Chronic myelogenous leukaemia (CML)
2008-08-01 Ali G Turhan Affiliation1.Pole de Biologie-Sante - 40 avenue du Recteur Pineau - 86022 Poitiers Cedex, France
2.Genetics, Dept Medical Information, University of Poitiers, CHU Poitiers Hospital, F-86021 Poitiers, France
Clinics and Pathology
Disease
CML is a malignant chronic myeloproliferative disorder (MPD) of the hematopoietic stem cell.
Phenotype stem cell origin
Evidence exists for the involvement of the most primitive and quiescent hematopoietic stem cell compartiment (CD34+/CD38-, Thy1+): t(9;22) is found in myeloid progenitor and in B-lymphocytes progenitors, but, involvement of the T-cell lineage is extremely rare. The existence of a highly quiescent stem cell population has been demonstrated in patients with CML. More recently, the presence of Ph chromosome has been demonstrated in vascular endothelium of CML patients at diagnosis. Ph+ stem cells with stem markers of endothelial cells have been shown to be present at the level of single stem cells. These findings have suggested that a putative "hemangioblast" giving rise to both hematopoietic and endothelial cells could be present on adult marrow and be a target of t(9;22) translocation.
Epidemiology
Annual incidence: 10/106 (from 1/106 in childhood to 30/106 after 60 yrs);
median age: 30-60 yrs;
sex ratio: 1.2M/1F.
median age: 30-60 yrs;
sex ratio: 1.2M/1F.
Clinics
The disease is currently discovered after a routine blood count revealing hyperleucocytosis and circulating immature white blood cells. A splenomegaly might be present. Bone marrow aspirate with cytogenetic analysis is required, as well as molecular evaluation for the detection of BCR-ABL oncogene which is quantified by calculating BCR-ABL / ABL ratio. The disease is classified most commonly using Sokal or Hasford scores. The natural history of the disease including classically three phases (Chronic phase, accelerated phase and blast crisis) has been profoundly modified by the current therapy regimens using tyrosine kinase inhibitors. For instance, recent update of the IRIS study demonstrates the progressive reduction of secondary events over time with no blast crisis occurring after 6 years. Most patients now seen in accelerated phase or in blastic phase are those who relapse after IM and/or dasatinib/Nilotinib therapies. The major problems in CML are the resistance encountered as first line therapy as well as intolerance to TKI therapy leading to discontinuation of the drugs. In the IRIS trial, approximately 30% of patients discontinue imatinib for reasons of resistance (15%) or intolerance. Finally, it is clear that currently available TKI therapies do not eradicate the most primitive stem cells, explaining relapses occurring after discontinuation of treatment (see below).
Cytology
Hyperplastic bone marrow; myeloid proliferation with maturation; some myelodysplastic features can be seen on Imatinib therapy after disappearance of the Ph clone. The significance of these abnormalities is not known but very rare patients evolve into myelodysplastic or leukemic phase. The typical AL cytology can be seen when patients evolve into accelerated or blast phase (see: t(9;22)(q34;q11) in ALL, t(9;22)(q34;q11) in AML).
Treatment
The treatment of CML has been revolutionized by the introduction of targeted therapies to the clinical practice. The first of these drugs was imatinib mesylate (IM) targeting the tyrosine kinase activity of BCR-ABL. In a large international multi-center trial, the use of IM as a first line therapy has been compared to standard IFN-ARA-C regimen (IRIS trial). This trial has clealy shown the advantages of IM in terms of complete haematological response, (CHR) complete cytogenetic response (CCR) and molecular response (MR) as compared to IFN-ARA-C regimen. Interestingly, the outcome on IM therapy has been shown to be correlate with the Sokal score at diagnosis. The most recent update of this trial has shown for the first time, a reduction of secondary events (accelerated phase, blast crisis) over time, with no patients progressing towards blast crisis after 6 years of therapy. However, several questions remain to be solved before demonstration of a "cure" under IM therapy: 1-The attempts to interrupt IM therapy has been followed in most cases by a cytogenetic and haematological relapse; 2-The most primitive stem cells seem to be resistant to IM therapy at least in vitro ; 3-The resistance to IM-therapy has been found to be associated, especially in patients who received it as second line treatment, with the occurrence of mutations in the ABL-kinase domain, impeding the binding of the drug to its target. Some of these mutations, occurring in the P-loop or in the ATP binding pocket ("gatekeeper" mutation T315I ) lead to a total resistance to IM; requiring the interruption of the drug. If the mutations reside outside these regions (C-lobe of the SH1 kinase domain), the increase of IM dose could lead to molecular responses. Detection of ABL-kinase mutations has therefore become a clinically useful molecular test. The majority of ABL-kinase mutations other than T315I have been shown to be targetable by second generation of TK inhibitors, essentially Dasatinib (Sprycell) and Nilotinib (Tasigna). Dasatinib is a combined SRC and ABL-inhibitor which is indicated as a second line therapy in CML patients failing on IM therapy or intolerant to IM-therapy. Nilotinib is a TK inhibitor with high affinity for ABL-tyrosine kinase. Novel therapies to eliminate the stem cells with T315I mutation remain a major futur challenge.
Prognosis
TKI therapies have changed the prognosis of CML as well as the natural history of the disease. Results from the IRIS trial suggest that the majority of patients with CML at first chronic phase will attain complete cytogenetic and major molecular responses with however persistence of minimal residual leukemic stem cells. Although it is difficult to qualify CML as an indolent disease, the estimated median survival with the available therapies is 25 years.
Cytogenetics
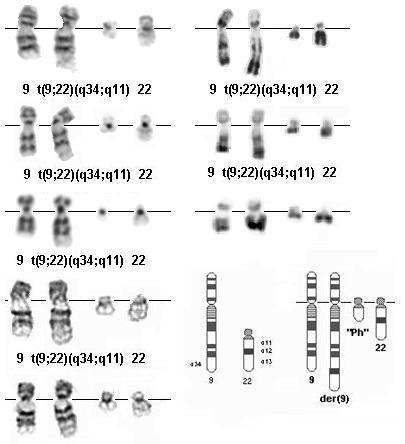
t(9;22)(q34;q11) G- banding (left) - Courtesy Jean-Luc Lai and Alain Vanderhaegen (3 top) and Diane H. Norback, Eric B. Johnson, and Sara Morrison-Delap, UW Cytogenetic Services (2 bottom); R-banding (right) top: Editor; 2 others Courtesy Jean-Luc Lai and Alain Vanderhaegen); diagram and breakpoints (Editor).
Cytogenetics morphological
all CML patients have a t(9;22), at least at the molecular level (see below); but not all t(9;22) are found in CML: this translocation may also be seen in ALL, and in AML (see: t(9;22)(q34;q11) in ALL, t(9;22)(q34;q11) in AML), t(9;22) is also the hallmark of chronic neutrophilic leukemia which presents clinically with absence of circulating immature myeloid cells. In this translocation BCR breakpoint occurs in the micro-BCR region, with the e19-a2 junction at the DNA level and a large protein (BCR-ABL p230) with increased TK activity. It should be noted that the product of the reciprocal translocation ABL-BCR can also be detected in CML cells, the signification of which is unclear.
Cytogenetics molecular
Is a useful tool for diagnostic ascertainment in the case of a masked Philadelphia chromosome, where chromosomes 9 and 22 all appear to be normal, but where cryptic insertion of 3 ABL within a chromosome 22 can be demonstrated.
Additional anomalies
Deletion of the derivative chromosome 9: Detected at diagnosis (n 10% of patients), probably indicating a genetic instability phenotype, this finding has been associated to the more aggressive behavior of the disease, a poor prognostic factor potentially reversed by the use of imatinib mesylate.
Since the introduction of imatinib mesylate to the clinical practice, chromosomal abnormalities have been detected in Ph-negative metaphases in patients in CCR. The most frequent abnormalities are -7, +8 and in very rare cases they are associated to the development of MDS or AML. The direct role of TKI therapies in these abnormalities is difficult to assess in vivo. However, in vitro, it has been demonstrated that imatinib induces centrosome and chromosome aberrations in normal human dermal fibroblasts.
Variants
Chromosome, are found in 5-10% of cases; however, 9q34-3ABL always joins 22q11-5BCR in true CML; the third chromosome and breakpoint is, at times, not random. In a way, masked Philadelphia chromosomes (see above) are also variants.
Genes Involved and Proteins
Gene name
ABL1 (v-abl Abelson murine leukemia viral oncogene homolog 1)
Location
9q34.12
Dna rna description
Alternate splicing (1a and 1b) in 5.
Protein description
Giving rise to 2 proteins of 145 kDa; contains SH (SRC homology) domains; N-term SH3 and SH2 - SH1 (tyrosine kinase) - DNA binding motif - actin binding domain C-term; widely expressed; localisation is mainly nuclear; inhibits cell growth.
Gene name
BCR (Breakpoint cluster region)
Location
22q11.23
Dna rna description
Various splicings.
Protein description
Main form: 160 KDa; N-term Serine-Treonine kinase domain, SH2 binding, and C-term domain which functions as a GTPase activating protein for p21rac; widely expressed; cytoplasmic localisation; protein kinase; probable role in signal transduction.
Result of the Chromosomal Anomaly
Description
1. The crucial event lies on der(22), id est 5 BCR/3 ABL hybrid gene is pathogenic, while ABL/BCR may or may not be expressed;
2. Breakpoint in ABL is variable over a region of 200 kb, often between the two alternative exons 1b and 1a, sometimes 5 of 1b, or 3 of 1a, but always 5 of exon 2;
3. Breakpoint in BCR is in a narrow region, therefore called M-bcr (for major breakpoint cluster region), a cluster of 5.8 kb, between exons 12 and 16, also called b1 to b5 of M-bcr; most breakpoints being either between b2 and b3, or between b3 and b4
2. Breakpoint in ABL is variable over a region of 200 kb, often between the two alternative exons 1b and 1a, sometimes 5 of 1b, or 3 of 1a, but always 5 of exon 2;
3. Breakpoint in BCR is in a narrow region, therefore called M-bcr (for major breakpoint cluster region), a cluster of 5.8 kb, between exons 12 and 16, also called b1 to b5 of M-bcr; most breakpoints being either between b2 and b3, or between b3 and b4
Transcript
8.5 kb mRNA, resulting in a 210 KDa chimeric protein with increased tyrosine kinase activity.
Detection protocole
RT-PCR for minimal residual disease detection. Current detection methods of BCR-ABL make use of quantitative PCR analyses, using essentially TaqMan or SybR green technologies. The amount of BCR-ABL is compared to an endogenous gene, most of laboratories using ABL or BCR. The ratio of BCR-ABL / ABL at diagnosis is then followed during the treatment which induces a reduction of this ratio which is expressed by two different manners: 1-An absolute number resulting from the BCR-ABL/ABL ratio is quantified, and the reduction of this ratio by 3-log under therapy represents "major molecular response" or MMR. Or 2- The BCR-ABL /ABL ratio is quantified at diagnosis, representing 100% of BCR-ABL amount detected for a given patient using the RQ-PCR technology used in the laboratory. This ratio expressed as a percentage is calculated at each timepoint during the follow-up on therapy, a percentage of 0.1% (3-logreduction from the diagnosis) signifies MMR. The introduction of this international scale allows the comparison of results from different laboratories.
Description
P210 with the first 902 or 927 amino acids from BCR; BCR/ABL has a cytoplasmic localization, in contrast with ABL, mostly nuclear. The shuttling of ABL between the nucleus and cytoplasm is accomplished by the presence of import and export signal sequences in the COOH-terminal region of ABL. Interestingly, these sequences are conserved in BCR-ABL. It has been shown that the inhibition of BCR-ABL export from the nucleus has been shown to induce apoptosis in BCR-ABL-expressing cells. This inhibition is accomplished by the use of export inhibitors such as leptomycin.
Oncogenesis
A- Major molecular pathways activated by BCR-ABL.BCR/ABL activates RAS signaling through the GRB2 adaptor molecule which interacts specifically with the Y177 of BCR. PI3-K (phosphatidyl inositol 3 kinase) pathway is also activated with secondary activation of the AKT/PKB pathway. Integrity of transcription machinery induced by MYC is necessary for the transforming action of BCR-ABL. More recently, activation of STAT (Signal transducers and activators of transcription) molecules has been described as a major molecular signaling event induced by BCR-ABL, with activation of essentially STAT5, STAT1, and STAT6. Activation of the molecules of the focal adhesion complex ( PAXILLIN, FAK) by BCR-ABL requires the role of the adaptor molecule CRK-L. BCR-ABL activates negative regulatory molecules such as PTP1B and Abi-1 and their inactivation could be associated with progression into blast crisis.B- Correlations between molecular pathways and leukemic phenotype observed in primary CML cells or in BCR-ABL-transduced cells are currently limited. BCR-ABL has anti-apoptotic activity (PI63K/Akt/STAT5) . BCR/ABL induces cell adhesive and migratory abnormalities in vitro in the presence of fibronection or in transwell assays (Abnormal integrin signaling/FAK/CRK-L/Abnormal response to chemokine SDF-1). The involvement of CXCR-4 and SDF-1 axis has also recently been shown: BCR-ABL down-regulates CXCR-4 expression with a dose-dependent effect. This could explain the inability of BCR-Abl-expressing leukemic cells and progenitors to the marrow niches where SDF-1 (the ligand of CXCR-4) is present. Under the IM-therapy, the up-regulation of CXCR-4 expression could induce the migration of some stem cells which escape from the effect of IM-therapy, to the bone marrow niches where these cells become quiescent. Other adhesion receptors such as CD44 have been shown to be altered in CML and in a murine CML model generated in CD44 K/O mice, the leukemogenic ability of the BCR-ABL-expressing stem cells is impaired. BCR-ABL induces a dose-effect relationship in CML cells with increased BCR-ABL mRNA during progression into blast crisis, with induction of genetic instability. During recent years molecular pathways involved in the genetic instability phenotype have been extensively studied. BCR-ABL has been shown to contribute directly to the generation of a "mutator" phenotype as well as to the overexpression of DNA-polymerase-beta which is prone to copying errors during DNA replication. BCR-ABL is directly responsible of the down-regulation of the major DNA repair protein DNA-PKcs and that of BRCA1, both of these events taking place at the post-transcriptional level. Conversely, BCR-ABL increases expression of RAD51, through a STAT5-induced mechanism, and might contribute to drug resistance. Recent data suggest that BCR-ABL not only interferes with DNA repair pathways but also contributes to DNA damage by increasing the production of reactive oxygen species (ROS). There is evidence suggesting that ROS activity induced by BCR-ABL could contribute to the occurrence of ABL-kinase mutations. Leukemic cell populations expressing high levels of BCR-ABL TK activity (such as stem cells) might be predisposed to the occurrence of these mutations. Thus, BCR-ABL generates conditions of genetic damage in the presence of inappropriate DNA repair capacity. Beside DNA -PKcs and Rad 51, DNA repair pathways have been shown to be altered by BCR-ABL activity including Bloom protein, WRN helicase, ATM and BRCA1. Molecular events associated with blast crisis: BCR-ABL expression increases during progression of CML, from CP to blastic phase. This increased expression plays potentially a role, in the downstream signalling events as this has been shown in in vitro models. Several molecular events have now been explored in the last decade such as P53 mutation (30% of patients) methylation of internal ABL promoter, telomere shortening, Abi-1 inactivation. Some molecular events are detected more frequently in lymphoid blast crisis ((p16/ARF mutations, Rb mutations, Ikaros deletions). In a minority of patients, there are additional translocations detected such as t(3;21)(q26;q22) translated into a AML1 / EVI1 fusion protein. It should be noted however that in the TKI therapy era, the most common genetic abnormality detected is the occurrence of ABL-kinase mutations. Interaction of BCR-ABL with RNA binding proteins. Recently, there has been progress in the understanding of the mechanisms of impaired differentiation. BCR-ABL has been shown to downregulate the expression of CCAAT enhancer-binding protein alpha (CEBP-alpha) at the protein level by acting on hnRNPE2, which belongs to a family of RNA binding proteins. CEBP-alpha expression is required for normal granulocytic differentiation via induction of G-CSF-R gene expression. BCR-ABL has been shown to increase the expression of several classes of RNA binding proteins either at the transcriptional level (hnRNPA2/B1, hnRNPK, hnRNPD1) or by stablizing the protein (hnRNPA1, hnRNPE2). These RNA binding proteins interact with RNA molecules exported from the nucleus and change the fate of the proteins by inhibition or stimulating the translation, or by stabilizing the protein levels by inhibition of the proteasome-induced degradation. Some RNA bindings proteins regulated by BCR-ABL lead to enhanced proliferation and survival of CML cells, possibly contributing the occurrence of blast crisis. Among those, increased levels of la/SSB, lead to an increased translation of MDM with low p53 levels and inhibition of apoptosis. Via hnRNPA1, BCR-ABL has been shown to increase the nuclear oncogene SET (TAF-1B) which is an inhibitor of PP2A, a serine threonine phosphatase which is a negative regulator of RAS / MAPK and PI-3K pathways. PP2A plays also a major role in controlling cell cycle and DNA replication. PP2A is thought to be a tumor suppressor protein whose expression is reduced in tumors. By stimulating the PP2A inhibitor SET, BCR-ABL lead to reduced levels of PP2A which could contribute to blast crisis as suggested by the foowing facts: 1- In blast crisis cells SET activity is high with low PP2A levels; 2- Inhibition of SET by forskolin or an shRNA approach, lead to restoration of PP2A levels and inhibition of several signalling pathways controlled by BCR-ABL, including AKT, Jak2, STAT and Myc. The global result in different models leads to the reversion of BC phenotype (reduced proliferation, increased differentiation) even in imatinib resistant (including T315I) leukemic cells. A clinical grade PP2A activator, fingolimod is currently in clinical trials. Target cell for blast crisis: There is recent data suggesting that blast crisis could occur not at the level of hematopoietic stem cell but at the level of a CFU-GM. The data in favour of this hypothesis have been obtained by analyzing the levels of beta-catenin activation (target of WNT signalling) in CFU-C obtained from patients in blast crisis. However, stem cell characteristics of these populations have been tested only by in vitro replating assays.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16709930 | 2006 | Evolving concepts in the management of chronic myeloid leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. | Baccarani M et al |
| 12829601 | 2003 | p210 BCR/ABL kinase regulates nucleotide excision repair (NER) and resistance to UV radiation. | Canitrot Y et al |
| 9585502 | 1998 | Oncogenic Abl and Src tyrosine kinases elicit the ubiquitin-dependent degradation of target proteins through a Ras-independent pathway. | Dai Z et al |
| 11264175 | 2001 | BCR-ABL down-regulates the DNA repair protein DNA-PKcs. | Deutsch E et al |
| 12576338 | 2003 | Down-regulation of BRCA1 in BCR-ABL-expressing hematopoietic cells. | Deutsch E et al |
| 17151364 | 2006 | Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. | Druker BJ et al |
| 15990860 | 2005 | Induction of centrosome and chromosome aberrations by imatinib in vitro. | Fabarius A et al |
| 15993098 | 2005 | Osteopontin is upregulated by BCR-ABL. | Flamant S et al |
| 15902293 | 2005 | Occurrence of de novo ABL kinase domain mutations in primary bone marrow cells after BCR-ABL gene transfer and Imatinib mesylate selection. | Flamant S et al |
| 15805265 | 2005 | p210BCR-ABL inhibits SDF-1 chemotactic response via alteration of CXCR4 signaling and down-regulation of CXCR4 expression. | Geay JF et al |
| 11756187 | 2002 | Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. | Graham SM et al |
| 9227927 | 1997 | Interferon alfa-2b combined with cytarabine versus interferon alone in chronic myelogenous leukemia. French Chronic Myeloid Leukemia Study Group. | Guilhot F et al |
| 17662883 | 2007 | Chronic myeloid leukaemia. | Hehlmann R et al |
| 2227945 | 1990 | Complex translocations, simple variant translocations and Ph-negative cases in chronic myelogenous leukaemia. | Huret JL et al |
| 15306667 | 2004 | Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. | Jamieson CH et al |
| 17330101 | 2007 | Chronic myeloid leukemia stem cells possess multiple unique features of resistance to BCR-ABL targeted therapies. | Jiang X et al |
| 16527898 | 2006 | BCR/ABL kinase induces self-mutagenesis via reactive oxygen species to encode imatinib resistance. | Koptyra M et al |
| 16809614 | 2006 | Myelodysplastic syndromes and acute leukemia developing after imatinib mesylate therapy for chronic myeloid leukemia. | Kovitz C et al |
| 16998483 | 2006 | Requirement for CD44 in homing and engraftment of BCR-ABL-expressing leukemic stem cells. | Krause DS et al |
| 1931706 | 1991 | The molecular pathology of chronic myelogenous leukaemia. | Kurzrock R et al |
| 9566916 | 1998 | Protein tyrosine phosphatase 1B antagonizes signalling by oncoprotein tyrosine kinase p210 bcr-abl in vivo. | LaMontagne KR Jr et al |
| 17717597 | 2007 | FTY720, a new alternative for treating blast crisis chronic myelogenous leukemia and Philadelphia chromosome-positive acute lymphocytic leukemia. | Neviani P et al |
| 12637609 | 2003 | Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. | O'Brien SG et al |
| 17363515 | 2007 | From mRNA metabolism to cancer therapy: chronic myelogenous leukemia shows the way. | Perrotti D et al |
| 18385684 | 2008 | Getting to the stem of chronic myeloid leukaemia. | Savona M et al |
| 11684015 | 2001 | BCR/ABL regulates mammalian RecA homologs, resulting in drug resistance. | Slupianek A et al |
| 3164637 | 1988 | Prognostic significance of additional cytogenetic abnormalities at diagnosis of Philadelphia chromosome-positive chronic granulocytic leukemia. | Sokal JE et al |
| 17996650 | 2007 | Rac guanosine triphosphatases represent integrating molecular therapeutic targets for BCR-ABL-induced myeloproliferative disease. | Thomas EK et al |
Citation
Ali G Turhan
Chronic myelogenous leukaemia (CML)
Atlas Genet Cytogenet Oncol Haematol. 2008-08-01
Online version: http://atlasgeneticsoncology.org/haematological/1117/chronic-myelogenous-leukaemia-(cml)
Historical Card
2000-10-01 Chronic myelogenous leukaemia (CML) by Ali G Turhan Affiliation
Pole de Biologie-Sante - 40 avenue du Recteur Pineau - 86022 Poitiers Cedex, France
1997-12-01 Chronic myelogenous leukaemia (CML) by Jean-Loup Huret Affiliation
Genetics, Dept Medical Information, University of Poitiers, CHU Poitiers Hospital, F-86021 Poitiers, France
