CCND1 (B-cell leukemia/lymphoma 1)
2015-04-01 Shreya Sarkar , Chinmay Kumar Panda AffiliationDepartment of Oncogene Regulation, Chittaranjan National Cancer Institute, Kolkata, West Bengal, India; [email protected]
Identity


Abstract
Review on CCND1, with data on DNA, on the protein encoded, and where the gene is implicated.
DNA/RNA
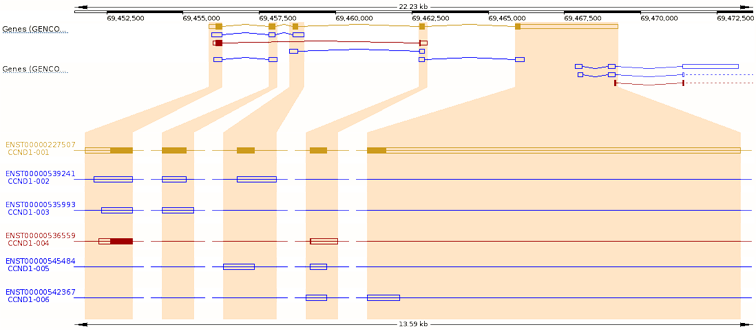
Description
Transcription
Pseudogene
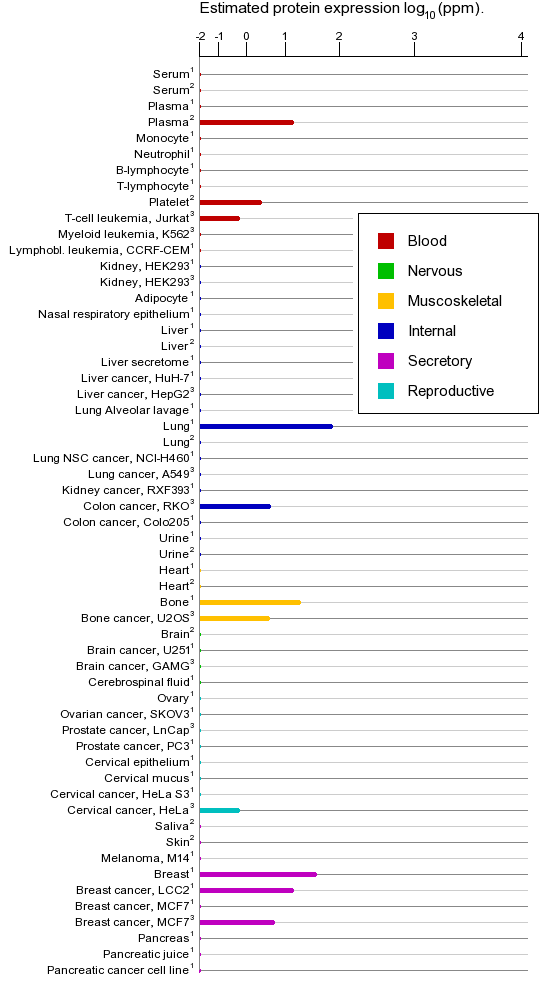
Proteins
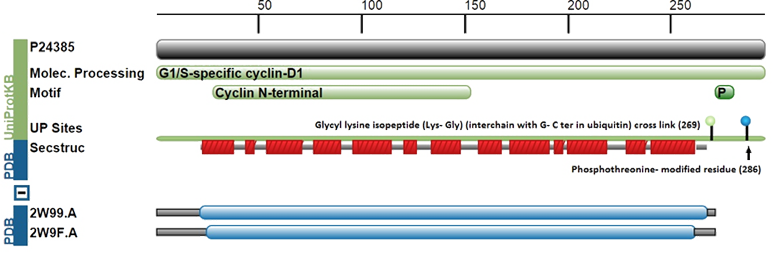
Description

.
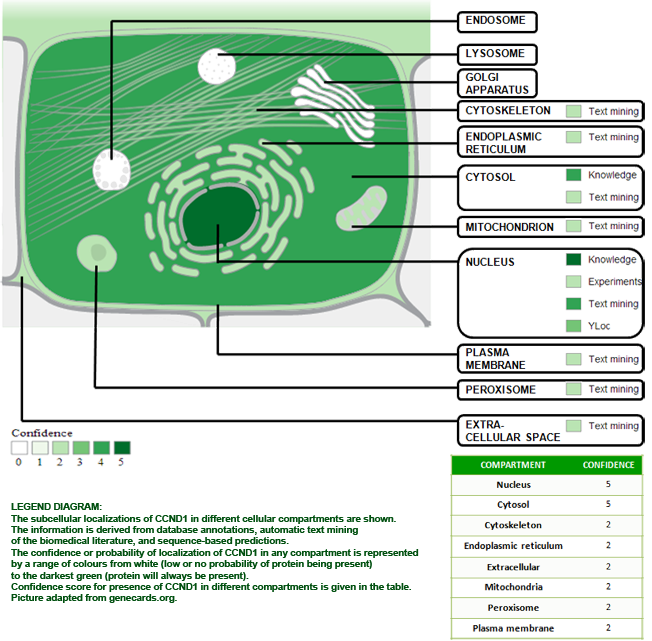
Localisation
NOTE: Accumulation of CCND1- CDK4 complexes occur in the nuclear membrane, which are then transported to the nucleus through interactions with KIP-CIP family member proteins (By similarity, a LaBaer et. al.,1997).
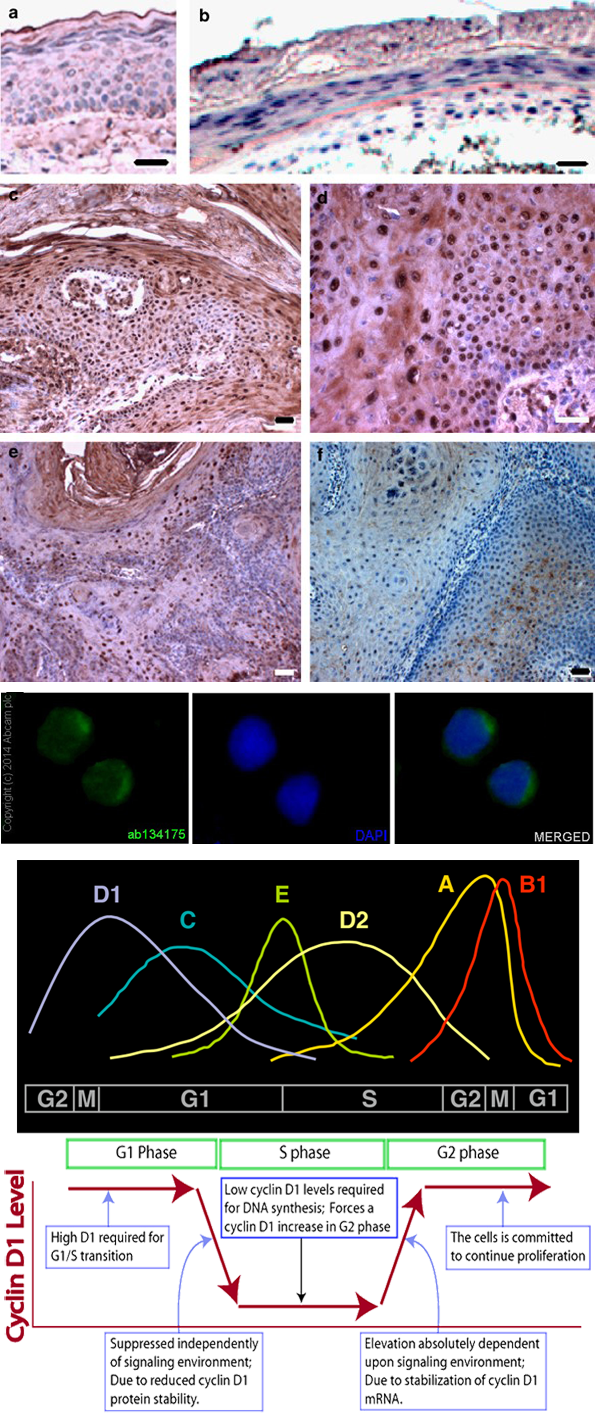
Function
CCND1 has a kinase-independent function of sequestering CDK inhibitors such as p27 Kip1 and p21Cip1and promoting efficient activation of Cyclin E/CDK2-containing complexes (Polyak et al., 1994; Sherr and Roberts, 1999).
CCND1 phosphorylates Smad3 and inhibits its transcriptional activity and antiproliferative function (Matsuura et. al., 2004).
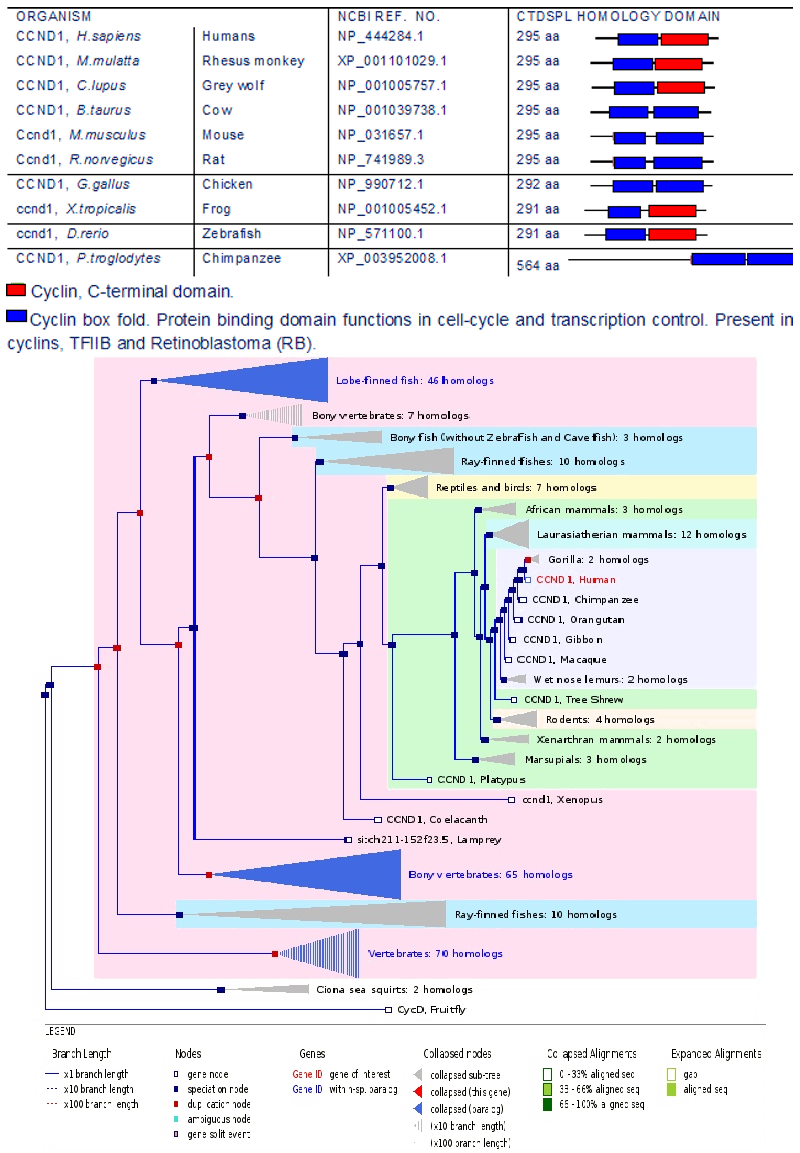
Homology

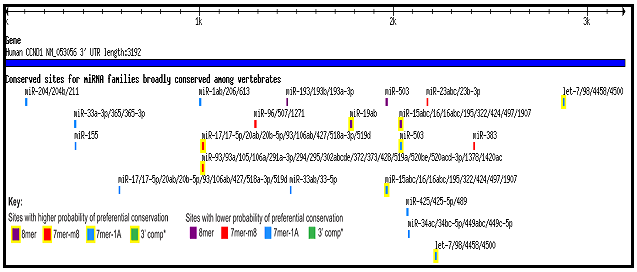
Mutations

Epigenetics
miR365
in Gastric cancer cell line BGC-823
Binds to 3 UTR of CCND1 in gastric cancer. miR-365 markedly decreased the expression (mRNA and protein) of CCND1. Conversely, miR365 knockdown repressed cell growth, which can be overcome by CCND1 over-expression. Similar inverse co-relation was obtained between miR-365 and CCND1 expression in patient samples. (Long-Guo et. al., 2013).
in Vascular smooth muscle cell (VSMC)
miR-365 suppresses CCND1 significantly in mRNA and protein levels in primary rat VSMC. CCND1 is a direct target of miR-365 in vascular smooth muscle cells, as shown by significant inhibition of the luciferase activity of wild type CCND1 3 UTR, but not the mutant cyclinD1 3 UTR with the mutant biding site of miR-365 (Zhang et. al., 2014). CCND1 is a potential target of mir-365 through direct binding. (Kim et. al., 2014).
in Colon cancer
miRNA directly binds to the 3UTR of CCND1, proved by luciferase reporter assay. Transfection of miR365 significantly decreased CCND1 expression in HT29 and LoVo cells. Pearsons co-relation between miR-365 levels and CCND1 expression by qRT-PCR and western blot showed that they were inversely correlated (Nie et. al., 2012).
miR-338-3p in Hepatocyte cell line LO2
miR-338-3p binds at two regions in the 3 UTR of CCND1( mainly at the site spanning nucleotides 2397-2403). Overexpression of miR-338-3p downregulates endogenous CyclinD1 protein, while inhibition upregulates CyclinD1 protein, without any change in CCND1 mRNA levels. miR-338-3p post-transcriptionally regulates CCND1 (Fu et. al., 2012).
miR-19a in Human umbilical vein endothelial cells (HUVECs)
miR-19a binding site (nucleotides 1,778-1,785 in human CCND1) identified by sequence alignment, which is highly conserved among different species. Binding of miR-19a to 3 UTR of CCND1 verified by luciferase assay. CCND1 protein expression markedly reduced upon over-expression of miR-19a, although no change in RNA expression. miR-19a post-transcriptionally regulates CCND1 expression (Qin et. al., 2010).
miR-490-3p in A549 Lung cancer cell line
miR-490-3p binds to 3 UTR of CCND1. Over-expression decreased the expression of CCND1, both at the RNA and protein levels (Gu et. al., 2014).
miR-302 in Endometrial cell line Ishikawa
Directly targets CCND1 and significantly inhibited protein expression (Yan et. al., 2014).
miR-449-a in Gastric cancer cell line SGC7901
miR-449a inhibited SGC7901 cells proliferation and enhanced cisplatin chemosensitivity by downregulating expression of CCND1, respectively, via directly targeting the 3-untranslated regions of CCND1 mRNA (Hu et. al., 2014).
miR-16 in Bladder cancer cell line TCHu-1
Binding of miR16 to 3 UTR of CCND1 and its reduced expression was validated by luciferase assay, while the reverse result was obtained by mutation of the conserved miR-16 binding motif. Overexpression of miR-16 in TCHu-1 cells led to reduced CCND1 protein expression, whereas its inhibition led to an increased expression of CCND1 (Jiang et. al., 2013).
miR-9 in Gastric cancer
Databases indicated potential binding site of miR-9 with high complementarity at CCND1 39-UTR (bases 2974-2995), which was validated by luciferase reporter assay. Significant inverse correlation between miR-9 expression and CCND1 transcript levels in gastric cancer tissues and cell lines. Overexpression of miR9 in gastric cancer cell lines SGC-7901 and AGS resulted in reduced RNA and protein expression of CCND1, whereas knockdown of miR-9 produced the opposite result, proving that miR-9 considerably inhibited the expression of CCND1 through post-transcriptional repression. Results validated by in-vitro experiments (Zheng et. al., 2013).
miR-195 in Glioma
Analysis using publicly available algorithms (TargetScan, Pictar, miRANDA) indicates that CCND1 is a predicted target of miR-195, which was validated by overexpression of miR- 195, which reduced, but inhibition of miR-195 increased, the luciferase activity of CCND1-39UTR in a consistent and dose-dependent manner. Upregulation of miR-195 decreased, but inhibition of miR-195 increased, the expression levels of CCND1 in LN18 and T98G glioma cells. The findings were also validated in a model system in mice (Hui et. al., 2013).
miR-155 in Human extravillous trophoblast derived HTR-8/SVneo cells
Bioinformatics analysis showed that, at the 3 untranslated region (UTR) of CCND1, six bases are complementary to the seed region of miR-155. Luciferase assays and CCND1 3UTR transfection assays validated that CCND1 3UTR was the target of miR-155 in HTR-8/SVneo cells. Overexpression of miR-155 in HTR-8/SVneo cells reduced the level of CCND1 protein (Dai et. al., 2012).
miR-143 in Mesenchymal stem cells from the bone marrow of male Fischer 344 rats
Ectopic expression of miR-143 also increased CCND1 in the native MSC as compared with scramble transfected cells .On the contrary, pre-treatment of AAMSC with miR-143 specific antagomir significantly abolished CCND1 expression (Lai et. al., 2012).
miR-21
in Mouse liver regeneration
Cyclin D expression and G1 phase transition of hepatocytes after 2/3 PH depend on induced miR-21 expression. Knockdown of miR-21 impaired progression of hepatocytes into S phase of the cell cycle, mainly through a decrease in levels of cyclinD1 protein, but not Ccnd1 mRNA, whereas increased miR-21 expression facilitated CCND1 translation in the early phase of liver regeneration (Ng et. al., 2012).
in Renal cancer
miR-21 controlled the expression of CCND1 through NF?B-dependent transcription and mediated renal cancer cell proliferation by CCND1 (Bera et. al., 2013).
miR-520-b in Hepatoma cell lines
miR520-b directly targets the 3 UTR of CCND1; proved by dual luciferase reporter system. Down-regulation of protein levels of CCND1 occurred on over-expression of miR520-b in HepG2 and H7402 cells, while the over-expression occurred on inhibition in miR520-b. Tumors in mice over-expressing miR520-b also showed lower CCND1 expression (Zhang et. al., 2012).
miR-193b in Melanoma
TargetScan showed that miR193b binds to the 3UTR of CCND1, which was proved by luciferase reporter assay. miR-193b over-expression led to nearly 50% reduction in CCND1 mRNA and protein levels in Malme-3M cells than in control (Chen et. al., 2010).
miR-17/20 in Breast cancer
Levels of the miR-17-5p/miR-20a miRNA cluster were inversely correlated to CCND1 abundance in human breast tumors and cell lines. miR 17/20 negatively regulates the expression of CCND1 by binding to a conserved 3UTR region (nucleotides 2,109-2,117) of the gene (Yu et. al., 2008).
miR-20 and miR106-a in Spermatogonial stem cells (SCC)
They promote renewal at the post-transcriptional level via targeting CCND1. Knockdown of CCND1 results in renewal of SCCs (He et. al., 2013).
miR-503 in Endometrioid endometrial cancer (EEC)
Binds to 5 UTR of CCND1 and its expression is inversely co-related with CCND1 in EEC tissues and cell lines (Xu et. al., 2013).
miR-449b in SW116 colon cancer stem cell
Transfecting pre-miR-449b and inhibiting miR-449b altered protein expression levels of CCND1 (Fang, 2013).
miR-15a and miR16-1 in Osteosarcoma
They bind to 3-UTR of CCND1 and suppress transcription of CCND1 (Cai et. al., 2012).
miR-138 in Nasopharyngeal carcinoma
CCND1 is a novel direct target of miR138. mRNA levels of CCND1 were inversely correlated with miR-138 expression (Liu et. al., 2012).
miR-34a in A549 cell line
Ectopic expression of miR-34a reduces both mRNA and protein levels of CCND1 by targeting the 3-untranslated mRNA region of CCND1 (Sun et. al., 2008).
miR-29a in Breast cancer cell lines
Over-expression of miR29a down-regulation of CCND1 expression in MDA-MB-453 cells, whereas in MCF-10A cells with Mir-29a knockdown, CCND1 was up-regulated (Wu et. al., 2013).
miR-7 in Colorectal cancer cell lines
Over-expression of miR-7 significantly decreased CCND1 expression (Xu et. al., 2014).
miR-545 in Lung cancer
miR-545 caused cell cycle arrest at the G0/G1 phase and induced cell apoptosis in lung cancer cells by targeting CCND1. The effects of CCND1 down-regulated by miR-545 were similar to those caused by siRNAs of CCND1 and over-expression of CCND1 could abolish the miR-545-induced inhibition of cell proliferation (Du et. al., 2014).
miR-125b in Melanoma
Cells over-expressing miR-125b exhibited reduced expression of CCND1 (Nyholm et. al., 2014).
miR-147 in Colon and lung cancer cells.
Transfection of miR147 led to down-regulation of CCND1 (Lee et. al., 2014).
Implicated in
Over-expression of Pin X1 in T24 cells leads to greater than 2 fold increase in mRNA expression of CCND1 than in control cell, with similar results obtained by Western blotting. A significant correlation between the immune-histochemical expression of PinX1 and CCND1 was also observed in the UCB tissues (Liu et. al., 2013).
Ursane triterpenoid isopropyl 3?-hydroxyurs-12-en-28-oat (UA17) (Natural compound) : Protein level of CCND1 was down-regulated in a dose-dependent manner when treated with UA17or Cisplatin in NTUB1 cells. Enhanced decrease of level of CCND1 when treated with a combination of Cisplatin (20 ?M) + UA17 (20 ?M) (Lin et. al., 2014)
Metformin : Treatment with metformin leads to reduction in expression of CCND1 in a dose-dependent manner. Metformin treatment also markedly reduced the expression of CCND1 in Human Bladder Tumor Xenografts in Nude Mice compared to control (Zhang et. al., 2013).
Enhanced expression of Vav1 led to the elevation of CCND1 and the progression of cell cycle (Du et. al., 2014).
Tea polyphenols (Natural compound): Tea polyphenols did not significantly alter the expression of CCND1 in breast cancer cell lines (Chen et. al., 2014).
Acylglycerol kinase (AGK) over-expression led to concurrent increase in levels of CCND1 (Wang et. al., 2014).
Activation of Notch-1 signaling up-regulated expression of CCND1 through NF-kB (Li et. al., 2014).
Panepoxydone (Natural compound): CCND1 was down-regulated by dose-dependent treatment of Panepoxydone (Arora et. al., 2014).
Gallotannin (Natural compound): Nanostring and qPCR data showed that CCND1 was exclusively downregulated on treatment with gallotannin in triple negative breast cancer (Zhao et. al., 2014).
Progesterone induced the assembly of a transcriptional complex among AP-1, Stat3, PR, and ErbB-2 at the CCND1 promoter, which functions as an enhanceosome to drive breast cancer growth (Flaqué et. al., 2013).
Obatoclax analog SC-2001 : SC-2001 down-regulated CCND1 in TNBC cell lines in a dose- dependent manner (Liu et. al., 2014). Euginol (Natural compound): Treatment of euginol decreased CCND1 level 3 fold in MDA-MB-231 cells and 20 fold in MCF7 cells compared to control (Sharif et. al., 2013).
8u001ebromou001e7u001emethoxychrysin (BrMC) (Natural compound): BrMC caused a doseu001edependent reduction of CCND1 in HER2/neu over-expressing breast cancer cells (Cao et. al., 2014).
Fenofibrate : Fenofibrate decreased the expression of CCND1 in a time and dose dependent manner in Triple negative breast cancer cells (Li et. al., 2014).
Quercetin (Natural compound): CML KBM7 Cells demonstrated reduction in expression on CCND1 on treatment with quercetin ((Li et. al., 2014).
Quercetin (Natural compound): CML KBM7 Cells demonstrated reduction in expression on CCND1 on treatment with quercetin ((Li et. al., 2014).
High height and weight was associated with risk of CCND1 positive CRC in women. Increased hip circumference, high BMI, high WHR and high waist circumference was associated with CCND1 positive tumours in men (Brändstedt et. al., 2013).
CCND1 over-expression was significantly associated with both poor OS, DFS, relatively older patients (?60 years), T3,4 tumor invasion, N positive and distant metastasis (Li et. al., 2104).
Galectin-3 knockdown decreased the mRNA expression level of CCND1, whereas epirubicin significantly up-regulated their expression. Combined treatment effectively reduced the mRNA expression of CCND1 (Lee et. al., 2013).
HMGCR expression was significantly associated with expression of CCND1 (Bengtsson et. al., 2014).
CoCl2 : Treatment of COCl2 leads to dose-dependent decrease in expression of CCND1 and cell cycle arrest (Lopez-Sanchez et. al., 2014).
SW620-S and TGF-b1 : Fibroblasts induced by Colorectal cancer cells, treated with SW620-S and TGF-b-1 separately showed high expression of CCND1 (Rao et. al., 2014).
No significantly statistical differences between the two groups were observed in distribution of genotypes or alleles at CCND1 807 (Jang et. al., 2013).
Resveratrol (Res) (Natural compound) : Res reduced expression of CCND1 (Yang et. al., 2013). Knockdown of P115 led to reduction in expression of CCND1, whereas its over-expression led to up-regulation of CCND1 (Li et. al., 2013).
Caudatin 3-O-?-D-cymaropyranosyl-(1 ? 4)-?-D-oleandropyranosyl-(1 ? 4)-?-D-cymaropyranosyl-(1 ? 4)- ? -D-cymaropyranoside (CGII) (Drug): CGII induced down-regulation of expression of CCND1 in a dose-dependent manner in Gastric Cancer SGC-7901 Cells (Wang et. al., 2013)
Tetramethypyrazine (TMP) (Natural compound): Expression of CCND1 gradually decreased with increasing concentrations of TMP in Gastric cancer 7901 cells (Ji et. al., 2014).
Resveratrol (Res) (Natural compound) : Res reduced expression of CCND1 (Yang et. al., 2013). Knockdown of P115 led to reduction in expression of CCND1, whereas its over-expression led to up-regulation of CCND1 (Li et. al., 2013).
Caudatin 3-O-?-D-cymaropyranosyl-(1 ? 4)-?-D-oleandropyranosyl-(1 ? 4)-?-D-cymaropyranosyl-(1 ? 4)- ? -D-cymaropyranoside (CGII) (Drug): CGII induced down-regulation of expression of CCND1 in a dose-dependent manner in Gastric Cancer SGC-7901 Cells (Wang et. al., 2013)
Tetramethypyrazine (TMP) (Natural compound): Expression of CCND1 gradually decreased with increasing concentrations of TMP in Gastric cancer 7901 cells (Ji et. al., 2014).
Knockdown of TRIM24 led to decreased CCND1 expression (Liu et. al., 2014).
KIF14 knockdown suppresses tumor cell growth through decrease in levels of cyclins including CCND1 (Xu et. al., 2014).
Sorafenib and YC-1 : Treatment with the sorafenib and YC-1 combination led to a significant reduction in CCND1 (Kong et. al., 2014). 7. 3, 3u001eDiu001eOu001emethyl ellagic acidu001e4u001eOu001e?u001edu001exylopyranoside (JNE2). JNE2 induced down-regulation of expression of CCND1 in HepG2 cells (Zhang et. al., 2014).
SL1122-37: SL1122-37 induced down-regulation of expression of CCND1 in PLC/ PRF/5 HCC cells (Qin et. al., 2013).
Over-expression of Ubiquitin- conjugating enzyme E2C (UBE2C) increased expression of CCND1 in L-78 and SC-1680 cells, as well as in tumor transplants in nude mice (Tang et. al., 2014).
Met- F-AEA in combination with URB597 induced down-regulation of CCND1 and subsequent G0/ G1 cell cycle arrest (Ravi et. al., 2014).
Up-regulation of decorin led to significant decrease in expression of CCND1 (Liang et. al., 2013).
Polydatin: PD suppressed expression of CCND1 in A549 and NCIu001eH1975 lung cancer cell lines (Zhang et. al., 2014). Knockdown of JAM-A decreased protein levels of CCND1 (Zhang et. al., 2013).
Tea polyphenols (Natural compound): Epigallocatechin gallate, epicatechin gallate and theaflavin reduced the expression of CCND1 in benzo(a)pyrene-induced lung carcinogenesis in mice (Manna et al., 2009).
85% were weakly positive and 15%, moderately positive with labelled streptavidin biotin, whereas 75% were weakly positive and 25% moderately positive for CCND1 with EnVision. All 20 mantle cell lymphoma cases were strongly CCN D1 positive with catalyzed signal amplification. No evidence of CCND1 immunostaining was obtained in any of the small lymphocytic lymphoma and follicular centre cell lymphoma instances with any of the three methods used (Barranco et. al., 2003).
CCND1 showed exclusive nuclear staining and directly compared with the expression observed by immunoblot analysis with the same antibody, as well as with mRNA expression and with the occurrence of genomic rearrangements within the B CL-1 locus. 12/13 MCL showed over-expression by immunohistochemistry or immunoblot, with similar results for additional 13 MCLs, indicating its importance for routine diagnostic purposes (Boer et. al., 2014).
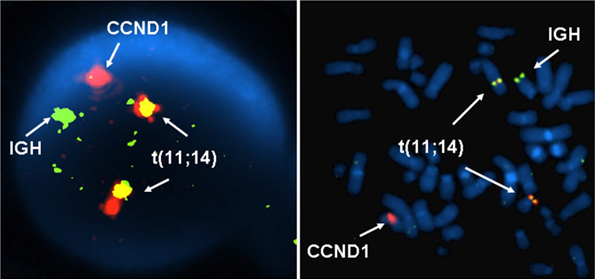
CCND1 mRNA could be detected in 23 of 24 mantle-cell lymphomas by reverse transcription polymerase chain reaction (RT-PCR) whereas only 9 of 24 demonstrated a t(11;14) by PCR (Aguilera et. al., 1998).
In 16 of 21 cases of MCL with overt disease, the ratio of CCND1 mRNA to ?2-microglobulin mRNA was increased, but all 21 cases showed increased ratios of CCND1 mRNA to CD19 mRNA (Howe et. al., 2004)
Indole-3-carbinol (I3C) : I3C induced G1 arrest by decreasing CCND1 expression (Chen et. al., 2013).
Over-expression of n-myc downstream regulated gene 2 (NDRG2) induced down-regulation of expression of CCND1 (Zhang et. al., 2014).
A negative co-relation existed between WWOX and CCND1 expression (Nowakowska et. al., 2014)
Clinico-pathological correlation showed that CCND1 over-expression was related to increase in tumor size, tumor differentiation and higher clinical stages and lymph node metastasis and adversely affected overall survival (Zhao et. al., 2014).
HPV-negative patients, heavy alcohol consumption was significantly associated with somatic copy-number alterations (SCNAs) in CCND1 (Urashima et. al., 2013).
The proportions of positive staining in well, moderately and poorly differentiated laryngeal SCC were 50, 66.7, 100%, respectively, for CCND1, and were statistically significant, with the expression being positively correlated with Ang-2 expression. Tumor grading and CCND1 were independent factors affecting laryngeal SCC patient survival by the Cox regression model of risk factors proportion analysis, which may possess clinical significance in evaluating the prognosis and guiding the clinical treatment of SCC (Liu et. al., 2013).
Knockdown of Nemo-like kinases (NLK) led to significant reduction in the levels of CCND1 (Dong et. al., 2013).
2,4-bis (p-hydroxyphenyl)-2-butenal : HPB 242 significantly decreased CCND1 expression in HN22 and HSC4 Oral squamous cell carcinoma cell lines (Chae et. al., 2014).
Down-regulation of miR-196a led to decrease in expression of CCND1 via Nuclear Factor Kappa-B-Inhibitor Alpha (Huang et. al., 2014).
Diallyl trisulfide (DATS) (Natural compound) : DATS reduced levels of CCND1 and DATS-induced apoptosis was correlated with down-regulation of CCND1 protein levels in Capan-2 cells (Ma et. al., 2014).
alpha-Mangostin (Natural compound) : alpha--Mangostin led to decrease in expression of CCND1 (Xu et. al., 2014).
Pristimerin (PM) : PM treatment produced decreased expression of CCND1 in MiaPaCa-2 and Panc-1 cells (Deeb et. al., 2014).
Univariate analyses showed that lymph node positivity, surgical margin positivity, non-localized tumor, age at prostatectomy and CCND1 in malignant epithelium were significantly associated with time to BF (Biochemical failure) (Rizzardi et. al., 2014).
Pifithrin (PFT) : Combination therapy with suboptimal doses of PFT-m and HT decreased expression of CCND1 (Sekihara et. al., 2013). Triptolide (Natural compound) : Triptolide induced significant decrease of expression of CCND1 through EZH2 (Tamgue et. al., 2014).
Salinomycin-: Salinomycin induced lowering of expression of CCND1 in Breast and prostate cancer cells (Lu et. al., 2014).
Human diploid fibroblast (HDFs): CCND1 gene was significantly up-regulated in irradiated (1 Gy) HDFs as compared to untreated control, while bothHDFs treated with Gelam honey and irradiated HDFs pre-treated with Gelam honey showed down- regulation of cyclin D1 gene as compared to irradiated HDFs. HDFs treated with Gelam honey during radiation and post-irradiation however showed significant up-regulation of cyclin D1 gene as compared to untreated control (Ahmed et. al., 2014).
Vascular smooth muscle cells: STS (sodium tanshinone IIA silate) decreased the expression of cell cycle-associated protein, CCND1 (Wu et. al., 2014)
. Vascular smooth muscle cells: PDGF-induced CCND1 mRNA and protein expression was inhibited by TGFb. PDGF-induced CCND1 expression requiring KLF5 was inhibited by TGFb via a Smad dependent mechanism, leading to G1 cell cycle arrest of VSMs (Garrido et. al., 2013).
Nuroectodermal stem cells: PGE2 (Prostaglandin E2) treatment significantly up-regulated CCND1 (Wong et. al., 2014).
Neurons: DYRK1A (dual specificity tyrosine-phosphorylation-regulated kinase 1A) reduced cellular CCND1 levels by phosphorylation on Thr286, which is known to induce proteasomal degradation (Soppa et. al., 2014).
Renal intestinal fibroblasts: Exposure of NRK-49F to resulted in reduced expression proliferation markers CCND1 in a dose and time dependent manner (Ponnusamy et. al., 2014).
Idiopathic pulmonary fibrosis (IPF): Cell cycle regulatory protein CCND1 was significantly enhanced in AEC (alveolar epithelial cell) within the remodelled fibrotic areas of IPF lungs but expression was negligible in myofibroblasts (Akram et. al., 2014).
Human Rheumatoid Arthritis Synovial Cells: The protein and mRNA levels of CCND1 decreased gradually with the increasing of thapsigargin concentration and treatment times (Wang et. al., 2014).
Rat liver fibrosis:
Decreased expression in CCND1 in the cerebellum of the hyperbilirubinemic Gunn rats led to significant increased cell cycle arrest in the late G0/G1 phase (Robert et. al., 2013).
Chicken fetal myoblasts (CFMs):
Following seventy percent partial hepatectomy (PH) in wild type (WT) mice IL-6 serum levels increased, resulting in increased CCND1 (Tachibana et. al., 2014).
CCND1 was more frequently up-regulated in mammary tumors from transgenic mice (expressing myristoylated-Akt1 (myr-Akt1) under the control of the MMTV-LTR promoter) compared to tumors from wild-type mice. Increased expression of CCND1 was incompletely dependent on Akt1 expression. Low expression of CCND1 and increased expression of Twist and Slug was observed in mammary tumors that had metastasized to secondary sites (Wu et. al., 2014).
Embelin-treated mice showed significant inhibition in tumor growth, which was associated with reduced expression of CCND1 (Huang et. al., 2014).
Nicotine significantly increased expression of CCND1 (He et. al., 2014).
In mice treated with hUCMSCs-LV-IL-21, Expression of cyclin-D1 was simultaneously low compared to control group, hUCMSCs group and hUCMSCs-LV-Vec group (Zhang et. al., 2014).
Rat:
Dairy Cow Mammary Epithelial Cells:
Treatment with leucine induced LeuRS, increasing CCND1 mRNA and protein expression (Wang et. al., 2014).
CCND1 accumulation due to differential effects of of PKC? and PKC? was likely contribute to the opposing tumor suppressive and tumor promoting activities in the intestinal epithelium (Pyfz et. al., 2014).
IGF-1R activation together with EGFR co-signaling decreased the percentage of cells in G1 and enhanced cell progression into S and G2 by increases in expression of CCND1 (Alagappan et. al., 2014).
CCND1 mRNA was significantly decreased by sodium ferulate in cells under serum stimulation (Zhang et. al., 2014).
Sophocarpine inhibited the proliferation of HSCs by a decrease in the expression of CCND1 (Qian et. al., 2014).
Rat Airway Smooth Muscle Cells:
Chicken:
Increased CCND1 expression during acceleration of cell cycle at G1/ S phase in CMF was due to CARP (cardiac ankyrin repeat protein) over-expression (Ma. et. al., 2014).
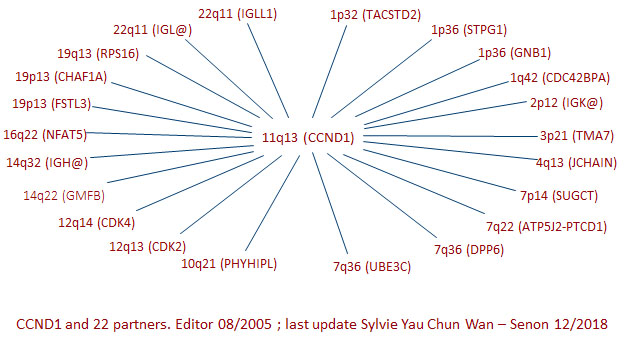
Breakpoints
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 9846986 | 1998 | Differential expression of cyclin D1 in mantle cell lymphoma and other non-Hodgkin's lymphomas. | Aguilera NS et al |
| 11743641 | 2002 | Chromosomal translocations involving 11q13 contribute to cyclin D1 overexpression in squamous cell carcinoma of the head and neck. | Akervall J et al |
| 9010112 | 1997 | Amplification of cyclin D1 in squamous cell carcinoma of the head and neck and the prognostic value of chromosomal abnormalities and cyclin D1 overexpression. | Akervall JA et al |
| 24551275 | 2014 | Alveolar epithelial cells in idiopathic pulmonary fibrosis display upregulation of TRAIL, DR4 and DR5 expression with simultaneous preferential over-expression of pro-apoptotic marker p53. | Akram KM et al |
| 24330704 | 2013 | Eugenol triggers apoptosis in breast cancer cells through E2F1/survivin down-regulation. | Al-Sharif I et al |
| 24904523 | 2014 | Insulin-Like Growth Factor Receptor Signaling is Necessary for Epidermal Growth Factor Mediated Proliferation of SVZ Neural Precursors in vitro Following Neonatal Hypoxia-Ischemia. | Alagappan D et al |
| 24896091 | 2014 | Panepoxydone targets NF-kB and FOXM1 to inhibit proliferation, induce apoptosis and reverse epithelial to mesenchymal transition in breast cancer. | Arora R et al |
| 12591969 | 2003 | Catalyzed signal amplification for cyclin D1 detection in mantle cell lymphoma. | Barranco C et al |
| 24708688 | 2014 | HMG-CoA reductase expression in primary colorectal cancer correlates with favourable clinicopathological characteristics and an improved clinical outcome. | Bengtsson E et al |
| 23981302 | 2013 | NFκB-mediated cyclin D1 expression by microRNA-21 influences renal cancer cell proliferation. | Bera A et al |
| 24885829 | 2014 | Associations of hormone replacement therapy and oral contraceptives with risk of colorectal cancer defined by clinicopathological factors, beta-catenin alterations, expression of cyclin D1, p53, and microsatellite-instability. | Brändstedt J et al |
| 16547504 | 2006 | Gain of 11q/cyclin D1 overexpression is an essential early step in skin cancer development and causes abnormal tissue organization and differentiation. | Burnworth B et al |
| 22922827 | 2012 | miR-15a and miR-16-1 downregulate CCND1 and induce apoptosis and cell cycle arrest in osteosarcoma. | Cai CK et al |
| 24765191 | 2014 | Inhibition of cell growth by BrMC through inactivation of Akt in HER-2/neu-overexpressing breast cancer cells. | Cao XZ et al |
| 24423061 | 2014 | Specificity protein 1 is a novel target of 2,4-bis (p-hydroxyphenyl)-2-butenal for the suppression of human oral squamous cell carcinoma cell growth. | Chae JI et al |
| 20304954 | 2010 | MicroRNA-193b represses cell proliferation and regulates cyclin D1 in melanoma. | Chen J et al |
| 24646833 | 2014 | Tea polyphenols induced apoptosis of breast cancer cells by suppressing the expression of Survivin. | Chen X et al |
| 24358165 | 2013 | Indole-3-carbinol inhibits nasopharyngeal carcinoma growth through cell cycle arrest in vivo and in vitro. | Chen Z et al |
| 24902855 | 2014 | PAR1-dependent COX-2/PGE2 production contributes to cell proliferation via EP2 receptors in primary human cardiomyocytes. | Chien PT et al |
| 24744797 | 2014 | Longer survival in patients with breast cancer with cyclin d1 over-expression after tumor recurrence: longer, but occupied with disease. | Chung J et al |
| 24345432 | 2013 | Progesterone receptor assembly of a transcriptional complex along with activator protein 1, signal transducer and activator of transcription 3 and ErbB-2 governs breast cancer growth and predicts response to endocrine therapy. | Díaz Flaqué MC et al |
| 22858023 | 2012 | MicroRNA-155 inhibits proliferation and migration of human extravillous trophoblast derived HTR-8/SVneo cells via down-regulating cyclin D1. | Dai Y et al |
| 24603988 | 2014 | Pristimerin, a quinonemethide triterpenoid, induces apoptosis in pancreatic cancer cells through the inhibition of pro-survival Akt/NF-κB/mTOR signaling proteins and anti-apoptotic Bcl-2. | Deeb D et al |
| 24457957 | 2014 | Discrepant NOXA (PMAIP1) transcript and NOXA protein levels: a potential Achilles' heel in mantle cell lymphoma. | Dengler MA et al |
| 24460265 | 2013 | Inhibition of nemo-like kinase increases taxol sensitivity in laryngeal cancer. | Dong JR et al |
| 9624412 | 1998 | Cyclin D1 and human neoplasia. | Donnellan R et al |
| 24847381 | 2014 | Predictive simulation approach for designing cancer therapeutic regimens with novel biological mechanisms. | Doudican NA et al |
| 24505359 | 2014 | MicroRNA-545 suppresses cell proliferation by targeting cyclin D1 and CDK4 in lung cancer cells. | Du B et al |
| 24905577 | 2014 | Estrogen induces Vav1 expression in human breast cancer cells. | Du MJ et al |
| 24797571 | 2014 | Microvesicles derived from human Wharton's jelly mesenchymal stem cells promote human renal cancer cell growth and aggressiveness through induction of hepatocyte growth factor. | Du T et al |
| 24812569 | 2014 | Proapoptotic and Antiproliferative Effects of Thymus caramanicus on Human Breast Cancer Cell Line (MCF-7) and Its Interaction with Anticancer Drug Vincristine. | Esmaeili-Mahani S et al |
| 23674142 | 2013 | miR-449b inhibits the proliferation of SW1116 colon cancer stem cells through downregulation of CCND1 and E2F3 expression. | Fang Y et al |
| 24804719 | 2014 | Piperine causes G1 phase cell cycle arrest and apoptosis in melanoma cells through checkpoint kinase-1 activation. | Fofaria NM et al |
| 22912826 | 2012 | The effect of miR-338-3p on HBx deletion-mutant (HBx-d382) mediated liver-cell proliferation through CyclinD1 regulation. | Fu X et al |
| 24331535 | 2013 | Resveratrol induces apoptosis and autophagy in T-cell acute lymphoblastic leukemia cells by inhibiting Akt/mTOR and activating p38-MAPK. | Ge J et al |
| 24396396 | 2014 | FHIT overexpression in HepG2 hepatoma cells affects growth and cyclin D1 expression in vitro. | Ge J et al |
| 24661491 | 2014 | Primary diffuse large B-cell lymphoma developing within a rectal tubular adenoma with low-grade dysplasia: a case report. | Genovese F et al |
| 19556426 | 2009 | How I treat mantle cell lymphoma. | Ghielmini M et al |
| 24312381 | 2013 | FABP4 induces vascular smooth muscle cell proliferation and migration through a MAPK-dependent pathway. | Girona J et al |
| 24912819 | 2014 | Therapeutic effect of a multi-targeted imidazolium compound in hepatocellular carcinoma. | Gopalan B et al |
| 24440705 | 2014 | MicroRNA-490-3p inhibits proliferation of A549 lung cancer cells by targeting CCND1. | Gu H et al |
| 24149576 | 2013 | Akt-p53-miR-365-cyclin D1/cdc25A axis contributes to gastric tumorigenesis induced by PTEN deficiency. | Guo SL et al |
| 24930892 | 2014 | Transcriptional profiles of SHH pathway genes in keratocystic odontogenic tumor and ameloblastoma. | Gurgel CA et al |
| 9671416 | 1998 | FLRG (follistatin-related gene), a new target of chromosomal rearrangement in malignant blood disorders. | Hayette S et al |
| 24690900 | 2014 | The pro-proliferative effects of nicotine and its underlying mechanism on rat airway smooth muscle cells. | He F et al |
| 24648939 | 2013 | Genetic polymorphism of CCND1 G870A and esophageal cancer susceptibility: A meta-analysis. | He W et al |
| 23836497 | 2013 | MiRNA-20 and mirna-106a regulate spermatogonial stem cell renewal at the post-transcriptional level via targeting STAT3 and Ccnd1. | He Z et al |
| 14633913 | 2004 | Real-time quantitative reverse transcription-PCR for cyclin D1 mRNA in blood, marrow, and tissue specimens for diagnosis of mantle cell lymphoma. | Howe JG et al |
| 24802408 | 2014 | JMJD2A predicts prognosis and regulates cell growth in human gastric cancer. | Hu CE et al |
| 24248414 | 2014 | miR-449a Regulates proliferation and chemosensitivity to cisplatin by targeting cyclin D1 and BCL2 in SGC7901 cells. | Hu J et al |
| 24504166 | 2014 | MiR-196a promotes pancreatic cancer progression by targeting nuclear factor kappa-B-inhibitor alpha. | Huang F et al |
| 24694877 | 2014 | Embelin suppresses growth of human pancreatic cancer xenografts, and pancreatic cancer cells isolated from KrasG12D mice by inhibiting Akt and Sonic hedgehog pathways. | Huang M et al |
| 23383003 | 2013 | MicroRNA-195 inhibits the proliferation of human glioma cells by directly targeting cyclin D1 and cyclin E1. | Hui W et al |
| 24907662 | 2014 | Nuclear distribution of claudin-2 increases cell proliferation in human lung adenocarcinoma cells. | Ikari A et al |
| 24391010 | 2013 | Genetic polymorphisms of CCND1 and PTEN in progression of esophageal squamous carcinoma. | Jang Y et al |
| 24454966 | 2014 | Drug targets for cell cycle dysregulators in leukemogenesis: in silico docking studies. | Jayaraman A et al |
| 24870761 | 2014 | Anti-proliferation effects and molecular mechanisms of action of tetramethypyrazine on human SGC-7901 gastric carcinoma cells. | Ji AJ et al |
| 23991964 | 2013 | MicroRNA-16 inhibits bladder cancer proliferation by targeting Cyclin D1. | Jiang QQ et al |
| 8449399 | 1993 | Direct binding of cyclin D to the retinoblastoma gene product (pRb) and pRb phosphorylation by the cyclin D-dependent kinase CDK4. | Kato J et al |
| 24470823 | 2013 | Primary congenital sacrococcygeal neuroblastoma: A case report with immunohistochemical study and review of literature. | Khandeparkar SG et al |
| 24878898 | 2014 | A novel herbal medicine, KIOM-C, induces autophagic and apoptotic cell death mediated by activation of JNK and reactive oxygen species in HT1080 human fibrosarcoma cells. | Kim A et al |
| 24648690 | 2014 | Expression of sfrp2 is increased in catagen of hair follicles and inhibits keratinocyte proliferation. | Kim BK et al |
| 24260381 | 2013 | Scoparone exerts anti-tumor activity against DU145 prostate cancer cells via inhibition of STAT3 activity. | Kim JK et al |
| 24819721 | 2014 | MicroRNA-365 inhibits the proliferation of vascular smooth muscle cells by targeting cyclin D1. | Kim MH et al |
| 24418169 | 2014 | YC-1 enhances the anti-tumor activity of sorafenib through inhibition of signal transducer and activator of transcription 3 (STAT3) in hepatocellular carcinoma. | Kong J et al |
| 9106657 | 1997 | New functional activities for the p21 family of CDK inhibitors. | LaBaer J et al |
| 22374674 | 2012 | MicroRNA-143 is a critical regulator of cell cycle activity in stem cells with co-overexpression of Akt and angiopoietin-1 via transcriptional regulation of Erk5/cyclin D1 signaling. | Lai VK et al |
| 24454732 | 2014 | MicroRNA-147 induces a mesenchymal-to-epithelial transition (MET) and reverses EGFR inhibitor resistance. | Lee CG et al |
| 24715915 | 2014 | One Patient, Two Uncommon B-Cell Neoplasms: Solitary Plasmacytoma following Complete Remission from Intravascular Large B-Cell Lymphoma Involving Central Nervous System. | Lee J et al |
| 24303084 | 2013 | Galectin-3 silencing inhibits epirubicin-induced ATP binding cassette transporters and activates the mitochondrial apoptosis pathway via β-catenin/GSK-3β modulation in colorectal carcinoma. | Lee YK et al |
| 24475272 | 2014 | Reevesioside A, a cardenolide glycoside, induces anticancer activity against human hormone-refractory prostate cancers through suppression of c-myc expression and induction of G1 arrest of the cell cycle. | Leu WJ et al |
| 24319687 | 2013 | CD59 underlines the antiatherosclerotic effects of C-phycocyanin on mice. | Li B et al |
| 10329598 | 1999 | Detection of translocation t(11;14)(q13;q32) in mantle cell lymphoma by fluorescence in situ hybridization. | Li JY et al |
| 24760075 | 2014 | Notch-1 signaling promotes the malignant features of human breast cancer through NF-κB activation. | Li L et al |
| 24222746 | 2013 | Cyclin D1 G870A polymorphism and risk of nasopharyngeal carcinoma: a meta-analysis. | Li M et al |
| 24529079 | 2014 | Fenofibrate induces apoptosis of triple-negative breast cancer cells via activation of NF-κB pathway. | Li T et al |
| 24928376 | 2014 | Effects of quercetin on hedgehog signaling in chronic myeloid leukemia KBM7 cells. | Li W et al |
| 24379579 | 2013 | P115 promotes growth of gastric cancer through interaction with macrophage migration inhibitory factor. | Li XJ et al |
| 24728073 | 2014 | Prognostic significance of cyclin D1 expression in colorectal cancer: a meta-analysis of observational studies. | Li Y et al |
| 24342321 | 2013 | Human decorin regulates proliferation and migration of human lung cancer A549 cells. | Liang S et al |
| 24933647 | 2014 | Anti-cancer effects of ursane triterpenoid as a single agent and in combination with cisplatin in bladder cancer. | Lin KW et al |
| 24903225 | 2014 | Obatoclax analog SC-2001 inhibits STAT3 phosphorylation through enhancing SHP-1 expression and induces apoptosis in human breast cancer cells. | Liu CY et al |
| 24268029 | 2013 | PinX1 suppresses bladder urothelial carcinoma cell proliferation via the inhibition of telomerase activity and p16/cyclin D1 pathway. | Liu JY et al |
| 24409330 | 2014 | Overexpression of TRIM24 is associated with the onset and progress of human hepatocellular carcinoma. | Liu X et al |
| 22739938 | 2012 | MiR-138 suppressed nasopharyngeal carcinoma growth and tumorigenesis by targeting the CCND1 oncogene. | Liu X et al |
| 24223635 | 2013 | Expression and significance of angiopoietin-2 and cyclin D1 in laryngeal squamous cell carcinoma and the correlation with prognosis. | Liu YF et al |
| 24932611 | 2014 | CoCl2, a mimic of hypoxia, induces formation of polyploid giant cells with stem characteristics in colon cancer. | Lopez-Sánchez LM et al |
| 24905570 | 2014 | Salinomycin suppresses LRP6 expression and inhibits both Wnt/β-catenin and mTORC1 signaling in breast and prostate cancer cells. | Lu W et al |
| 24644428 | 2014 | CARP, a myostatin-downregulated gene in CFM Cells, is a novel essential positive regulator of myogenesis. | Ma G et al |
| 24415872 | 2014 | Apoptotic pathway induced by diallyl trisulfide in pancreatic cancer cells. | Ma HB et al |
| 17041754 | 2006 | Epigallocatechin gallate induced apoptosis in Sarcoma180 cells in vivo: mediated by p53 pathway and inhibition in U1B, U4-U6 UsnRNAs expression. | Manna S et al |
| 18656336 | 2009 | Tea polyphenols can restrict benzo[a]pyrene-induced lung carcinogenesis by altered expression of p53-associated genes and H-ras, c-myc and cyclin D1. | Manna S et al |
| 24236150 | 2013 | Transforming growth factor β inhibits platelet derived growth factor-induced vascular smooth muscle cell proliferation via Akt-independent, Smad-mediated cyclin D1 downregulation. | Martin-Garrido A et al |
| 15241418 | 2004 | Cyclin-dependent kinases regulate the antiproliferative function of Smads. | Matsuura I et al |
| 24755523 | 2014 | Extracellular activation of Wnt signaling through epigenetic dysregulation of Wnt inhibitory factor-1 (Wif-1) is associated with pathogenesis of adrenocortical tumor. | Mitsui Y et al |
| 24523791 | 2013 | Role of cyclin D1 in breast carcinoma. | Mohammadizadeh F et al |
| 24862759 | 2014 | Prolactin-induced protein is required for cell cycle progression in breast cancer. | Naderi A et al |
| 22326957 | 2012 | A microRNA-21 surge facilitates rapid cyclin D1 translation and cell cycle progression in mouse liver regeneration. | Ng R et al |
| 22072615 | 2012 | microRNA-365, down-regulated in colon cancer, inhibits cell cycle progression and promotes apoptosis of colon cancer cells by probably targeting Cyclin D1 and Bcl-2. | Nie J et al |
| 24455756 | 2014 | The correlation analysis of WWOX expression and cancer related genes in neuroblastoma- a real time RT-PCR study. | Nowakowska M et al |
| 24762088 | 2014 | miR-125b induces cellular senescence in malignant melanoma. | Nyholm AM et al |
| 24931004 | 2014 | Odd-skipped related 1 is a novel tumour suppressor gene and a potential prognostic biomarker in gastric cancer. | Otani K et al |
| 24604959 | 2013 | Cyclin D1 expression in multiple myeloma by immunohistochemistry: Case series of 14 patients and literature review. | Padhi S et al |
| 24820071 | 2014 | Cyclin D1 expression in prostate carcinoma. | Pereira RA et al |
| 23790801 | 2013 | New roles of cyclin D1. | Pestell RG et al |
| 8288131 | 1994 | p27Kip1, a cyclin-Cdk inhibitor, links transforming growth factor-beta and contact inhibition to cell cycle arrest. | Polyak K et al |
| 24833701 | 2014 | Blocking sirtuin 1 and 2 inhibits renal interstitial fibroblast activation and attenuates renal interstitial fibrosis in obstructive nephropathy. | Ponnusamy M et al |
| 24914206 | 2014 | Differential regulation of cyclin D1 expression by protein kinase C α and ϵ signaling in intestinal epithelial cells. | Pysz MA et al |
| 24587659 | 2014 | Sophocarpine attenuates liver fibrosis by inhibiting the TLR4 signaling pathway in rats. | Qian H et al |
| 20133739 | 2010 | MicroRNA-19a mediates the suppressive effect of laminar flow on cyclin D1 expression in human umbilical vein endothelial cells. | Qin X et al |
| 24241174 | 2013 | SL1122-37, a novel derivative of sorafenib, has greater effects than sorafenib on the inhibition of human hepatocellular carcinoma (HCC) growth and prevention of angiogenesis. | Qin Y et al |
| 24574651 | 2013 | Cyclin D1 an early biomarker in oral carcinogenesis. | Ramakrishna A et al |
| 24427302 | 2014 | High expression of IGFBP7 in fibroblasts induced by colorectal cancer cells is co-regulated by TGF-β and Wnt signaling in a Smad2/3-Dvl2/3-dependent manner. | Rao C et al |
| 24811863 | 2014 | FAAH inhibition enhances anandamide mediated anti-tumorigenic effects in non-small cell lung cancer by downregulating the EGF/EGFR pathway. | Ravi J et al |
| 24602161 | 2014 | The impact of cyclin D1 overexpression on the prognosis of bladder cancer: a meta-analysis. | Ren B et al |
| 8204893 | 1994 | Rearrangement of CCND1 (BCL1/PRAD1) 3' untranslated region in mantle-cell lymphomas and t(11q13)-associated leukemias. | Rimokh R et al |
| 24708576 | 2014 | Evaluation of protein biomarkers of prostate cancer aggressiveness. | Rizzardi AE et al |
| 24223883 | 2013 | Alterations in the cell cycle in the cerebellum of hyperbilirubinemic Gunn rat: a possible link with apoptosis? | Robert MC et al |
| 16531763 | 2006 | Genetic alterations (amplification and rearrangement) of D-type cyclins loci in head and neck squamous cell carcinoma of Indian patients: prognostic significance and clinical implications. | Sabbir MG et al |
| 18367489 | 2008 | Genomic aberrations in mantle cell lymphoma detected by interphase fluorescence in situ hybridization. Incidence and clinicopathological correlations. | Sander S et al |
| 24678876 | 2014 | Calcitriol restores antiestrogen responsiveness in estrogen receptor negative breast cancer cells: a potential new therapeutic approach. | Santos-Martínez N et al |
| 24377544 | 2013 | Reduction of proliferation and induction of apoptosis are associated with shrinkage of head and neck squamous cell carcinoma due to neoadjuvant chemotherapy. | Sarkar S et al |
| 24860672 | 2014 | Histological Characterization of Biliary Intraepithelial Neoplasia with respect to Pancreatic Intraepithelial Neoplasia. | Sato Y et al |
| 24244355 | 2013 | Pifithrin-μ, an inhibitor of heat-shock protein 70, can increase the antitumor effects of hyperthermia against human prostate cancer cells. | Sekihara K et al |
| 24667650 | 2014 | ERRβ signalling through FST and BCAS2 inhibits cellular proliferation in breast cancer cells. | Sengupta D et al |
| 24618206 | 2014 | Therapeutic effects of lentivirus-mediated shRNA targeting of cyclin D1 in human gastric cancer. | Seo JH et al |
| 10385618 | 1999 | CDK inhibitors: positive and negative regulators of G1-phase progression. | Sherr CJ et al |
| 15856299 | 2005 | Deletion in chromosome 11 and Bcl-1/Cyclin D1 alterations are independently associated with the development of uterine cervical carcinoma. | Singh RK et al |
| 24348821 | 2014 | Expression of p-Akt in ovarian serous carcinoma and its association with proliferation and apoptosis. | Song T et al |
| 24650096 | 2014 | Alpha-enolase as a potential cancer prognostic marker promotes cell growth, migration, and invasion in glioma. | Song Y et al |
| 24806449 | 2014 | The Down syndrome-related protein kinase DYRK1A phosphorylates p27(Kip1) and Cyclin D1 and induces cell cycle exit and neuronal differentiation. | Soppa U et al |
| 12231535 | 2002 | Identification of cyclin D1 mRNA overexpression in B-cell neoplasias by real-time reverse transcription-PCR of microdissected paraffin sections. | Specht K et al |
| 9619726 | 1998 | Molecular cytogenetic analysis of B-cell chronic lymphocytic leukemia. | Stilgenbauer S et al |
| 24932295 | 2014 | Resveratrol inhibits the phosphatidylinositide 3-kinase/protein kinase B/mammalian target of rapamycin signaling pathway in the human chronic myeloid leukemia K562 cell line. | Sui T et al |
| 18406353 | 2008 | Downregulation of CCND1 and CDK6 by miR-34a induces cell cycle arrest. | Sun F et al |
| 24903677 | 2015 | A comprehensive immunophenotypic marker analysis of hairy cell leukemia in paraffin-embedded bone marrow trephine biopsies--a tissue microarray study. | Tóth-Lipták J et al |
| 24484634 | 2014 | Interleukin-6 is required for cell cycle arrest and activation of DNA repair enzymes after partial hepatectomy in mice. | Tachibana S et al |
| 24289559 | 2013 | Triptolide inhibits histone methyltransferase EZH2 and modulates the expression of its target genes in prostate cancer cells. | Tamgue O et al |
| 24821722 | 2014 | Repression of cyclin D1 expression is necessary for the maintenance of cell cycle exit in adult mammalian cardiomyocytes. | Tane S et al |
| 24815438 | 2014 | Effects of ubiquitin-conjugating enzyme 2C on invasion, proliferation and cell cycling of lung cancer cells. | Tang XK et al |
| 24289556 | 2013 | Targeting SHCBP1 inhibits cell proliferation in human hepatocellular carcinoma cells. | Tao HC et al |
| 24655584 | 2014 | Gelam honey attenuated radiation-induced cell death in human diploid fibroblasts by promoting cell cycle progression and inhibiting apoptosis. | Tengku Ahmad TA et al |
| 24348824 | 2014 | Frizzled-2: A potential novel target for molecular pancreatic cancer therapy. | Tomizawa M et al |
| 24278325 | 2013 | Distinct effects of alcohol consumption and smoking on genetic alterations in head and neck carcinoma. | Urashima M et al |
| 15723720 | 2005 | Numerical abnormalities of the Cyclin D1 gene locus on chromosome 11q13 in non-melanoma skin cancer. | Utikal J et al |
| 24816720 | 2014 | The suppressive role of SOX7 in hepatocarcinogenesis. | Wang C et al |
| 24568493 | 2014 | Fangchinoline inhibits cell proliferation via Akt/GSK-3beta/ cyclin D1 signaling and induces apoptosis in MDA-MB-231 breast cancer cells. | Wang CD et al |
| 24688409 | 2014 | Effects of thapsigargin on the proliferation and survival of human rheumatoid arthritis synovial cells. | Wang H et al |
| 24559156 | 2014 | Estrogen receptor α-coupled Bmi1 regulation pathway in breast cancer and its clinical implications. | Wang H et al |
| 24722568 | 2014 | Leucyl-tRNA synthetase regulates lactation and cell proliferation via mTOR signaling in dairy cow mammary epithelial cells. | Wang L et al |
| 24886245 | 2014 | Acylglycerol kinase promotes cell proliferation and tumorigenicity in breast cancer via suppression of the FOXO1 transcription factor. | Wang X et al |
| 24454488 | 2013 | A C 21 -Steroidal Glycoside Isolated from the Roots of Cynanchum auriculatum Induces Cell Cycle Arrest and Apoptosis in Human Gastric Cancer SGC-7901 Cells. | Wang YQ et al |
| 24811890 | 2014 | Unique molecular signatures as a hallmark of patients with metastatic breast cancer: implications for current treatment paradigms. | Wheler JJ et al |
| 8172823 | 1994 | Cyclin D1 overexpression in non-Hodgkin's lymphoma with chromosome 11 bcl-1 rearrangement. | Williams ME et al |
| 15306823 | 2004 | Variant t(2;11)(p11;q13) associated with the IgK-CCND1 rearrangement is a recurrent translocation in leukemic small-cell B-non-Hodgkin lymphoma. | Wlodarska I et al |
| 24656144 | 2014 | Prostaglandin E2 alters Wnt-dependent migration and proliferation in neuroectodermal stem cells: implications for autism spectrum disorders. | Wong CT et al |
| 24558429 | 2014 | Mir-184 post-transcriptionally regulates SOX7 expression and promotes cell proliferation in human hepatocellular carcinoma. | Wu GG et al |
| 24694542 | 2014 | RETRACTED: Silencing of Ether à go-go 1 by shRNA inhibits osteosarcoma growth and cell cycle progression. | Wu J et al |
| 24739942 | 2014 | Sodium tanshinone IIA silate inhibits high glucose-induced vascular smooth muscle cell proliferation and migration through activation of AMP-activated protein kinase. | Wu WY et al |
| 24742286 | 2014 | Activation of Akt1 accelerates carcinogen-induced tumorigenesis in mammary gland of virgin and post-lactating transgenic mice. | Wu Y et al |
| 24289849 | 2013 | The inhibitory role of Mir-29 in growth of breast cancer cells. | Wu Z et al |
| 24854087 | 2014 | Silencing of KIF14 interferes with cell cycle progression and cytokinesis by blocking the p27(Kip1) ubiquitination pathway in hepatocellular carcinoma. | Xu H et al |
| 24570594 | 2014 | miR-7 inhibits colorectal cancer cell proliferation and induces apoptosis by targeting XRCC2. | Xu K et al |
| 24812621 | 2014 | α-Mangostin suppresses the viability and epithelial-mesenchymal transition of pancreatic cancer cells by downregulating the PI3K/Akt pathway. | Xu Q et al |
| 23731275 | 2013 | MicroRNA-503 suppresses proliferation and cell-cycle progression of endometrioid endometrial cancer by negatively regulating cyclin D1. | Xu YY et al |
| 24333727 | 2014 | MicroRNA miR-302 inhibits the tumorigenicity of endometrial cancer cells by suppression of Cyclin D1 and CDK1. | Yan GJ et al |
| 17176475 | 2006 | Variations in cyclin D1 levels through the cell cycle determine the proliferative fate of a cell. | Yang K et al |
| 24278101 | 2013 | Resveratrol inhibits the growth of gastric cancer by inducing G1 phase arrest and senescence in a Sirt1-dependent manner. | Yang Q et al |
| 18695042 | 2008 | A cyclin D1/microRNA 17/20 regulatory feedback loop in control of breast cancer cell proliferation. | Yu Z et al |
| 24287487 | 2013 | Cyclin D1 induction of Dicer governs microRNA processing and expression in breast cancer. | Yu Z et al |
| 24396481 | 2014 | Antitumor effect and mechanism of an ellagic acid derivative on the HepG2 human hepatocellular carcinoma cell line. | Zhang H et al |
| 24489938 | 2014 | Sodium ferulate inhibits neointimal hyperplasia in rat balloon injury model. | Zhang J et al |
| 24265754 | 2013 | Overexpression of JAM-A in non-small cell lung cancer correlates with tumor progression. | Zhang M et al |
| 24936138 | 2014 | MicroRNA-365 inhibits vascular smooth muscle cell proliferation through targeting cyclin D1. | Zhang P et al |
| 24386256 | 2013 | Silybin-mediated inhibition of Notch signaling exerts antitumor activity in human hepatocellular carcinoma cells. | Zhang S et al |
| 24351837 | 2013 | The antidiabetic drug metformin inhibits the proliferation of bladder cancer cells in vitro and in vivo. | Zhang T et al |
| 22319632 | 2012 | MicroRNA-520b inhibits growth of hepatoma cells by targeting MEKK2 and cyclin D1. | Zhang W et al |
| 24444073 | 2014 | Gene therapy of ovarian cancer using IL-21-secreting human umbilical cord mesenchymal stem cells in nude mice. | Zhang Y et al |
| 24348867 | 2014 | Polydatin inhibits growth of lung cancer cells by inducing apoptosis and causing cell cycle arrest. | Zhang Y et al |
| 24528032 | 2014 | Overexpression of NDRG2 can inhibit neuroblastoma cell proliferation through negative regulation by CYR61. | Zhang ZG et al |
| 24658335 | 2014 | Gallotannin imposes S phase arrest in breast cancer cells and suppresses the growth of triple-negative tumors in vivo. | Zhao T et al |
| 24454925 | 2014 | Downregulation of PAX6 by shRNA inhibits proliferation and cell cycle progression of human non-small cell lung cancer cell lines. | Zhao X et al |
| 24675814 | 2014 | Cyclin D1 overexpression is associated with poor clinicopathological outcome and survival in oral squamous cell carcinoma in Asian populations: insights from a meta-analysis. | Zhao Y et al |
| 23383271 | 2013 | microRNA-9 suppresses the proliferation, invasion and metastasis of gastric cancer cells through targeting cyclin D1 and Ets1. | Zheng L et al |
| 9209653 | 1997 | Bcl-1/cyclin D1 in malignant lymphoma. | de Boer CJ et al |
Other Information
Locus ID:
NCBI: 595
MIM: 168461
HGNC: 1582
Ensembl: ENSG00000110092
Variants:
dbSNP: 595
ClinVar: 595
TCGA: ENSG00000110092
COSMIC: CCND1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000110092 | ENST00000227507 | P24385 |
| ENSG00000110092 | ENST00000227507 | Q6FI00 |
| ENSG00000110092 | ENST00000536559 | F5H437 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA10040 | cetuximab | Chemical | ClinicalAnnotation | associated | PD | 16788380, 18349392, 22117530 | |
| PA128406956 | fluorouracil | Chemical | ClinicalAnnotation | associated | PD | 23567490 | |
| PA152241907 | lapatinib | Chemical | VariantAnnotation | associated | PD | ||
| PA443560 | Breast Neoplasms | Disease | VariantAnnotation | associated | PD | ||
| PA443756 | Colonic Neoplasms | Disease | ClinicalAnnotation | associated | PD | 23567490 | |
| PA446108 | Colorectal Neoplasms | Disease | ClinicalAnnotation | associated | PD | 16788380, 18349392, 22117530 | |
| PA446155 | Precursor Cell Lymphoblastic Leukemia-Lymphoma | Disease | ClinicalAnnotation | associated | PD | 12972956, 16870553 | |
| PA450428 | methotrexate | Chemical | ClinicalAnnotation | associated | PD | 12972956, 16870553 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 36066507 | 2024 | MiR-623 links lncRNA RP11-89 and cyclin D1 to regulate the proliferation of glioblastoma cells. | 0 |
| 37610179 | 2024 | Different expression of DACT1, DACT2, and CYCLIN D1 genes in human colorectal cancer tissues and its association with clinicopathological characteristics. | 0 |
| 37930255 | 2024 | ORAOV1, CCND1, and MIR548K Are the Driver Oncogenes of the 11q13 Amplicon in Squamous Cell Carcinoma. | 1 |
| 37992567 | 2024 | NEIL3 promotes cell proliferation of ccRCC via the cyclin D1-Rb-E2F1 feedback loop regulation. | 0 |
| 38301962 | 2024 | Optical genomic mapping is a helpful tool for detecting CCND1 rearrangements in CD5-negative small B-cell lymphoma: Two cases of leukemic non-nodal mantle cell lymphoma. | 0 |
| 38316778 | 2024 | G-quadruplexes promote the motility in MAZ phase-separated condensates to activate CCND1 expression and contribute to hepatocarcinogenesis. | 1 |
| 38349334 | 2024 | Periodic changes of cyclin D1 mRNA stability are regulated by PC4 modifications in the cell cycle. | 0 |
| 38396691 | 2024 | Overexpression of Alpha-1 Antitrypsin Increases the Proliferation of Mesenchymal Stem Cells by Upregulation of Cyclin D1. | 0 |
| 38478555 | 2024 | The oncogene cyclin D1 promotes bipolar spindle integrity under compressive force. | 0 |
| 38488492 | 2024 | FAK mediates hypoxia-induced pulmonary artery smooth muscle cell proliferation by modulating mitochondrial transcription termination factor 1/cyclin D1. | 0 |
| 38497174 | 2024 | LncRNA FOXD3-AS1 Contributes to Glioblastoma Progression Via Sponging miR-3918 to Upregulate CCND1. | 0 |
| 38556083 | 2024 | Increased expression of long non-coding RNA FIRRE promotes hepatocellular carcinoma by HuR-CyclinD1 axis signaling. | 0 |
| 38557526 | 2024 | Expression of Cyclin D1 in Urothelial Carcinoma of Urinary Bladder and its Association with Tumour Grade. | 0 |
| 38862441 | 2024 | [Overexpression of lncRNA FEZF1-AS1 promotes progression of non-small cell lung cancer via the miR-130a-5p/CCND1 axis]. | 0 |
| 36066507 | 2024 | MiR-623 links lncRNA RP11-89 and cyclin D1 to regulate the proliferation of glioblastoma cells. | 0 |
Citation
Shreya Sarkar ; Chinmay Kumar Panda
CCND1 (B-cell leukemia/lymphoma 1)
Atlas Genet Cytogenet Oncol Haematol. 2015-04-01
Online version: http://atlasgeneticsoncology.org/gene/36/ccnd1-(b-cell-leukemia-lymphoma-1)
Historical Card
1998-05-01 CCND1 (B-cell leukemia/lymphoma 1) by Jean-Loup Huret Affiliation
