53 E3 ligase HDM2, enhancing HDM2-p53 interactions and promoting p53 degradation (Kim et al., 2011).
The long (p48) and short (p42) forms of EBP1 have opposing biological effects, with the longer form inducing cell survival and the shorter form inhibiting cell growth (Liu et al., 2006). The long form binds HDM2, promoting degradation of p53 (Kim et al., 2010).
EBP1 was isolated as an ErbB3 binding protein using a yeast-two hybrid screen (Yoo et al., 2000). The interactions of EBP1 with ErbB3 is disrupted by the ErbB3 ligand heregulin, leading to EBP1 nuclear translocation. This leads to the eventual inhibition of heregulin-stimulated proliferation, presumably due to the repression of proliferation associated genes (Zhang et al., 2008a).
EBP1 also binds RNA and associates with 28S, 18S and 5.8S mature rRNAs, several rRNA precursors and probably U3 small nucleolar RNA. It has been implicated in the regulation of intermediate and late steps of rRNA processing (Squatrito et al., 2004; Squatrito et al., 2006). EBP1 also mediates cap-independent translation of specific viral IRES (internal ribosome entry site) (Pilipenko et al., 2000). EBP1 regulates translation of AR mRNA (Zhou et al., 2010).
EBP1 has also been implicated in protein stability via its interaction with the proteasome. Overexpression of EBP1 results in decreased stability of ErbB2 protein in breast cancer cells via a proteasome-mediated pathway (Lu et al., 2011). The long form of EBP1 binds to the p53 E3 ligase HDM2, enhancing HDM2-p53 interactions and promoting p53 degradation (Kim et al., 2011).
The long (p48) and short (p42) forms of EBP1 have opposing biological effects, with the longer form inducing cell survival and the shorter form inhibiting cell growth (Liu et al., 2006). The long form binds HDM2, promoting degradation of p53 (Kim et al., 2010).
Homology
Similar (30% identity) to the 42 kDA DNA binding protein SF00553 in S. pombe yeast (Yamada et al., 1994) and StEBP1 in potato (Horvath et al., 2006). The orthologue in potatoes (StEBP1) has 69% sequence similarity to human EBP1 and can inhibit growth of human breast cancer cell lines and E2F1 expression in these cells.
Mutations
Germinal
No mutations in the PA2G4 gene have been reported.
Somatic
None reported.
Implicated in
Entity name
Prostate cancer
Prognosis
Decreased expression of EBP1 is associated with higher tumor grade and metastasis in prostate cancer (Zhang et al., 2008b). However, another study indicated EBP1 expression increased with disease progression (Gannon et al., 2008).
Entity name
Breast cancer
Prognosis
Deletion of EBP1 results in tamoxifen resistance in breast cancer (Zhang et al., 2008a). However, patients with a high level of EBP1 protein have a poor clinical outcome (Ou et al., 2006).
Entity name
Glioblastoma
Prognosis
Glioblastoma patients expressing a high level of p48 EBP1 have a worse prognosis than those expressing lower levels of the protein (Kim et al., 2010; Kwon and Ahn, 2011).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16642037 | 2006 | Nuclear Akt associates with PKC-phosphorylated Ebp1, preventing DNA fragmentation by inhibition of caspase-activated DNase. | Ahn JY et al |
| 18283314 | 2008 | Phosphorylation of the ErbB3 binding protein Ebp1 by p21-activated kinase 1 in breast cancer cells. | Akinmade D et al |
| 19025630 | 2008 | Ebp1 expression in benign and malignant prostate. | Gannon PO et al |
| 17024182 | 2006 | EBP1 regulates organ size through cell growth and proliferation in plants. | Horváth BM et al |
| 21085677 | 2010 | EBP1 is a novel E2F target gene regulated by transforming growth factor-β. | Judah D et al |
| 21098709 | 2010 | Negative regulation of p53 by the long isoform of ErbB3 binding protein Ebp1 in brain tumors. | Kim CK et al |
| 17765895 | 2007 | The crystal structure of Ebp1 reveals a methionine aminopeptidase fold as binding platform for multiple interactions. | Kowalinski E et al |
| 21145366 | 2011 | p48 Ebp1 acts as a downstream mediator of Trk signaling in neurons, contributing neuronal differentiation. | Kwon IS et al |
| 16832058 | 2006 | Ebp1 isoforms distinctively regulate cell survival and differentiation. | Liu Z et al |
| 19037095 | 2009 | Human BRE1 is an E3 ubiquitin ligase for Ebp1 tumor suppressor. | Liu Z et al |
| 20379846 | 2011 | The ErbB3 binding protein EBP1 regulates ErbB2 protein levels and tamoxifen sensitivity in breast cancer cells. | Lu Y et al |
| 17690690 | 2007 | Structural insights into the transcriptional and translational roles of Ebp1. | Monie TP et al |
| 19946338 | 2010 | Ebp1 sumoylation, regulated by TLS/FUS E3 ligase, is required for its anti-proliferative activity. | Oh SM et al |
| 16944931 | 2006 | Quantitative profiling of drug-associated proteomic alterations by combined 2-nitrobenzenesulfenyl chloride (NBS) isotope labeling and 2DE/MS identification. | Ou K et al |
| 10950867 | 2000 | A cell cycle-dependent protein serves as a template-specific translation initiation factor. | Pilipenko EV et al |
| 7556453 | 1995 | Molecular cloning of a murine cDNA encoding a novel protein, p38-2G4, which varies with the cell cycle. | Radomski N et al |
| 16631606 | 2006 | Ebp1 is a dsRNA-binding protein associated with ribosomes that modulates eIF2alpha phosphorylation. | Squatrito M et al |
| 11268000 | 2001 | Ebp1, an ErbB-3 binding protein, interacts with Rb and affects Rb transcriptional regulation. | Xia X et al |
| 7985416 | 1994 | A fission yeast gene encoding a protein that preferentially associates with curved DNA. | Yamada H et al |
| 10682683 | 2000 | Interaction of the PA2G4 (EBP1) protein with ErbB-3 and regulation of this binding by heregulin. | Yoo JY et al |
| 18355957 | 2008 | Inhibition of heregulin mediated MCF-7 breast cancer cell growth by the ErbB3 binding protein EBP1. | Zhang Y et al |
| 15073182 | 2004 | Heregulin regulates the ability of the ErbB3-binding protein Ebp1 to bind E2F promoter elements and repress E2F-mediated transcription. | Zhang Y et al |
| 18852121 | 2008 | EBP1, an ErbB3-binding protein, is decreased in prostate cancer and implicated in hormone resistance. | Zhang Y et al |
| 20159994 | 2010 | Post-transcriptional regulation of androgen receptor mRNA by an ErbB3 binding protein 1 in prostate cancer. | Zhou H et al |
Other Information
Locus ID:
NCBI: 5036
MIM: 602145
HGNC: 8550
Ensembl: ENSG00000170515
Variants:
dbSNP: 5036
ClinVar: 5036
TCGA: ENSG00000170515
COSMIC: PA2G4
RNA/Proteins
Expression (GTEx)
Pathways
| Pathway | Source | External ID |
|---|---|---|
| Immune System | REACTOME | R-HSA-168256 |
| Innate Immune System | REACTOME | R-HSA-168249 |
| Neutrophil degranulation | REACTOME | R-HSA-6798695 |
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38834613 | 2024 | Orthogonal proteogenomic analysis identifies the druggable PA2G4-MYC axis in 3q26 AML. | 0 |
| 38834613 | 2024 | Orthogonal proteogenomic analysis identifies the druggable PA2G4-MYC axis in 3q26 AML. | 0 |
| 36423778 | 2023 | Loci on chromosome 12q13.2 encompassing ERBB3, PA2G4 and RAB5B are associated with polycystic ovary syndrome. | 2 |
| 36606322 | 2023 | LINC00173 facilitates tumor progression by stimulating RAB1B-mediated PA2G4 and SDF4 secretion in nasopharyngeal carcinoma. | 3 |
| 36423778 | 2023 | Loci on chromosome 12q13.2 encompassing ERBB3, PA2G4 and RAB5B are associated with polycystic ovary syndrome. | 2 |
| 36606322 | 2023 | LINC00173 facilitates tumor progression by stimulating RAB1B-mediated PA2G4 and SDF4 secretion in nasopharyngeal carcinoma. | 3 |
| 33150374 | 2021 | Progression signature underlies clonal evolution and dissemination of multiple myeloma. | 20 |
| 33220287 | 2021 | Pseudogene PA2G4P4 promotes oncogene PA2G4 expression and nuclear translocation to affect glioblastoma cell viability and apoptosis. | 3 |
| 33529238 | 2021 | HER2-intronic miR-4728-5p facilitates HER2 expression and accelerates cell proliferation and migration by targeting EBP1 in breast cancer. | 3 |
| 33605574 | 2021 | Increased PA2G4 Expression Is an Unfavorable Factor in Nasopharyngeal Carcinoma. | 5 |
| 33634940 | 2021 | Ebp1 p48 promotes oncogenic properties in hepatocellular carcinoma through p38 MAPK/HIF1α activation and p53 downregulation. | 5 |
| 33691908 | 2021 | EBP1 regulates Suv39H1 stability via the ubiquitin-proteasome system in neural development. | 4 |
| 33150374 | 2021 | Progression signature underlies clonal evolution and dissemination of multiple myeloma. | 20 |
| 33220287 | 2021 | Pseudogene PA2G4P4 promotes oncogene PA2G4 expression and nuclear translocation to affect glioblastoma cell viability and apoptosis. | 3 |
| 33529238 | 2021 | HER2-intronic miR-4728-5p facilitates HER2 expression and accelerates cell proliferation and migration by targeting EBP1 in breast cancer. | 3 |
Citation
Anne Hamburger ; Arundhati Ghosh ; Smita Awasthi
PA2G4 (proliferation-associated 2G4, 38kDa)
Atlas Genet Cytogenet Oncol Haematol. 2011-09-01
Online version: http://atlasgeneticsoncology.org/gene/41628/pa2g4
