GATA1 (GATA binding protein 1 (globin transcription factor1))
2019-07-01 Winston Y. Lee , Olga K. Weinberg AffiliationLee, WY : Department of Pathology, University of Michigan Medical School, Ann Arbor, MI, (WYL); Department of Pathology, Brigham and Womens Hospital, Harvard Medical School, Boston, MA, (OKW) USA
Identity
Abstract
We provide a survey of the disease entities associated with GATA1 mutations.
DNA/RNA
Description
Transcription
Proteins
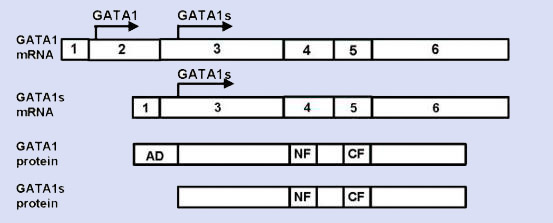
Description
Expression
Localisation
Function
Homology
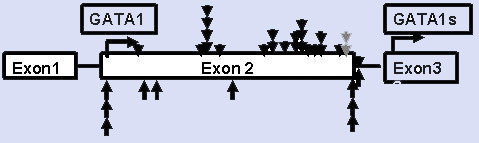
Mutations
Germinal
Somatic
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 30914438 | 2019 | Impaired human hematopoiesis due to a cryptic intronic GATA1 splicing mutation. | Abdulhay NJ et al |
| 14656875 | 2004 | Natural history of GATA1 mutations in Down syndrome. | Ahmed M et al |
| 16103636 | 2005 | Dyserythropoietic anemia and thrombocytopenia due to a novel mutation in GATA-1. | Del Vecchio GC et al |
| 11809723 | 2002 | Different substitutions at residue D218 of the X-linked transcription factor GATA1 lead to altered clinical severity of macrothrombocytopenia and anemia and are associated with variable skewed X inactivation. | Freson K et al |
| 14744791 | 2004 | The role of cytidine deaminase and GATA1 mutations in the increased cytosine arabinoside sensitivity of Down syndrome myeloblasts and leukemia cell lines. | Ge Y et al |
| 12747884 | 2003 | Acquired mutations in GATA1 in neonates with Down's syndrome with transient myeloid disorder. | Groet J et al |
| 26186939 | 2015 | The biology of pediatric acute megakaryoblastic leukemia. | Gruber TA et al |
| 14512321 | 2004 | Recent insights into the mechanisms of myeloid leukemogenesis in Down syndrome. | Gurbuxani S et al |
| 15070711 | 2004 | The GATA1 mutation in an adult patient with acute megakaryoblastic leukemia not accompanying Down syndrome. | Harigae H et al |
| 15630411 | 2005 | Origins of leukaemia in children with Down syndrome. | Hitzler JK et al |
| 23940284 | 2013 | Congenital dyserythropoietic anemias: molecular insights and diagnostic approach. | Iolascon A et al |
| 15198727 | 2004 | Leukaemia -- a developmental perspective. | Izraeli S et al |
| 29551269 | 2018 | Ribosome Levels Selectively Regulate Translation and Lineage Commitment in Human Hematopoiesis. | Khajuria RK et al |
| 24766296 | 2014 | Recurrent GATA1 mutations in Diamond-Blackfan anaemia. | Klar J et al |
| 17713552 | 2008 | Congenital transfusion-dependent anemia and thrombocytopenia with myelodysplasia due to a recurrent GATA1(G208R) germline mutation. | Kratz CP et al |
| 29481579 | 2018 | Loss of Full-Length GATA1 Expression in Megakaryocytes Is a Sensitive and Specific Immunohistochemical Marker for the Diagnosis of Myeloid Proliferative Disorder Related to Down Syndrome. | Lee WY et al |
| 15895080 | 2005 | Developmental stage-selective effect of somatically mutated leukemogenic transcription factor GATA1. | Li Z et al |
| 12172549 | 2002 | A leukemogenic twist for GATA1. | Look AT et al |
| 24952648 | 2014 | Altered translation of GATA1 in Diamond-Blackfan anemia. | Ludwig LS et al |
| 10700180 | 2000 | Familial dyserythropoietic anaemia and thrombocytopenia due to an inherited mutation in GATA1. | Nichols KE et al |
| 23733339 | 2013 | Exome sequencing identifies putative drivers of progression of transient myeloproliferative disorder to AMKL in infants with Down syndrome. | Nikolaev SI et al |
| 24453067 | 2014 | Loss of GATA-1 full length as a cause of Diamond-Blackfan anemia phenotype. | Parrella S et al |
| 12649131 | 2003 | Mutations in exon 2 of GATA1 are early events in megakaryocytic malignancies associated with trisomy 21. | Rainis L et al |
| 22706301 | 2012 | Exome sequencing identifies GATA1 mutations resulting in Diamond-Blackfan anemia. | Sankaran VG et al |
| 15317736 | 2004 | Prenatal origin of GATA1 mutations may be an initiating step in the development of megakaryocytic leukemia in Down syndrome. | Taub JW et al |
| 10216081 | 1999 | Consequences of GATA-1 deficiency in megakaryocytes and platelets. | Vyas P et al |
| 12172547 | 2002 | Acquired mutations in GATA1 in the megakaryoblastic leukemia of Down syndrome. | Wechsler J et al |
| 24056718 | 2013 | The landscape of somatic mutations in Down syndrome-related myeloid disorders. | Yoshida K et al |
| 26713410 | 2016 | A Child With Dyserythropoietic Anemia and Megakaryocyte Dysplasia Due to a Novel 5'UTR GATA1s Splice Mutation. | Zucker J et al |
Other Information
Locus ID:
NCBI: 2623
MIM: 305371
HGNC: 4170
Ensembl: ENSG00000102145
Variants:
dbSNP: 2623
ClinVar: 2623
TCGA: ENSG00000102145
COSMIC: GATA1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000102145 | ENST00000376665 | B7WNQ9 |
| ENSG00000102145 | ENST00000376670 | P15976 |
| ENSG00000102145 | ENST00000651144 | P15976 |
Expression (GTEx)
Pathways
| Pathway | Source | External ID |
|---|---|---|
| Hemostasis | REACTOME | R-HSA-109582 |
| Factors involved in megakaryocyte development and platelet production | REACTOME | R-HSA-983231 |
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37171759 | 2024 | ARHGAP18 is Upregulated by Transcription Factor GATA1 Promotes the Proliferation and Invasion in Hepatocellular Carcinoma. | 1 |
| 38103735 | 2024 | Aggregates of nonmuscular myosin IIA in erythrocytes associate with GATA1- and GFI1B-related thrombocytopenia. | 0 |
| 38159323 | 2024 | A novel GATA1 variant p.G229D causing the defect of procoagulant platelet formation. | 0 |
| 38350611 | 2024 | miR-1202 acts as anti-oncomiR in myeloid leukaemia by down-modulating GATA-1(S) expression. | 1 |
| 38695236 | 2024 | An intricate regulatory circuit between FLI1 and GATA1/GATA2/LDB1/ERG dictates erythroid vs. megakaryocytic differentiation. | 0 |
| 37171759 | 2024 | ARHGAP18 is Upregulated by Transcription Factor GATA1 Promotes the Proliferation and Invasion in Hepatocellular Carcinoma. | 1 |
| 38103735 | 2024 | Aggregates of nonmuscular myosin IIA in erythrocytes associate with GATA1- and GFI1B-related thrombocytopenia. | 0 |
| 38159323 | 2024 | A novel GATA1 variant p.G229D causing the defect of procoagulant platelet formation. | 0 |
| 38350611 | 2024 | miR-1202 acts as anti-oncomiR in myeloid leukaemia by down-modulating GATA-1(S) expression. | 1 |
| 38695236 | 2024 | An intricate regulatory circuit between FLI1 and GATA1/GATA2/LDB1/ERG dictates erythroid vs. megakaryocytic differentiation. | 0 |
| 36120774 | 2023 | Transcription factor GATA1 represses oxidized-low density lipoprotein-induced pyroptosis of human coronary artery endothelial cells. | 0 |
| 36542956 | 2023 | LncRNA RNA XIST binding to GATA1 contributes to rheumatoid arthritis through its effects on proliferation of synovial fibroblasts and angiogenesis via regulation of CCN6. | 3 |
| 36749159 | 2023 | GATA-1 Promotes Erythroid Differentiation Through the Upregulation of miR-451a and miR-210-3p Expressions in CD34(+) Cells in High-Altitude Polycythemia. | 1 |
| 36987811 | 2023 | SATB1 Chromatin Loops Regulate Megakaryocyte/Erythroid Progenitor Expansion by Facilitating HSP70 and GATA1 Induction. | 0 |
| 37254808 | 2023 | Multidimensional profiling reveals GATA1-modulated stage-specific chromatin states and functional associations during human erythropoiesis. | 3 |
Citation
Winston Y. Lee ; Olga K. Weinberg
GATA1 (GATA binding protein 1 (globin transcription factor1))
Atlas Genet Cytogenet Oncol Haematol. 2019-07-01
Online version: http://atlasgeneticsoncology.org/gene/40689/gata1-(gata-binding-protein-1-(globin-transcription-factor1))
Historical Card
2005-02-01 GATA1 (GATA binding protein 1 (globin transcription factor1)) by Shai Izraeli Affiliation
