MSH6 (mutS homolog 6 (E. Coli))
2006-11-01 Sreeparna Banerjee AffiliationDepartment of Biology, Office: Z-16\\\/Lab: B-59, Middle East Technical University, 06531 Ankara, Turkey
Identity
HGNC
LOCATION
2p16.3
LOCUSID
ALIAS
GTBP,GTMBP,HNPCC5,HSAP,MMRCS3,p160
FUSION GENES
DNA/RNA
Note
The genes for MSH2 and MSH6 which form the major mismatch recognition MutSalpha complex functional in the mismatch repair (MMR) pathway are located within 1 Mb of each other. MSH2 and MSH6 may have been produced by duplication of a primordial mutS repair gene.
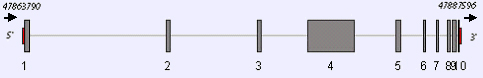
Exons are represented by gray boxes (in scale) with exon numbers on the bottom. The arrows show the ATG and the stop codons respectively.
Description
MSH6 gene maps to NC_000002.10 and spans a region of 23.8 kilo bases.
MSH6 has 10 exons, the sizes being 347, 197, 170, 2545, 266, 119, 89, 155, 200 and 176 bps.
MSH6 has 10 exons, the sizes being 347, 197, 170, 2545, 266, 119, 89, 155, 200 and 176 bps.
Transcription
Human MSH6 gene is transcriptionally upregulated 2.5 fold at late G1/early S phase while the amount of protein remains unchanged during the whole cell cycle.
The promoter region has a high GC content, as well as multiple start sites. Sequence analysis of 3.9 kb of the 5-upstream region of the MSH6 gene revealed the absence of TATAA- or CAAT-boxes. Seven consensus binding sequences for the ubiquitous transcription factor Sp1 were found in the promoter region. This factor is implicated in positioning the RNA polymerase II complex at the transcriptional start sites of promoters lacking TATA- and CAAT-boxes. The proximal promoter region of MSH6 gene also contains several consensus binding sites of the embryonic TEA domain-containing factor ETF. This transcription factor has also been reported to stimulate transcription from promoters lacking the TATA box. In addition, the trancription of MSH6 gene is downregulated by CpG methylation of the promoter region.
Three common polymorphic variants (-557 T G, -448 G A, and -159 C T) of the MSH6 promoter have been identified in which different Sp1 sites were inactivated by single-nucleotide polymorphisms (SNPs) resulting in altered promoter activity.
The promoter region has a high GC content, as well as multiple start sites. Sequence analysis of 3.9 kb of the 5-upstream region of the MSH6 gene revealed the absence of TATAA- or CAAT-boxes. Seven consensus binding sequences for the ubiquitous transcription factor Sp1 were found in the promoter region. This factor is implicated in positioning the RNA polymerase II complex at the transcriptional start sites of promoters lacking TATA- and CAAT-boxes. The proximal promoter region of MSH6 gene also contains several consensus binding sites of the embryonic TEA domain-containing factor ETF. This transcription factor has also been reported to stimulate transcription from promoters lacking the TATA box. In addition, the trancription of MSH6 gene is downregulated by CpG methylation of the promoter region.
Three common polymorphic variants (-557 T G, -448 G A, and -159 C T) of the MSH6 promoter have been identified in which different Sp1 sites were inactivated by single-nucleotide polymorphisms (SNPs) resulting in altered promoter activity.
Pseudogene
No pseudogene has been reported for the MSH6 gene.
Proteins
Note
Eukaryotic MutSalpha is a heterodimer of the 100-kDa MSH2 and the 160-kDa MSH6 that participates in the mismatch repair pathway. The proteins are required for single base and frameshift mispair specific binding, a result consistent with the finding that tumour-derived cell lines devoid of either protein have a mutator phenotype.
Description
The MSH6 protein maps to NP_000170 and has 1360 amino acids. The molecular weight is 152786 Da. The protein contains a highly conserved helix-turn-helix domain associated with a Walker-A motif (an adenine nucleotide and magnesium binding motif) with ATPase activity.
The breast cancer 1 gene (BRCA1) product is part of a large multisubunit protein complex of tumor suppressors, DNA damage sensors, and signal transducers. This complex is called BASC, for BRCA1-associated genome surveillance complex and the mismatch repair protein MSH6 was found to be a part of this complex.
The breast cancer 1 gene (BRCA1) product is part of a large multisubunit protein complex of tumor suppressors, DNA damage sensors, and signal transducers. This complex is called BASC, for BRCA1-associated genome surveillance complex and the mismatch repair protein MSH6 was found to be a part of this complex.
Localisation
The subcellular localisation of MSH6 is the nucleus.
Function
hMSH6 gene product with hMSH2, hMSH3 gene products play role in strand specific repair of DNA replication errors. Studies show that hMSH2-hMSH6 complex functions in the recognition step of the repair of base-base mismatches or single frameshifts. The ADP/ATP binding domain of the heterodimer and the associated ATPase activity function to regulate mismatch binding as a molecular switch. Both MSH2 and MSH6 can simultaneously bind ATP. The MSH6 subunit contains the high-affinity ATP binding site and MSH2 contains a high-affinity ADP binding site. Stable binding of ATP to MSH6 results in a decreased affinity of MSH2 for ADP, and binding to mispaired DNA stabilizes the binding of ATP to MSH6. Mispair binding encourages a dual-occupancy state with ATP bound to Msh6 and Msh2; following which there is a hydrolysis-independent sliding along DNA. Subsequent steps result in the excision of the mispaired region followed by DNA synthesis and ligation.
Homology
H.sapiens: MSH6 (mutS homolog 6 (E. coli)).
C.familiaris: LOC474585 (similar to mutS homolog 6).
M.musculus: Msh6 (mutS homolog 6 (E. coli)).
C.elegans: msh-6 (MSH (MutS Homolog) family).
S.pombe: SPCC285.16c (hypothetical protein).
S.cerevisiae: MSH6 (Mismatch repair protein).
A.thaliana: MSH6 (MSH6).
C.familiaris: LOC474585 (similar to mutS homolog 6).
M.musculus: Msh6 (mutS homolog 6 (E. coli)).
C.elegans: msh-6 (MSH (MutS Homolog) family).
S.pombe: SPCC285.16c (hypothetical protein).
S.cerevisiae: MSH6 (Mismatch repair protein).
A.thaliana: MSH6 (MSH6).
Mutations
Note
The MSH6 gene plays a role in the development of inherited cancers, especially the colorectum and endometrial cancers.
Germinal
MSH6 germline mutations have variable penetration. Atypical hereditary non polyposis colorectal cancer (HNPCC) can result from germline mutations in MSH6; however, disease-causing germline mutations of MSH6 are rare in HNPCC and HNPCC-like families. Other studies have indicated that germline MSH6 mutations may contribute to a subset of early-onset colorectal cancer.
Somatic
The involvement of somatic or epigenetic inactivation of hMSH6 is rare in colorectal cancer and missense mutations in MSH6 are often clinically innocuous or have a low penetrance. However, somatic mutations of MSH6 have been shown to confer resistance to alkylating agents such as temozolomide in malignant gliomas in vivo. This concurrently results in accelerated mutagenesis in resistant clones as a consequence of continued exposure to alkylating agents in the presence of defective mismatch repair. Therefore, when MSH6 is inactivated in gliomas, there is a change in status of the alkylating agents from induction of tumour cell death to promotion of neoplastic progression.
Implicated in
Entity name
Disease
Mutations in the mismatch repair genes MSH2, MSH6, < CC: TXT: MLH1 ID: 149> and PMS2 results in hereditary non polyposis colorectal cancer (HNPCC, Lynch syndrome). Individuals predisposed to this syndrome have increased lifetime risk of developing colorectal, endometrial and other cancers. The resulting mismatch repair deficiency leads to microsatellite instability which is the hallmark of tumors arising within this syndrome, as well as a variable proportion of sporadic tumors.
Clinically, HNPCC can be divided into two subgroups:
Type I: a young onset age for hereditary colorectal cancer, and carcinoma of the proximal colon.
Type II: patients are susceptible to cancers in tissues such as the colon, uterus, ovary, breast, stomach, small intestine and skin.
Diagnosis of classical HNPCC is based on the Amsterdam criteria:
- 3 or more relatives affected by colorectal cancer, one a first degree relative of the other two;
- 2 or more generation affected;
- 1 or more colorectal cancers presenting before 50 years of age; exclusion of hereditary polyposis syndromes.
Clinically, HNPCC can be divided into two subgroups:
Type I: a young onset age for hereditary colorectal cancer, and carcinoma of the proximal colon.
Type II: patients are susceptible to cancers in tissues such as the colon, uterus, ovary, breast, stomach, small intestine and skin.
Diagnosis of classical HNPCC is based on the Amsterdam criteria:
- 3 or more relatives affected by colorectal cancer, one a first degree relative of the other two;
- 2 or more generation affected;
- 1 or more colorectal cancers presenting before 50 years of age; exclusion of hereditary polyposis syndromes.
Entity name
Disease
Turcot syndrome is a condition whereby central nervous system malignant tumours are associated with familial colorectal cancer. A homozygous mutation in MSH6 has been reported in a family with childhood-onset brain tumour, lymphoma, colorectal cancer, and neurofibromatosis type 1 phenotype.
Entity name
Colorectal cancer.
Disease
Mutations in four mismatch repair genes MSH2, MLH1, MSH6, and PMS2, have been convincingly linked to susceptibility of hereditary nonpolyposis colorectal cancer (HNPCC)/Lynch syndrome. Of the 500 different HNPCC-associated MMR gene mutations known, approximately 10% are associated with mutations in the MSH6 gene.
Entity name
Endometrial cancer
Disease
Germline mutations in the MSH6 gene are often observed in HNPCC-like families with an increased frequency of endometrial cancer. Sequence analysis of the MSH6 coding region revealed the presence of three putative missense mutations in patients with atypical family histories that do not meet HNPCC criteria. MSH6 mutations may contribute to the etiology of double primary carcinomas of the colorectum and endometrium.
Entity name
Ovarian cancer
Disease
Late-onset endometrioid type of ovarian cancer can be linked to MSH6 germline mutations.
Entity name
Lung cancer
Disease
Early onset lung cancer (before age 50) has been associated with polymorphisms in the MSH6 gene. Cadmium, an environmental and occupational carcinogen associated with lung cancer development was shown to inhibit the ATPase activity of MSH2-MSH6 heterodimer.
Entity name
Breast cancer
Disease
Mutations in the MSH6 gene are not usually connected with breast cancer, even when associated with endometrial or colorectal cancer.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16434208 | 2006 | The genetics of HNPCC: application to diagnosis and screening. | Abdel-Rahman WM et al |
| 8942985 | 1996 | hMSH2 forms specific mispair-binding complexes with hMSH3 and hMSH6. | Acharya S et al |
| 15746000 | 2005 | Cadmium inhibits mismatch repair by blocking the ATPase activity of the MSH2-MSH6 complex. | Banerjee S et al |
| 16807412 | 2006 | Identification and survival of carriers of mutations in DNA mismatch-repair genes in colon cancer. | Barnetson RA et al |
| 11153917 | 2000 | Do MSH6 mutations contribute to double primary cancers of the colorectum and endometrium? | Charames GS et al |
| 14585961 | 2003 | Regulation of the human MSH6 gene by the Sp1 transcription factor and alteration of promoter activity and expression by polymorphisms. | Gazzoli I et al |
| 9428522 | 1997 | The human mismatch recognition complex hMSH2-hMSH6 functions as a novel molecular switch. | Gradia S et al |
| 10078208 | 1999 | hMSH2-hMSH6 forms a hydrolysis-independent sliding clamp on mismatched DNA. | Gradia S et al |
| 16000562 | 2005 | A homozygous mutation in MSH6 causes Turcot syndrome. | Hegde MR et al |
| 16618716 | 2006 | A hypermutation phenotype and somatic MSH6 mutations in recurrent human malignant gliomas after alkylator chemotherapy. | Hunter C et al |
| 16612326 | 2006 | The multifaceted mismatch-repair system. | Jiricny J et al |
| 15354210 | 2004 | MSH6 missense mutations are often associated with no or low cancer susceptibility. | Kariola R et al |
| 17108146 | 2006 | DNA repair and cell cycle control genes and the risk of young-onset lung cancer. | Landi S et al |
| 16600868 | 2006 | Inhibition of Msh6 ATPase activity by mispaired DNA induces a Msh2(ATP)-Msh6(ATP) state capable of hydrolysis-independent movement along DNA. | Mazur DJ et al |
| 7604265 | 1995 | GTBP, a 160-kilodalton protein essential for mismatch-binding activity in human cells. | Palombo F et al |
| 7604266 | 1995 | Mutations of GTBP in genetically unstable cells. | Papadopoulos N et al |
| 16136382 | 2005 | Lynch syndrome genes. | Peltomäki P et al |
| 14520694 | 2003 | MSH6 germline mutations are rare in colorectal cancer families. | Peterlongo P et al |
| 16940983 | 2006 | MSH6 germline mutations in early-onset colorectal cancer patients without family history of the disease. | Pinto C et al |
| 11807791 | 2002 | Involvement of hMSH6 in the development of hereditary and sporadic colorectal cancer revealed by immunostaining is based on germline mutations, but rarely on somatic inactivation. | Plaschke J et al |
| 15660526 | 2005 | Inherited susceptibility to colorectal cancer. | Rowley PT et al |
| 16270383 | 2005 | Low prevalence of germline hMSH6 mutations in colorectal cancer families from Spain. | Sánchez de Abajo A et al |
| 12376742 | 2002 | Ovarian cancer of endometrioid type as part of the MSH6gene mutation phenotype. | Suchy J et al |
| 11746986 | 2002 | Identification and functional characterization of the promoter region of the human MSH6 gene. | Szadkowski M et al |
| 15805151 | 2005 | No MSH6 germline mutations in breast cancer families with colorectal and/or endometrial cancer. | Vahteristo P et al |
| 10783165 | 2000 | BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures. | Wang Y et al |
Other Information
Locus ID:
NCBI: 2956
MIM: 600678
HGNC: 7329
Ensembl: ENSG00000116062
Variants:
dbSNP: 2956
ClinVar: 2956
TCGA: ENSG00000116062
COSMIC: MSH6
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA443761 | Colorectal Neoplasms, Hereditary Nonpolyposis | Disease | Literature, MultilinkAnnotation | associated | 23788249 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38390329 | 2024 | Mismatch repair gene MSH6 correlates with the prognosis, immune status and immune checkpoint inhibitors response of endometrial cancer. | 2 |
| 38840222 | 2024 | TDP43 interacts with MLH1 and MSH6 proteins in a DNA damage-inducible manner. | 0 |
| 38390329 | 2024 | Mismatch repair gene MSH6 correlates with the prognosis, immune status and immune checkpoint inhibitors response of endometrial cancer. | 2 |
| 38840222 | 2024 | TDP43 interacts with MLH1 and MSH6 proteins in a DNA damage-inducible manner. | 0 |
| 36931573 | 2023 | Molecular Profile of MSH6-Associated Colorectal Carcinomas Shows Distinct Features From Other Lynch Syndrome-Associated Colorectal Carcinomas. | 3 |
| 37307877 | 2023 | Discordant Staining Patterns and Microsatellite Results in Tumors of MSH6 Pathogenic Variant Carriers. | 0 |
| 36931573 | 2023 | Molecular Profile of MSH6-Associated Colorectal Carcinomas Shows Distinct Features From Other Lynch Syndrome-Associated Colorectal Carcinomas. | 3 |
| 37307877 | 2023 | Discordant Staining Patterns and Microsatellite Results in Tumors of MSH6 Pathogenic Variant Carriers. | 0 |
| 33901323 | 2022 | DNA methylation status of MutS genes in ameloblastoma. | 1 |
| 35346574 | 2022 | Early age of onset and broad cancer spectrum persist in MSH6- and PMS2-associated Lynch syndrome. | 4 |
| 35855666 | 2022 | Novel MSH6 mutation predicted metastasis in eyelid and periocular squamous cell carcinoma. | 1 |
| 35868444 | 2022 | Structural, functional analysis and association of MSH6 rs1800932, rs1042821 polymorphisms with clinical outcome in North Indian lung cancer patients treated with platinum-based doublet chemotherapy. | 0 |
| 36073783 | 2022 | Germline variants screening of MLH1, MSH2, MSH6 and PMS2 genes in 64 Algerian Lynch syndrome families: The first nationwide study. | 0 |
| 36189922 | 2022 | S6K1 phosphorylates Cdk1 and MSH6 to regulate DNA repair. | 2 |
| 33901323 | 2022 | DNA methylation status of MutS genes in ameloblastoma. | 1 |
Citation
Sreeparna Banerjee
MSH6 (mutS homolog 6 (E. Coli))
Atlas Genet Cytogenet Oncol Haematol. 2006-11-01
Online version: http://atlasgeneticsoncology.org/gene/344/msh6-(muts-homolog-6-(e-coli))
