DNA/RNA
Note
Transcription
Several transcript variants encoding different FAK isoforms have been found for the PTK2 gene. The full-length nature of the following three has been determined.
(Sources: http://useast.ensembl.org/index.html; http://www.ncbi.nlm.nih.gov/pubmed/)
- Variant 1 differs in the 5 UTR and coding sequence compared to variant 2. The resulting isoform (a) is shorter at the N-terminus compared to isoform (b).
4414 bp - 33 exons - 1065 aa.
Ave. residue weight: 113.521.
Charge: 3.5.
Isoelectric point: 6.7311.
Molecular weight: 120899.54.
Number of residues: 1065.
- Variant 2 encodes the longest isoform (b).
4286 bp - 31 exons - 1073 aa.
Ave. residue weight: 113.366.
Charge: 2.0.
Isoelectric point: 6.6317.
Molecular weight: 121641.45.
Number of residues: 1073.
- Variant 3 differs in the 5 UTR and coding sequence, and contains two additional in-frame segments near the 3 end of the coding sequence, compared to variant 2. The resulting isoform (c) is shorter at the N-terminus and contains two additional segments in the C-terminus compared to isoform b.
Proteins
Note
Description
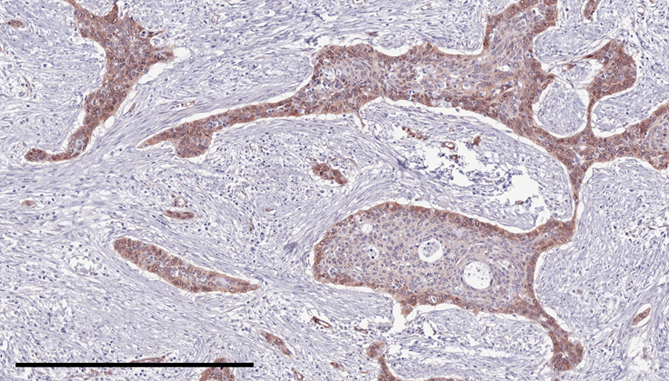
Expression
Localisation
Function
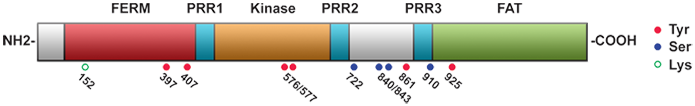
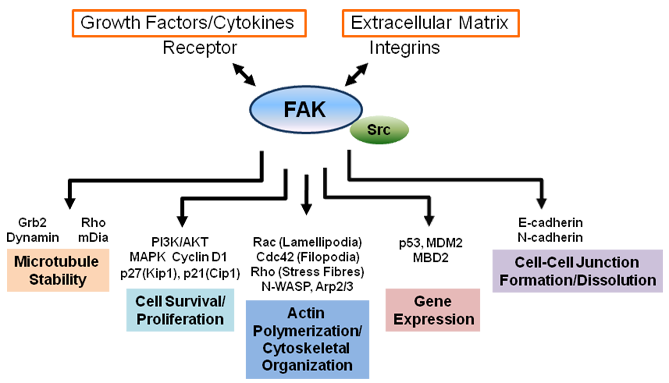
Integrin engagement with the extracellular matrix results in integrin clustering and a sequence of inter- and intramolecular events that permit the autophosphorylation of FAK at Tyr397 (Dunty et al., 2004). Subsequent recruitment of Src-family kinases through SH2-domain binding is followed by a mutual activation of both kinases. In the case of FAK this further activation is accomplished by phosphorylation of other tyrosine residues, specifically Tyr407, 576, 577, 861 and 925. Phosphorylation of Tyr576 and 577 increases FAK kinase activity whereas the remaining tyrosine residues serve as docking sites for SH2-containing factors such as Grb2 which links FAK into the MAPK pathway. Tyr397 also constitutes a docking site for the p85 subunit of PI3K (Chen and Guan, 1994) and phospholipase C gamma (Zhang et al., 1999). The PRRs are sites of interaction with SH3-containing factors which transmit signals downstream of the kinase and regulate the activity of Rho-family GTPases in charge of cell motility through the formation of stress fibres (RhoA), lamellipodia (Rac) and filopodia (Cdc42). Crk-associated substrate (p130Cas), initially identified in a two-hybrid screen, is one of the main downstream factors that bind to the PRRs of FAK (Polte and Hanks, 1995). Signaling via p130Cas towards Crk, DOCK180 and Rac has been linked to membrane ruffling, lamellipodia formation and cell motility (Harte et al., 1996; Cho and Klemke, 2002). A second essential downstream target of FAK is paxillin (Bellis et al., 1995), an adaptor protein lacking intrinsic kinase activity which can be phosphorylated at two sites, Tyr31 and Tyr118, and binds FAK within the C-terminal focal adhesion targeting (FAT) region (Hayashi et al., 2002). Mutations in FAK that disrupt binding to paxillin affect the localization of the kinase to focal contacts. Paxillin may also be involved in regulation of MAPK downstream signaling due to an overlapping binding site with Grb2 which binds to Tyr925 within the FAT region (Liu et al., 2002).
More recent studies link FAK to the regulation of cell-cell contacts, microtubule stability and control of gene transcription. Conflicting results have been reported from different experimental systems implicating FAK either with the dissolution (Avizienyte et al., 2002; Cicchini et al., 2008) or the promotion (Yano et al., 2004; Playford et al., 2008) of cell-cell contacts which suggests a dependency of this feature on the specific cellular context. Ezratty et al. (2005) reported on the role of FAK, Grb2 and dynamin in microtubule-induced focal adhesion disassembly. Earlier studies demonstrated an integrin-mediated activation of FAK at the leading edge of migrating cells as requirement for microtubule stabilization mediated by Rho and mDia (Palazzo et al., 2004). This mechanism also involves localization of a lipid raft marker, ganglioside GM1, to the leading edge. Xie et al. (2003) showed that Cdk5-mediated serine-phosphorylation of FAK was linked to the localization of the kinase at microtubule fork structures which contribute to nuclear repositioning in migrating neuronal cells. Recently, serine-phosphorylated FAK was shown to co-localize with centrosomes in mitotic endothelial cells. In this study by Park et al. (2009), FAK was also found associated with cytoplasmic dynein, and deletion of FAK resulted in mitotic defects. In 2005, Golubovskaya et al. reported results indicating a physical interaction between the N-terminal fragment of FAK and the N-terminal transactivation domain of p53. This interaction led to suppression of p53-mediated apoptosis and inhibition of the transcriptional activity of p53. Lim et al. (2008) subsequently provided data demonstrating a scaffolding role of nuclear FAK for MDM2-mediated p53 degradation mediated by the different lobes of the FERM domain. Also, basic sequences in the F2 lobe of the FERM domain were implicated in the nuclear localization of FAK (Lim et al., 2008), but alternative mechanisms independent from this putative nuclear localization signal are thought to exist (Schaller, 2010). Another nuclear function was recently uncovered by Luo et al. (2009) who described a role of FAK in chromatin remodelling via its interaction with MBD2 leading to increased myogenin expression and muscle-terminal differentiation. Liu et al. (2004), Li et al. (2004) and Ren et al. (2004) reported an involvement of FAK in netrin-1 signaling downstream of the netrin receptor DCC with consequences for axonal outgrowth and guidance in the developing brain.
- Mouse Models
Several mouse models have been generated to elucidate the functions of FAK both during normal development and neoplasia. A role of FAK in embryonal development was first observed in fak-/- mice which displayed defects in mesoderm development and anterior-posterior axis formation with embryonic lethality around day E8.5 (Ilic et al., 1995).
A conditional knockout model using a Cre-loxP system with Cre recombinase under the control of the nkx2-5 promoter was generated by Hakim et al. (2007). The major abnormality reported from this study was a profound disturbance of the development of the cardiac outflow tract. Knockout mice from this study died shortly after birth and displayed a range of cardiac defects which resemble the human congenital heart defects Tetralogy of Fallot and persistent truncus arteriosus. Peng et al. (2006) and DiMichele et al. (2006) reported results obtained with conditional knockout mice which carried Cre-recombinase under the control of the myosin light chain 2v promoter. Peng et al. (2006) found that knockout mice developed eccentric cardiac hypertrophy upon stimulation with angiotensin II or pressure overload. In contrast, the results by DiMichele et al. (2006) suggest that FAK functions to promote cardiac hypertrophy. In a later study by Peng at al. (2008) with myosin light chain-2a Cre mice they observed cardiac developmental abnormalities with thin ventricular walls and ventricular septal defects in the knockout mice, the majority of which died in the embryonic stage. Endothelial cell-specific knockout of FAK, again using a Cre-loxP approach, has been reported by Shen et al. (2005) and Braren et al. (2006). The observed phenotypes in knockout mice from both studies strongly suggest a role of FAK in vascular morphogenesis, particularly vascular remodelling and sprouting angiogenesis. The roles of FAK in the cardiovascular system were reviewed by Vadali et al. (2007).
A series of mouse models suggest an essential role of FAK during the development of the central nervous system. Beggs et al. (2003) created dorsal forebrain-specific conditional knockout mice using the Cre/loxP approach and observed an essential function of FAK for the formation of a normal basal lamina at the interface between radial glial end-feet and meningeal fibroblasts. They noted that the cortical changes seen in their study resembled lissencephaly phenotypes seen in some forms of human congenital muscular dystrophy.
Van Miltenburg et al. (2009) investigated the role of FAK in normal mammary gland using a conditional FAK-knockout mammary epithelial cell transplantation model based on FAK(lox/lox)/Rosa26Cre-ERT2 donor mice with loss of FAK in all mammary cells. They observed an abnormal mammary duct development with a disruption of myoepithelial and luminal epithelial cell layer and aberrant ductal morphogenesis during pregnancy.
Comprehensive reviews focused on the cellular functions of FAK have been published by Mitra et al. (2005) and Schaller (2010). Figure 3 schematically summarizes some of the diverse cellular functions of FAK.
- Regulation
The level of FAK expression is negatively and positively regulated by several transcription factors including p53, NF-kB and N-Myc (Golubovskaya et al., 2004; Beierle et al., 2007). Aside from the tyrosine residues implicated with FAK activation, at least four different serine phosphorylation sites (Ser722, 840, 843 and 910) have been recognized within FAK. Although the function of these serine sites has been examined less comprehensively, their phosphorylation has generally been associated with FAK inactivation, such as during mitosis (Ma et al., 2001), in suspension and under conditions that disturb the integrity of the actin cytoskeleton (Jacamo et al., 2007). FAK signaling is subject to additional levels of regulation which involve proteolytic cleavage (Dourdin et al., 2001), sumoylation (Kadaré et al., 2003), inhibition by FAK family interacting protein of 200 kDa (FIP200) (Abbi et al., 2002), dephosphorylation by protein-tyrosine phosphatases (Zeng et al., 2003), and generation of alternatively spliced isoforms such as FAK-related non-kinase (FRNK) (Schaller et al., 1993).
- Other protein family members: Pyk2.
Homology
| Homo sapiens PTK2 | % Identity for | Protein | DNA |
| vs. Pan troglodytes PTK2 | 99.8 | 99.7 | |
| vs. Canis lupus familiaris PTK2 | 97.0 | 91.7 | |
| vs. Mus musculus Ptk2 | 97.2 | 90.7 | |
| vs. Rattus norvegicus Ptk2 | 97.0 | 90.8 | |
| vs. Gallus gallus PTK2 | 94.9 | 83.9 | |
| vs. Danio rerio ptk2.1 | 83.2 | 74.2 | |
| vs. Drosophila melanogaster Fak56D | 42.9 | 48.9 | |
| vs. Caenorhabditis elegans kin-32 | 36.5 | 47.8 |
Implicated in
Beierle et al. (2007) reported on the relevance of FAK as cellular survival factor in N-myc-amplified neuroblastoma and identified N-myc binding sites in the FAK promoter. Recently, the same group also provided data indicating greater in vivo-therapeutic efficacy of pharmacologic FAK inhibition in N-myc-positive model systems (Beierle et al., 2010a; Beierle et al., 2010b). Efficacy of a novel small molecule dual IGF1R/FAK tyrosine kinase inhibitor (TAE226) leading to decreased FAK phosphorylation and cellular viability, cell cycle arrest and apoptosis has been described in human neuroblastoma cell lines (Beierle et al., 2008b). FAK expression was demonstrated in 51 of 70 clinical neuroblastoma samples by immunohistochemistry. FAK protein levels correlated with mRNA transcript levels and with advanced disease stage in this study (Beierle et al., 2008a).
FAK has been implicated in invasion and metastasis as well as chemoresistance in pancreatic cancer (Duxbury et al., 2003; Duxbury et al., 2004). FAK overexpression by immunohistochemistry, demonstrated in 24 of 50 (48%) patient samples, correlated with tumor size, but no other features including grade, lymph node involvement or metastasis in a study by Furuyama et al. (2006). Another study included an examination of both FAK and Src protein levels. FAK expression correlated significantly with tumor stage while Src expression correlated with both tumor stage and patient survival, and was identified as an independent prognostic factor by multivariate analysis (Chatzizacharias et al., 2010).
Hayashi et al. (2010) demonstrated high levels of cytoplasmic FAK expression in normal biliary epithelium and observed a gradual loss of staining from dysplasia to extra-hepatic bile duct carcinoma. In this study, positive FAK staining was associated with a significantly better survival. Increased levels of FAK mRNA and protein have also been observed in a study of 60 patients with hepatocellular cancers. Increased mRNA levels correlated with tumor size, serum AFP and inferior disease-free and overall survival (Fujii et al., 2004).
RNA interference studies in colorectal cancer cell line xenografts demonstrated that FAK inhibition resulted in inhibition of cell proliferation and angiogenesis, induction of apoptosis and tumor growth suppression (Lei et al., 2010). Elevated levels of FAK mRNA and protein were noted in a small cohort of 34 matched primary colon cancers and liver metastases (Lark et al., 2003). More recently a larger series of colorectal cancers with matched liver metastases was used to evaluate the correlation of FAK staining with clinical outcome. In this study, FAK staining was equivalent in primary and metastatic lesions, and elevated levels of FAK and Src were associated with a reduced time to recurrence (de Heer et al., 2008).
Immunohistochemical analysis of 134 cases of endometrial cancer demonstrated moderate to strong staining in the majority (89%) of cases. Weak FAK staining was noted in the remaining 11% and associated with a trend to improved survival. Increased FAK staining, however, correlated with measures of poor outcome including tumor grade, lymphovascular invasion and lymph node metastases (Gabriel et al., 2009). A different study demonstrated high levels of FAK expression in endometrial cancers of different histologies (endometrioid, serous and clear cell) as well as in regions of endometrial hyperplasia. The authors concluded that their data implicate FAK in endometrial carcinogenesis (Livasy et al., 2004).
An analysis of 166 surgical samples demonstrated cytoplasmic and membranous FAK staining in regions of cervical dysplasia and frankly invasive cancer of the uterine cervix with absent staining in adjacent normal cervical epithelium (Gabriel et al., 2006). One third of the patients, with tumors exhibiting weak FAK staining, had an inferior survival compared to those with moderate/strong FAK staining, and weak FAK staining correlated with lymph node positivity in this study. Oktay et al. (2003) demonstrated positive FAK staining in premalignant lesions. Similarly, Schwock et al. (2009) demonstrated an increase in FAK expression and concurrent decrease of E-cadherin in metastatic cervical cancer and carcinoma in-situ compared to normal cervical epithelium. An association between E-cadherin loss and FAK was also noted in an earlier study that included 26 carcinomas and 5 carcinoma in situ cases (Moon et al., 2003). Although FAK protein expression remained constant in this study, elevated levels of phosphorylated FAK were found in carcinoma samples.
McLean et al. (2004) demonstrated a role for FAK in the malignant progression from papilloma to squamous cell carcinoma in a transgenic mouse model combined with chemical carcinogenesis. No effect of the FAK deletion was noted on wound re-epithelialization.
Immunohistochemical analysis of normal and neoplastic hemato-lymphoid tissues demonstrated FAK staining in B cells of the germinal center, marginal and mantle zones (Ozkal et al., 2009). Corresponding staining was present in most B-cell lymphomas while T-cell lymphomas were predominantly negative. Neoplastic cells of classical Hodgkins lymphoma were negative for FAK while those of lymphocyte-predominant Hodgkins lymphoma were positive in the same study.
A study of 60 primary acute myeloid leukemia samples demonstrated FAK transcript and protein expression in 48% cases and Pyk2 expression in 81% cases (Recher et al., 2004). FAK-positive acute myeloid leukemia cells displayed a higher migratory efficiency and lower sensitivity to chemotherapy. FAK expression positively correlated with white blood count at diagnosis, death rate and median survival.
Chen et al. (2001) found that keratinocytes from patients with psoriasis have elevated levels of phosphorylated FAK and concluded that integrin/FAK signaling contributes to a pre-activation of uninvolved keratinocytes that predisposes to the development of psoriatic plaques in response to certain stimuli.
FAK has a role in the development of the cardiovascular system since FAK-null mice are embryonically lethal with phenotypic abnormalities approximating those seen in human congenital heart defects (Vadali et al., 2007). FAK also appears to be involved cardiac hypertrophy and heart failure through its involvement in the cardiac response to biochemical stress and hypertrophic agonists. The relevance of FAK to cardiac physiology likely differs with the cellular context. Although FAK activation has been suggested to accelerate function deterioration of an overloaded heart, selective FAK deletion in cardiomyoctes has also been associated with maladaptive cardiac remodeling (Franchini et al., 2009).
FAK appears to be essential for normal glucose transport and glycogen synthesis due to cross talk between integrin and insulin signaling pathways (Huang et al., 2002; Huang et al., 2006). FAK has also been implicated in hyperglycemia-related vascular complications in Diabetes mellitus (Mori et al., 2002). Two independent studies reported on increased levels of activated FAK in the glomeruli from diabetic rats that could be abrogated by insulin treatment (Clark et al., 1995; Shikano et al., 1996).
FAK signaling has been implicated with non-neoplastic renal disease. Holzapfel et al. (2007) documented a role for FAK during restoration of tubular integrity in renal ischemia and reperfusion, and an earlier study by Morino et al. (1999) indicated activated FAK-signaling during the development and progression of autoimmune-mediated nephritis in an animal model.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12221124 | 2002 | Regulation of focal adhesion kinase by a novel protein inhibitor FIP200. | Abbi S et al |
| 12110680 | 2002 | Focal adhesion kinase activated by beta(4) integrin ligation to mCLCA1 mediates early metastatic growth. | Abdel-Ghany M et al |
| 8422239 | 1993 | Expression of an N-terminally truncated form of human focal adhesion kinase in brain. | André E et al |
| 12134161 | 2002 | Src-induced de-regulation of E-cadherin in colon cancer cells requires integrin signalling. | Avizienyte E et al |
| 18348298 | 2008 | Dual focal adhesion kinase/Pyk2 inhibitor has positive effects on bone tumors: implications for bone metastases. | Bagi CM et al |
| 14642275 | 2003 | FAK deficiency in cells contributing to the basal lamina results in cortical abnormalities resembling congenital muscular dystrophies. | Beggs HE et al |
| 19885861 | 2010 | Inhibition of focal adhesion kinase and src increases detachment and apoptosis in human neuroblastoma cell lines. | Beierle EA et al |
| 7615549 | 1995 | Characterization of tyrosine phosphorylation of paxillin in vitro by focal adhesion kinase. | Bellis SL et al |
| 16391003 | 2006 | Endothelial FAK is essential for vascular network stability, cell survival, and lamellipodial formation. | Braren R et al |
| 18349846 | 2008 | Involvement of focal adhesion kinase in cellular invasion of head and neck squamous cell carcinomas via regulation of MMP-2 expression. | Canel M et al |
| 20431421 | 2010 | Evaluation of the clinical significance of focal adhesion kinase and SRC expression in human pancreatic ductal adenocarcinoma. | Chatzizacharias NA et al |
| 11886520 | 2001 | Basal keratinocytes from uninvolved psoriatic skin exhibit accelerated spreading and focal adhesion kinase responsiveness to fibronectin. | Chen G et al |
| 7937853 | 1994 | Association of focal adhesion kinase with its potential substrate phosphatidylinositol 3-kinase. | Chen HC et al |
| 11839772 | 2002 | Purification of pseudopodia from polarized cells reveals redistribution and activation of Rac through assembly of a CAS/Crk scaffold. | Cho SY et al |
| 17949712 | 2008 | TGFbeta-induced EMT requires focal adhesion kinase (FAK) signaling. | Cicchini C et al |
| 8690164 | 1995 | Increased phosphorylation of focal adhesion kinase in diabetic rat kidney glomeruli. | Clark S et al |
| 16902179 | 2006 | Myocyte-restricted focal adhesion kinase deletion attenuates pressure overload-induced hypertrophy. | DiMichele LA et al |
| 20621036 | 2010 | Expression of focal adhesion kinase and phosphorylated focal adhesion kinase in human gliomas is associated with unfavorable overall survival. | Ding L et al |
| 11602605 | 2001 | Reduced cell migration and disruption of the actin cytoskeleton in calpain-deficient embryonic fibroblasts. | Dourdin N et al |
| 15169899 | 2004 | FERM domain interaction promotes FAK signaling. | Dunty JM et al |
| 15118593 | 2004 | Focal adhesion kinase gene silencing promotes anoikis and suppresses metastasis of human pancreatic adenocarcinoma cells. | Duxbury MS et al |
| 15895076 | 2005 | Microtubule-induced focal adhesion disassembly is mediated by dynamin and focal adhesion kinase. | Ezratty EJ et al |
| 7766995 | 1995 | Mapping of the focal adhesion kinase (Fadk) gene to mouse chromosome 15 and human chromosome 8. | Fiedorek FT Jr et al |
| 19219296 | 2009 | Focal adhesion kinase signaling in cardiac hypertrophy and failure. | Franchini KG et al |
| 8707856 | 1996 | Control of adhesion-dependent cell survival by focal adhesion kinase. | Frisch SM et al |
| 15246215 | 2004 | Focal adhesion kinase is overexpressed in hepatocellular carcinoma and can be served as an independent prognostic factor. | Fujii T et al |
| 16425085 | 2006 | Clinical significance of focal adhesion kinase in resectable pancreatic cancer. | Furuyama K et al |
| 19823058 | 2009 | Expression of focal adhesion kinase in patients with endometrial cancer: a clinicopathologic study. | Gabriel B et al |
| 16638855 | 2006 | Weak expression of focal adhesion kinase (pp125FAK) in patients with cervical cancer is associated with poor disease outcome. | Gabriel B et al |
| 18987997 | 2009 | Expression and clinical significance of focal adhesion kinase in the two distinct histological types, intestinal and diffuse, of human gastric adenocarcinoma. | Giaginis CT et al |
| 19521985 | 2009 | FAK overexpression and p53 mutations are highly correlated in human breast cancer. | Golubovskaya VM et al |
| 15455382 | 2005 | Differential expression of protease activated receptor 1 (Par1) and pY397FAK in benign and malignant human ovarian tissue samples. | Grisaru-Granovsky S et al |
| 1379699 | 1992 | Regulation of focal adhesion-associated protein tyrosine kinase by both cellular adhesion and oncogenic transformation. | Guan JL et al |
| 17526730 | 2007 | Conditional deletion of focal adhesion kinase leads to defects in ventricular septation and outflow tract alignment. | Hakim ZS et al |
| 16914580 | 2006 | Focal adhesion kinase targeting using in vivo short interfering RNA delivery in neutral liposomes for ovarian carcinoma therapy. | Halder J et al |
| 16361572 | 2005 | Focal adhesion kinase silencing augments docetaxel-mediated apoptosis in ovarian cancer cells. | Halder J et al |
| 18006843 | 2007 | Therapeutic efficacy of a novel focal adhesion kinase inhibitor TAE226 in ovarian carcinoma. | Halder J et al |
| 16320111 | 2005 | Focal adhesion kinase is activated in invading fibrosarcoma cells and regulates metastasis. | Hanada M et al |
| 1528852 | 1992 | Focal adhesion protein-tyrosine kinase phosphorylated in response to cell attachment to fibronectin. | Hanks SK et al |
| 8662921 | 1996 | p130Cas, a substrate associated with v-Src and v-Crk, localizes to focal adhesions and binds to focal adhesion kinase. | Harte MT et al |
| 12456636 | 2002 | FRNK blocks v-Src-stimulated invasion and experimental metastases without effects on cell motility or growth. | Hauck CR et al |
| 20185162 | 2010 | Decreased expression of focal adhesion kinase is associated with a poor prognosis in extrahepatic bile duct carcinoma. | Hayashi A et al |
| 11799401 | 2002 | The focal adhesion targeting (FAT) region of focal adhesion kinase is a four-helix bundle that binds paxillin. | Hayashi I et al |
| 19729215 | 2009 | 3D cell cultures of human head and neck squamous cell carcinoma cells are radiosensitized by the focal adhesion kinase inhibitor TAE226. | Hehlgans S et al |
| 16552181 | 2006 | Focal adhesion kinase signaling and the aggressive melanoma phenotype. | Hess AR et al |
| 16267008 | 2005 | Focal adhesion kinase promotes the aggressive melanoma phenotype. | Hess AR et al |
| 21321210 | 2011 | Endothelial focal adhesion kinase mediates cancer cell homing to discrete regions of the lungs via E-selectin up-regulation. | Hiratsuka S et al |
| 17549512 | 2007 | Role of focal adhesion kinase (FAK) in renal ischaemia and reperfusion. | Holzapfel K et al |
| 18974065 | 2008 | Apigenin inhibited migration and invasion of human ovarian cancer A2780 cells through focal adhesion kinase. | Hu XW et al |
| 11809746 | 2002 | Focal adhesion kinase (FAK) regulates insulin-stimulated glycogen synthesis in hepatocytes. | Huang D et al |
| 16574795 | 2006 | Reduced expression of focal adhesion kinase disrupts insulin action in skeletal muscle cells. | Huang D et al |
| 7566154 | 1995 | Reduced cell motility and enhanced focal adhesion contact formation in cells from FAK-deficient mice. | Ilić D et al |
| 18303050 | 2008 | FAK, PDZ-RhoGEF and ROCKII cooperate to regulate adhesion movement and trailing-edge retraction in fibroblasts. | Iwanicki MP et al |
| 17096371 | 2007 | FAK phosphorylation at Ser-843 inhibits Tyr-397 phosphorylation, cell spreading and migration. | Jacamo R et al |
| 9023046 | 1996 | Integrin expression on cell adhesion function and up-regulation of P125FAK and paxillin in metastatic renal carcinoma cells. | Jenq W et al |
| 18922979 | 2008 | Focal adhesion kinase controls aggressive phenotype of androgen-independent prostate cancer. | Johnson TR et al |
| 14500712 | 2003 | PIAS1-mediated sumoylation of focal adhesion kinase activates its autophosphorylation. | Kadaré G et al |
| 18606490 | 2008 | Mutation of Y925F in focal adhesion kinase (FAK) suppresses melanoma cell proliferation and metastasis. | Kaneda T et al |
| 2110361 | 1990 | Monoclonal antibodies to individual tyrosine-phosphorylated protein substrates of oncogene-encoded tyrosine kinases. | Kanner SB et al |
| 15483349 | 2004 | Increased expression of focal adhesion kinase in thyroid cancer: immunohistochemical study. | Kim SJ et al |
| 18941742 | 2009 | Focal adhesion kinase as an immunotherapeutic target. | Kobayashi H et al |
| 18056629 | 2007 | Mammary epithelial-specific disruption of the focal adhesion kinase blocks mammary tumor progression. | Lahlou H et al |
| 15861214 | 2005 | High focal adhesion kinase expression in invasive breast carcinomas is associated with an aggressive phenotype. | Lark AL et al |
| 21159635 | 2010 | Conditional deletion of the focal adhesion kinase FAK alters remodeling of the blood-brain barrier in glioma. | Lee J et al |
| 21179944 | 2010 | RNA interference-mediated silencing of focal adhesion kinase inhibits growth of human colon carcinoma xenograft in nude mice. | Lei K et al |
| 15494734 | 2004 | Activation of FAK and Src are receptor-proximal events required for netrin signaling. | Li W et al |
| 15564794 | 2004 | Upregulation of focal adhesion kinase (FAK) expression in ductal carcinoma in situ (DCIS) is an early event in breast tumorigenesis. | Lightfoot HM Jr et al |
| 18206965 | 2008 | Nuclear FAK promotes cell proliferation and survival through FERM-enhanced p53 degradation. | Lim ST et al |
| 18648907 | 2008 | Extended survival of Pyk2 or FAK deficient orthotopic glioma xenografts. | Lipinski CA et al |
| 15494732 | 2004 | Netrin requires focal adhesion kinase and Src family kinases for axon outgrowth and attraction. | Liu G et al |
| 11909967 | 2002 | Structural insight into the mechanisms of targeting and signaling of focal adhesion kinase. | Liu G et al |
| 15536334 | 2004 | Focal adhesion kinase overexpression in endometrial neoplasia. | Livasy CA et al |
| 19147559 | 2009 | Mammary epithelial-specific ablation of the focal adhesion kinase suppresses mammary tumorigenesis by affecting mammary cancer stem/progenitor cells. | Luo M et al |
| 11160818 | 2001 | Serine phosphorylation of focal adhesion kinase in interphase and mitosis: a possible role in modulating binding to p130(Cas). | Ma A et al |
| 18835089 | 2009 | Inhibition of T24 human bladder carcinoma cell migration by RNA interference suppressing the expression of HD-PTP. | Mariotti M et al |
| 15601818 | 2004 | Specific deletion of focal adhesion kinase suppresses tumor formation and blocks malignant progression. | McLean GW et al |
| 20405349 | 2010 | Evaluation of FAK and Src expression in human benign and malignant thyroid lesions. | Michailidi C et al |
| 21119598 | 2011 | Ligand-independent activation of c-Met by fibronectin and α(5)β(1)-integrin regulates ovarian cancer invasion and metastasis. | Mitra AK et al |
| 15688067 | 2005 | Focal adhesion kinase: in command and control of cell motility. | Mitra SK et al |
| 16682956 | 2006 | Intrinsic FAK activity and Y925 phosphorylation facilitate an angiogenic switch in tumors. | Mitra SK et al |
| 14675348 | 2003 | The expression and tyrosine phosphorylation of E-cadherin/catenin adhesion complex, and focal adhesion kinase in invasive cervical carcinomas. | Moon HS et al |
| 11872370 | 2002 | Hyperglycemia-induced alteration of vascular smooth muscle phenotype. | Mori S et al |
| 10457217 | 1999 | Glomerular overexpression and increased tyrosine phosphorylation of focal adhesion kinase p125FAK in lupus-prone MRL/MP-lpr/lpr mice. | Morino N et al |
| 20802517 | 2010 | DNA copy number aberrations in small-cell lung cancer reveal activation of the focal adhesion pathway. | Ocak S et al |
| 12673558 | 2003 | Focal adhesion kinase as a marker of malignant phenotype in breast and cervical carcinomas. | Oktay MH et al |
| 18070912 | 2007 | Regulation of lamellipodial persistence, adhesion turnover, and motility in macrophages by focal adhesion kinase. | Owen KA et al |
| 7796399 | 1995 | Overexpression of the focal adhesion kinase (p125FAK) in invasive human tumors. | Owens LV et al |
| 19647948 | 2009 | Focal adhesion kinase (FAK) expression in normal and neoplastic lymphoid tissues. | Ozkal S et al |
| 14764879 | 2004 | Localized stabilization of microtubules by integrin- and FAK-facilitated Rho signaling. | Palazzo AF et al |
| 19201755 | 2009 | Role of focal adhesion kinase Ser-732 phosphorylation in centrosome function during mitosis. | Park AY et al |
| 20869748 | 2010 | Focal adhesion kinase (FAK) gene amplification and its clinical implications in gastric cancer. | Park JH et al |
| 16374517 | 2006 | Inactivation of focal adhesion kinase in cardiomyocytes promotes eccentric cardiac hypertrophy and fibrosis in mice. | Peng X et al |
| 18448675 | 2008 | Cardiac developmental defects and eccentric right ventricular hypertrophy in cardiomyocyte focal adhesion kinase (FAK) conditional knockout mice. | Peng X et al |
| 18773890 | 2008 | Focal adhesion kinase regulates cell-cell contact formation in epithelial cells via modulation of Rho. | Playford MP et al |
| 7479864 | 1995 | Interaction between focal adhesion kinase and Crk-associated tyrosine kinase substrate p130Cas. | Polte TR et al |
| 20697346 | 2010 | MUC4 mucin-induced epithelial to mesenchymal transition: a novel mechanism for metastasis of human ovarian cancer cells. | Ponnusamy MP et al |
| 18845837 | 2008 | Mammary epithelial-specific disruption of focal adhesion kinase retards tumor formation and metastasis in a transgenic mouse model of human breast cancer. | Provenzano PP et al |
| 19147981 | 2009 | Ras- and PI3K-dependent breast tumorigenesis in mice and humans requires focal adhesion kinase signaling. | Pylayeva Y et al |
| 15126359 | 2004 | Expression of focal adhesion kinase in acute myeloid leukemia is associated with enhanced blast migration, increased cellularity, and poor prognosis. | Recher C et al |
| 15494733 | 2004 | Focal adhesion kinase in netrin-1 signaling. | Ren XR et al |
| 12242727 | 2002 | Expression of focal adhesion kinase in normal and pathologic human prostate tissues. | Rovin JD et al |
| 8423801 | 1993 | Autonomous expression of a noncatalytic domain of the focal adhesion-associated protein tyrosine kinase pp125FAK. | Schaller MD et al |
| 20332118 | 2010 | Cellular functions of FAK kinases: insight into molecular mechanisms and novel functions. | Schaller MD et al |
| 15743500 | 2005 | High expression of focal adhesion kinase (p125FAK) in node-negative breast cancer is related to overexpression of HER-2/neu and activated Akt kinase but does not predict outcome. | Schmitz KJ et al |
| 19458065 | 2009 | Targeting focal adhesion kinase with dominant-negative FRNK or Hsp90 inhibitor 17-DMAG suppresses tumor growth and metastasis of SiHa cervical xenografts. | Schwock J et al |
| 17963503 | 2007 | Differential expression of the FAK family kinases in rheumatoid arthritis and osteoarthritis synovial tissues. | Shahrara S et al |
| 15967814 | 2005 | Conditional knockout of focal adhesion kinase in endothelial cells reveals its role in angiogenesis and vascular development in late embryogenesis. | Shen TL et al |
| 17219439 | 2007 | A novel low-molecular weight inhibitor of focal adhesion kinase, TAE226, inhibits glioma growth. | Shi Q et al |
| 8717307 | 1996 | [Tyrosine phosphorylation of focal adhesion kinase (p125FAK) and paxillin in glomeruli from diabetic rats]. | Shikano T et al |
| 10806474 | 2000 | FAK integrates growth-factor and integrin signals to promote cell migration. | Sieg DJ et al |
| 19351861 | 2009 | Alphavbeta3/alphavbeta5 integrins-FAK-RhoB: a novel pathway for hypoxia regulation in glioblastoma. | Skuli N et al |
| 17724718 | 2007 | Osteonectin downregulates E-cadherin, induces osteopontin and focal adhesion kinase activity stimulating an invasive melanoma phenotype. | Smit DJ et al |
| 16179862 | 2005 | Effect of focal adhesion kinase (FAK) downregulation with FAK antisense oligonucleotides and 5-fluorouracil on the viability of melanoma cell lines. | Smith CS et al |
| 10821872 | 2000 | Anti-apoptotic role of focal adhesion kinase (FAK). Induction of inhibitor-of-apoptosis proteins and apoptosis suppression by the overexpression of FAK in a human leukemic cell line, HL-60. | Sonoda Y et al |
| 20389021 | 2010 | Adrenergic modulation of focal adhesion kinase protects human ovarian cancer cells from anoikis. | Sood AK et al |
| 15466376 | 2004 | Biological significance of focal adhesion kinase in ovarian cancer: role in migration and invasion. | Sood AK et al |
| 19482420 | 2009 | Doxycycline inhibits the adhesion and migration of melanoma cells by inhibiting the expression and phosphorylation of focal adhesion kinase (FAK). | Sun T et al |
| 15914540 | 2005 | Focal adhesion kinase is required for the spatial organization of the leading edge in migrating cells. | Tilghman RW et al |
| 8900422 | 1996 | Focal adhesion kinase (pp125FAK) expression, activation and association with paxillin and p50CSK in human metastatic prostate carcinoma. | Tremblay L et al |
| 20709760 | 2010 | CAV1 inhibits metastatic potential in melanomas through suppression of the integrin/Src/FAK signaling pathway. | Trimmer C et al |
| 17968709 | 2007 | Focal adhesion kinase: an essential kinase in the regulation of cardiovascular functions. | Vadali K et al |
| 16387351 | 2006 | Acquisition of chemoresistance following discontinuous exposures to cisplatin is associated in ovarian carcinoma cells with progressive alteration of FAK, ERK and p38 activation in response to treatment. | Villedieu M et al |
| 19242756 | 2009 | Expression of FAK and PTEN in bronchioloalveolar carcinoma and lung adenocarcinoma. | Wang C et al |
| 21247411 | 2011 | RNA interference-mediated silencing of focal adhesion kinase inhibits growth of human malignant glioma xenograft in nude mice. | Wang GJ et al |
| 7834423 | 1994 | Expression of growth factor receptors, the focal adhesion kinase, and other tyrosine kinases in human soft tissue tumors. | Weiner TM et al |
| 12941275 | 2003 | Serine 732 phosphorylation of FAK by Cdk5 is important for microtubule organization, nuclear movement, and neuronal migration. | Xie Z et al |
| 18852147 | 2008 | Heparin-binding epidermal growth factor-like growth factor promotes transcoelomic metastasis in ovarian cancer through epithelial-mesenchymal transition. | Yagi H et al |
| 18021457 | 2007 | Inhibition of cell motility by troglitazone in human ovarian carcinoma cell line. | Yang YC et al |
| 15263022 | 2004 | Roles played by a subset of integrin signaling molecules in cadherin-based cell-cell adhesion. | Yano H et al |
| 20814816 | 2011 | Clinical significance of high focal adhesion kinase gene copy number and overexpression in invasive breast cancer. | Yom CK et al |
| 20872237 | 2010 | Mesenchymal mode of migration participates in pulmonary metastasis of mouse osteosarcoma LM8. | Yui Y et al |
| 12515828 | 2003 | PTP alpha regulates integrin-stimulated FAK autophosphorylation and cytoskeletal rearrangement in cell spreading and migration. | Zeng L et al |
| 10430888 | 1999 | Focal adhesion kinase promotes phospholipase C-gamma1 activity. | Zhang X et al |
| 10197643 | 1999 | Prostatic carcinoma cell migration via alpha(v)beta3 integrin is modulated by a focal adhesion kinase pathway. | Zheng DQ et al |
| 18556171 | 2008 | Combined expression of the non-receptor protein tyrosine kinases FAK and Src in primary colorectal cancer is associated with tumor recurrence and metastasis formation. | de Heer P et al |
| 19584305 | 2009 | Complete focal adhesion kinase deficiency in the mammary gland causes ductal dilation and aberrant branching morphogenesis through defects in Rho kinase-dependent cell contractility. | van Miltenburg MH et al |
Other Information
Locus ID:
NCBI: 5747
MIM: 600758
HGNC: 9611
Ensembl: ENSG00000169398
Variants:
dbSNP: 5747
ClinVar: 5747
TCGA: ENSG00000169398
COSMIC: PTK2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37878047 | 2024 | HuR-induced circ_0082319 contributes to hepatocellular carcinoma by elevating PTK2 through miR-505-3p. | 1 |
| 37950040 | 2024 | VPS35 promotes gastric cancer progression through integrin/FAK/SRC signalling-mediated IL-6/STAT3 pathway activation in a YAP-dependent manner. | 0 |
| 38184221 | 2024 | DPP-4 exacerbates LPS-induced endothelial cells inflammation via integrin-α5β1/FAK/AKT signaling. | 0 |
| 38233791 | 2024 | ZDHHC5-mediated S-palmitoylation of FAK promotes its membrane localization and epithelial-mesenchymal transition in glioma. | 2 |
| 38269431 | 2024 | Exploring the therapeutic potential of focal adhesion kinase inhibition in overcoming chemoresistance in pancreatic ductal adenocarcinoma. | 0 |
| 38281617 | 2024 | ING3 inhibits the malignant progression of lung adenocarcinoma by negatively regulating ITGB4 expression to inactivate Src/FAK signaling. | 0 |
| 38326036 | 2024 | ICAM-1 Deletion Using CRISPR/Cas9 Protects the Brain from Traumatic Brain Injury-Induced Inflammatory Leukocyte Adhesion and Transmigration Cascades by Attenuating the Paxillin/FAK-Dependent Rho GTPase Pathway. | 0 |
| 38396816 | 2024 | THSD1 Suppresses Autophagy-Mediated Focal Adhesion Turnover by Modulating the FAK-Beclin 1 Pathway. | 0 |
| 38488492 | 2024 | FAK mediates hypoxia-induced pulmonary artery smooth muscle cell proliferation by modulating mitochondrial transcription termination factor 1/cyclin D1. | 0 |
| 38532364 | 2024 | A decrease in integrin α5β1/FAK is associated with increased apoptosis of aortic smooth muscle cells in acute type a aortic dissection. | 0 |
| 38648767 | 2024 | Effect of Focal Adhesion Kinase and Vinculin Expression on Migration Parameters of Normal and Tumor Epitheliocytes. | 0 |
| 38725858 | 2024 | The dysadherin/FAK axis promotes individual cell migration in colon cancer. | 0 |
| 38824643 | 2024 | FAK regulates tension transmission to the nucleus and endothelial transcriptome independent of kinase activity. | 0 |
| 38835117 | 2024 | T-plastin contributes to epithelial-mesenchymal transition in human lung cancer cells through FAK/AKT/Slug axis signaling pathway. | 0 |
| 37878047 | 2024 | HuR-induced circ_0082319 contributes to hepatocellular carcinoma by elevating PTK2 through miR-505-3p. | 1 |
Citation
Joerg Schwock ; Neesha Dhani
PTK2 (PTK2 protein tyrosine kinase 2)
Atlas Genet Cytogenet Oncol Haematol. 2011-03-01
Online version: http://atlasgeneticsoncology.org/gene/41898/ptk2-(ptk2-protein-tyrosine-kinase-2)
