Micronuclei : Pitfalls and Problems
John R K Savage
(34 city Road, Tilehurst, Reading, RG31 5HB, UK)
July 2000
Introduction
Ionizing radiation and numerous chemical mutagens cause structural chromosomal aberrations, many of which are visible at the light-microscope level. There are many types, and a complicated classification (Savage, 1976), so that specialised knowledge and training are required for reliable scoring. These aberrations form the basis of a large amount of radiobiological and DNA-repair theory and have many practical uses in the fields of biological dosimetry, clinical cytogenetics and environmental monitoring (Heddle, 1990; Streffer et al., 1998).
A proportion of the aberrations (usually referred to as "Asymmetrical events" or "Unstable aberrations" (Carrano and Heddle, 1973; Savage, 1976)) give rise to chromosome fragments without spindle attachment organelles (kinetochores, centromeres). These are termed "acentric fragments", (AF). When the cell divides, some of these fragments are excluded from the main daughter nuclei and form small extra nuclei within the cytoplasm, either on their own, or in conjunction with other fragments. Such "micronuclei" (MN) can appear in the cytoplasm of either, or both, daughter cells.
Depending on the origin of the excluded fragment, i.e. the type of aberration from which it was derived, both, or only one daughter cell will suffer genetic loss. In some cases, the aberrations lead to mechanical separation problems ("bridges") at anaphase as well as fragment loss. These events will ultimately kill the cell, though "death" (measured by cessation of division) may not occur until 2-4 divisions have taken place.
With time, therefore, when an acute radiation dose, or treatment with a very short-lived chemical clastogen has been given, MN production ultimately ceases, and the frequency in new cells returns to the control (un-treated) level.
It is logical to expect that there should be some numerical relationship between the number of AF per cell and the number of MN per cell derived from them. On the assumption that this relationship is fairly simple, MN scoring in interphase cells has been proposed, and used, as a quick and easy substitute for the more difficult and time-consuming metaphase aberration analysis ( Countryman and Heddle, 1976; Heddle, 1973; Heddle, 1975; Muller and Streffer, 1994).
In the context of mutagen screening, or when purely qualitative answers are required, this is to some extent valid, and meaningful results can be obtained (Heddle, 1990; Muller and Streffer, 1994). However, the MN system is full of pitfalls for the unwary and there are many factors which conspire to uncouple any simple relationship between AF and MN, making critical quantitative work very difficult.
The purpose of this paper is to highlight some of the MN-system problems, because, to appreciate them, will help in the design of meaningful experiments and applications, and in the interpretation of any results obtained.
Factors which can influence the AF Æ MN relationship
For convenience, I will group the factors that influence the observed frequencies of MN derived from a given frequency of AF under four broad headings:
- Production factors.
- Fragment-fate factors.
- Cell-kinetic factors.
- The time-displacement factor.
Production factors.
a) Fragment origin.
Following radiation, very few of the MN observed are derived from lagging whole chromosomes, most come from asymmetrical structural aberrations. In contrast, most of the spontaneously occurring ones appear to arise from whole chromosomes. This has been shown by the application of kinetochore-specific probes to micronuclei (Degrassi and Tanzarella, 1988; Fenech and Morley, 1989; Tucker and Eastmond, 1990).
There is only one type of MN but many different kinds of aberration can contribute to them. Figure 1 summarises the principal types of aberration which produce AF.
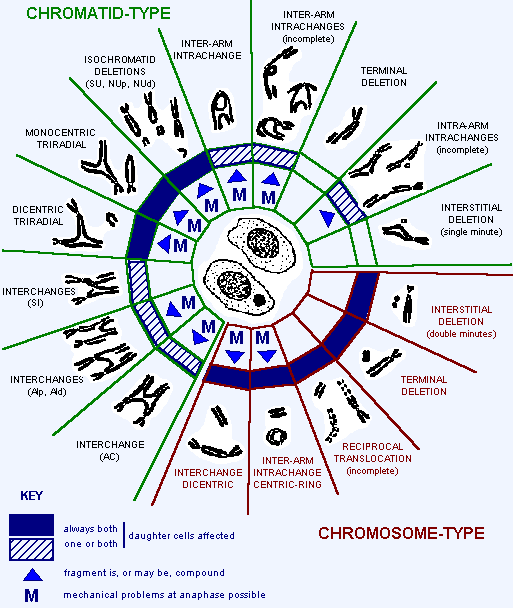
Figure 1 :
The principal structural chromosomal aberrations which contribute acentric fragments (AF) to form micronuclei (MN). The diagram also indicates those which produce compound fragments, i.e. composed of segments from more than one chromosome/chromatid and also those which produce mechanical separation problems ("bridges") at anaphase.
Fragment loss leads to genetic imbalance and ultimate cell death ; this may affect either one, or both daughter cells. Since there is frequently more than one aberration per cell, the probability of both daughters being affected is increased.
Which kinds predominate will depend upon the kind of cells used, the stage of the cell in the cycle when exposed to the clastogen, and the clastogen used. Ionizing radiation can produce all the types of structural change, but the vast majority of chemical clastogens produce, primarily, only chromatid-types.
Some of the fragments are compound, containing segments from more than one chromosome, others are simple. Thus, the extent of genetic loss varies considerably and may affect both daughters, or only one.
b) MN Generation.
Only AF excluded from the daughter nuclei at telophase can produce MN. Thus, cell division is a necessary condition for their appearance, and the MN frequency will be expected to increase with time after treatment as more and more aberration-bearing cells pass through mitosis. Exposure of a non- or very slowly dividing cell population, or a treatment that inhibits cell division, will mean that almost no MN are observed.
AF included in either daughter nucleus will duplicate along with the rest of the genome during S-phase and, if the cell divides again, will produce a second crop of MN, the duplication of the initial included fragment augmenting the observed frequency.
As mentioned above, neither fragment loss nor mechanical separation problems at anaphase necessarily kill the cell immediately (though the latter usually precludes further divisions) so quite a few AF-bearing cells divide 2 or 3 times before cessation leading to a continuous, but eventually declining, production of MN for some time after treatment.
In the majority of cell systems, both the absolute and the relative frequency of the various forms of primary aberrations vary as the cell transits the cycle. Consequently, the generation of excluded AF (and hence MN) will also fluctuate with time after treatment. In addition to this, we have to remember that with most clastogens, especially chemicals, a further crop of primary aberrations can arise in later divisions from unused long-lived lesions. These also will contribute AF for MN.
Fragment-fate factors.
a) The Inclusion probability, (PI).
The probability that an AF is included when the daughter nuclei re-form after division (Savage, 1988). The probability of exclusion, PE = (1.0 - PI ). When PI < 1.0, which appears to be the universal condition, then MN will form at successive divisions as outlined above. There is no recorded case of PI = 0.
A number of pertinent questions need to be asked, and answered, about PI :-
Is it a constant ? In some cases the answer is "No". It differs between cell type within species (fibroblasts versus lymphocytes, (Savage, 1988)), and between different species for the same cell type. Good information is lacking on effects of cell age, of karyotype composition, of fragment-size or of fragment number.
Is it affected by the clastogen used ?
Is it dependent upon the dose ? There is some evidence that fragment exclusion falls as the dose of ionizing radiation increases.
Finally, and importantly, is PI affected by any drug used to interfere with the cytoskeleton, and concomitant cytokinesis ? As, for example, cytochalasin-B (Fenech and Morley, 1985; Fenech and Morley, 1986), which is now almost universally used to overcome the generation/dilution problem discussed below.
b) Sister-fragment separation.
Chromosome-type (pre-replication) AF are always paired, whereas chromatid-type (post replication) AF are mixed, but predominantly single (Figure 1). It is assumed, especially for theoretical work, that all paired fragments retain their adherence, and are transmitted to MN as a unit pair (Braselmann et al., 1986; Carrano and Heddle, 1973; Wakata and Sasaki, 1987). Recent evidence from certain species indicates that this may not always be the case (Das and Sharma, 1987), so that in some systems, two MN may sometimes arise from one AF pair.
c) Fragment coalescence.
As the number of AF per cell increases, so also does the probability that one MN may contain several AF. Cytoplasmic currents around the spindle apparatus can lead to vortexes ("Sargasso Seas") where AF collect, enhancing this probability. Thus, at higher clastogen doses, any 1:1 AF:MN expectation breaks down. Several authors have noted the paucity of multi-MN cells and the tendency to under-dispersion of MN between-cell distributions.
Cell-kinetic Factors.
a) Dilution.
"Once an MN, always an MN". For practical purposes, MN, once formed, do not disappear for a long time, even in cells that have lost the ability to divide. Nor, in the vast majority of cases, do MN themselves divide, although when present at mitosis, they occasionally show "premature chromosome condensation" (PCC). Since nearly all cells that carry AF have a limited life span (~1-4 divisions), only a finite number of MN are produced, and the cells that are carrying them will soon be out-grown by "normal", non-AF producing, non-MN bearing, cells which will progressively "dilute" the observed MN/cell frequency.
This conflict between "generation" versus "dilution" with time after treatment leads to a "humped" yield-time curve, the profile of which is highly dose-dependent (Brock and Williams, 1985; Roberts et al., 1986). This means that often, there is no unique MN frequency that can be set against a given dose, with inevitable uncertainty in the shape of any dose-response curve. Consequently, since the rate of generation and of dilution are both affected by kinetic factors (see below), neither the peak frequency, nor the integrated area under the yield time curve, are reliable measures of damage. This is a problem all too familiar to those who attempt quantitative work with chromatid-type aberrations (Savage and Papworth, 1991).
b) Mitotic delay and perturbation.
Every known clastogen disturbs the orderly progression of cells towards division. The magnitude of the effect is dose, time, and probably stage dependent. Obviously, the changed cell-rates and orders will have a pronounced effect on factors like "generation" and "dilution" and, by changing the mixture of cells present in the scored sample, this will affect the observed frequency of MN.
c) Interphase "death".
The failure of a cell to reach the next mitosis after treatment. Sometimes loosely equated with "apoptosis", but this latter term should be reserved for the highly specialised "programmed" interphase death having certain well defined biomolecular characteristics. Interphase "death" as a general term, does not necessarily involve cessation of physiological activity, or nuclear degeneration (as an example consider the "feeder-layer" technique).
Interphase "death" is a regular feature of chemical clastogens, and some cell types show it after ionizing radiation (e.g. unstimulated lymphocytes). If extensive, a severe reduction in MN production will result. The phenomenon may not be random, affecting only certain developmental phases, so some bias of the AF source may be introduced.
The Time-displacement Factor.
This is probably the most frequently overlooked factor when quantitative comparisons between AF and MN are made (Savage, 1989).
It is the failure to remember that aberration frequencies are determined at a point in time (usually after a short colcemid metaphase accumulation) and are therefore "instantaneous" mean frequencies. Observed frequencies of MN are, however, "running" means, based on the cumulative number of MN derived from all divisions antecedent to the time of sampling.
Now, an instantaneous-mean and a running-mean are mathematically quite different things, and are not readily comparable. One of the dramatic differences is, that in sequential samples, significant fluctuations seen in the former, are thoroughly damped in the latter, and this effectively severs any meaningful relationship between AF and MN.
Thus, for example, in simple ratio comparisons of MN/AF, we need to remember that we are not comparing like with like, and the derived relationship can be wildly out. Predictable ratios exist only in the simplest hypothetical populations (Savage, 1989).
A technical innovation, Cytochalasin-B
The conflict between "generation" and "dilution" was recognised fairly quickly and several protocols were suggested to counteract it.
One obvious solution is to identify and confine scoring to those cells that have divided once, and once only, since the clastogen was given. This eliminates dilution by the overgrowth of undamaged cells coming round for the second time.
The most popular and widely used method to achieve this, utilises a drug called Cytochalasin-B (Cyt-B) (Fenech and Morley, 1985; Fenech and Morley, 1986; Wakata and Sasaki, 1987). In the presence of this compound at appropriate dilution (2-6 mg/ml) the nucleus goes through mitosis, but the daughter cells fail to separate, leading to a bi-nucleate cell. Obviously, such cells must have divided once since Cyt-B was added, and MN scoring can be confined to this sub-set.
Provided everything, apart from cell separation, is normal, the frequency of (MN/binucleate-cell) will be twice that which would have been found in once-divided mononucleate-cells. The frequency of binucleate-cells with MN will also be approximately twice that of once-divided mononucleate-cells.
The use of Cyt-B is, of course, only a partial solution, since we only collect the MN formed from one mitosis. Therefore, any treatment effects, or modifications in PI will be reflected in the observed MN frequency. However, it is a very useful method now almost universally used for micronucleus studies, and it has helped to clear up a lot of the problems which plagued the early work.
Cyt-B can be used in sequential "pulse" treatments to collect cohorts of cells at different times after treatment. This enables one to follow the generation and the dilution at different times after treatment.
There is still a lot to learn about the action of Cyt-B. Curiously, only a proportion of cells seems to be trapped, irrespective of concentration; mononucleate cells with MN are always contemporary with binucleate ones. Some of the trapped cells will divide again if treatment is prolonged, but spindles become multipolar and there is much non-disjunction so such cells are useless for longer-term MN studies.
Chronic irradiation and chemical treatments.
So far, we have viewed the problems of quantitative work on the relations of AF and MN assuming an acute dose of clastogen. If, however, a chronic dose is given - either by prolonged exposure time, or by incorporating a radioactive source into the nucleus, then we introduce an additional set of problems. This is a particular difficulty when chemical clastogens are used, as the majority of these are long-lived, and/or the molecules and lesions persist within the cells producing additional primary aberrations in subsequent cell generations. In practice, it is almost impossible to give anything approaching a clean acute treatment, as one can when using external radiation.
There is much intra-nuclear change going on as the cell transits the various phases of the mitotic cycle, so it is not surprising that sensitivity to clastogen effects (aberration type and frequency, mitotic delay and perturbation, and probably effectiveness of repair) varies from one stage to another. With chronic treatment, each cell must "run the gauntlet" of these changes whilst damage production and repair are taking place, and the various effects will accumulate and confound. The repair processes will be continually busy, stretched and probably over-stretched, whilst the cell is trying to cope with the continuous influx of problems (rather like mending a leaking pipe whilst someone is boring holes elsewhere !).
The overall effect is to introduce additional perturbations, frequent (often selective) cell death, and the conflict between "generation" and "dilution" will be considerably modified. Quite likely, the yield-time curve will have extra peaks and troughs and will not reflect the true sensitivity situation within the population.
We always have to remember that the observed frequency of any event, with which we construct our graphs, and from which we draw our inferences, depends entirely upon the mixture of cells which is present in the sample scored (Savage and Papworth, 1991). Changes in this cell mixture can produce effects as profound as real treatment-induced changes. It often requires much wisdom to tell the difference.
| Bibliography |
| Cell survival and radiation induced chromosome aberrations. I. Derivation of formulae for the determination of transmission and survival parameters of aberrations. |
| Braselmann H, Bauchinger M, Schmid E |
| Radiation and environmental biophysics. 1986 ; 25 (4) : 243-251. |
| PMID 3823371 |
| Kinetics of micronucleus expression in synchronized irradiated Chinese hamster ovary cells. |
| Brock WA, Williams M |
| Cell and tissue kinetics. 1985 ; 18 (3) : 247-254. |
| PMID 3986869 |
| Chromosome aberrations in patients treated with X-irradiation for ankylosing spondylitis. In: Radiation-induced Chromosome Damage in Man |
| Buckton KE |
| (eds. Ishihara, T. and Sasaki, M.) Alan Liss, New York. 1983 : 491-511. |
| The fate of chromosome aberrations. |
| Carrano AV, Heddle JA |
| Journal of theoretical biology. 1973 ; 38 (2) : 289-304. |
| PMID 4689998 |
| The production of micronuclei from chromosome aberrations in irradiated cultures of human lymphocytes. |
| Countryman PI, Heddle JA |
| Mutation research. 1976 ; 41 (2-3) : 321-332. |
| PMID 796719 |
| The fate of X-ray-induced chromosome aberrations in blood lymphocyte culture. |
| Das BC, Sharma T |
| Mutation research. 1987 ; 176 (1) : 93-104. |
| PMID 3796662 |
| Immunofluorescent staining of kinetochores in micronuclei: a new assay for the detection of aneuploidy. |
| Degrassi F, Tanzarella C |
| Mutation research. 1988 ; 203 (5) : 339-345. |
| PMID 3050494 |
| Measurement of micronuclei in lymphocytes. |
| Fenech M, Morley AA |
| Mutation research. 1985 ; 147 (1-2) : 29-36. |
| PMID 3974610 |
| Cytokinesis-block micronucleus method in human lymphocytes: effect of in vivo ageing and low dose X-irradiation. |
| Fenech M, Morley AA |
| Mutation research. 1986 ; 161 (2) : 193-198. |
| PMID 3724773 |
| Kinetochore detection in micronuclei: an alternative method for measuring chromosome loss. |
| Fenech M, Morley AA |
| Mutagenesis. 1989 ; 4 (2) : 98-104. |
| PMID 2659933 |
| A rapid in vivo test for chromosomal damage. |
| Heddle JA |
| Mutation research. 1973 ; 18 (2) : 187-190. |
| PMID 4351282 |
| Letter: Rapid screening of radioprotective drugs in vivo. |
| Heddle JA, Harris JW |
| Radiation research. 1975 ; 61 (2) : 350-353. |
| PMID 1167210 |
| Micronuclei in vivo. In: "Mutation and the environment, B |
| Heddle JA |
| (eds. Mendelsohn, M.L. and Albertini, R.J.), Wiley-Liss, New York. 1990 : 185-194. |
| Micronucleus assays. In: Advances in Mutagenesis Research |
| Müller W-U, Streffer C |
| (ed. Obe, G.) Springer-Verlag, Berlin. 1994 : 1-134. |
| A critical comparison of the micronucleus yield from high and low LET irradiation of plateau-phase cell populations. |
| Roberts CJ, Morgan GR, Holt PD |
| Mutation research. 1986 ; 160 (3) : 237-242. |
| PMID 3960037 |
| Classification and relationships of induced chromosomal structual changes. |
| Savage JR |
| Journal of medical genetics. 1976 ; 13 (2) : 103-122. |
| PMID 933108 |
| A comment on the quantitative relationship between micronuclei and chromosomal aberrations. |
| Savage JR |
| Mutation research. 1988 ; 207 (1) : 33-36. |
| PMID 3336377 |
| Acentric chromosomal fragments and micronuclei: the time-displacement factor. |
| Savage JR |
| Mutation research. 1989 ; 225 (4) : 171-173. |
| PMID 2927437 |
| Excogitations about the quantification of structural chromosomal aberrations, in: Advances in mutagenesis research, 3 |
| Savage JRK, Papworth DG |
| (ed. Obe, G.), Springer-Verlag, Berlin. 1991 : 162-189. |
| Micronuclei-biological indicator for retrospective dosimetry after exposure to ionizing radiation. |
| Streffer C, Müller WU, Kryscio A, Böcker W |
| Mutation research. 1998 ; 404 (1-2) : 101-105. |
| PMID 9729308 |
| Use of an antikinetochore antibody to discriminate between micronuclei induced by aneuploidogens and clastogens. In: "Mutation and the environment, B |
| Tucker JD, Eastmond DA |
| (eds. Mendelsohn, M.L. and Albertini, R.J.), Wiley-Liss, New York. 1990 : 275-284. |
| Measurement of micronuclei by cytokinesis-block method in cultured Chinese hamster cells: comparison with types and rates of chromosome aberrations. |
| Wakata A, Sasaki MS |
| Mutation research. 1987 ; 190 (1) : 51-57. |
| PMID 3099194 |
| Written | 2000-07 | John R K Savage |
| City Road, Tilehurst, Reading, RG31 5HB, UK |
| Citation |
| This paper should be referenced as such : |
| Savage, JRK |
| Micronuclei: pitfalls, problems |
| Atlas Genet Cytogenet Oncol Haematol. 2000;4(4):229-233. |
| Free journal version : [ pdf ] [ DOI ] |
| On line version : http://AtlasGeneticsOncology.org/Deep/MicronucleiID20016.htm |
Citation
Atlas of Genetics and Cytogenetics in Oncology and Haematology
Micronuclei : Pitfalls and Problems
Online version: http://atlasgeneticsoncology.org/deep-insight/20016/micronuclei-pitfalls-and-problems
