PIK3CA (phosphoinositide-3-kinase, catalytic, alpha polypeptide)
2018-06-01 Julia Phillips , Enric Domingo AffiliationDepartment of Oncology, University of Oxford, Oxford, United Kingdom \\\/ [email protected]
Identity
Abstract
Review on PIK3CA, with data on DNA, on the protein encoded, and where the gene is implicated.
DNA/RNA
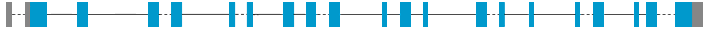
Description
Transcription
Pseudogene
Proteins
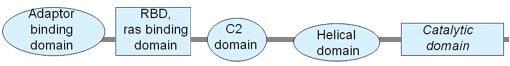
Description
Expression
Localisation
Function
Mutations
Somatic
The distribution of PIK3CA hotspot mutations can be significantly variable across tumour types in the kinase and helical domain. For example, cervical and bladder cancers share similar distributions of hotspots but they have far fewer H1047R kinase mutations than breast cancer. Similarly, endometrial and colorectal cancers share distribution patterns but display more R88Q mutations than other tumor types, maybe because this change is a target mutation of POLE genomic instability. However, colorectal cancers differ from endometrial cancers in their clonality. In endometrial tumors, both E545 helical domain mutations and H1047 kinase domain mutations are early clonal mutations whereas in colorectal cancer several E545 mutations are subclonal and H1047 mutations clonal (Chang, 2016). The variation in hotspot distributions suggests different functions need to be targeted in different tumour types.
Different domain mutations in PIK3CA have been associated with differential signaling. Kinase domain mutations have higher activation of PI3K complexes and increased G-alpha signaling events. However, some components of the G-alpha signaling pathway such as RHO GTPase complexes have lower activity in the kinase domain mutants relative to other PIK3CA mutants. Additionally, helical domain mutations are associated with lower activation of pathways related to proliferation, such as FOXM1, MYC, and PLK1, in comparison to other PIK3CA mutants. The different patterns of pathway activation associated with mutations of specific PIK3CA domains suggest that kinase domain mutations are more strongly associated with proliferation activity, whereas helical domain mutations are more strongly associated with motility activities. These differences in function across domain specific mutations may drive the varying distribution of mutations seen across cancer types (Yau, 2014).
Epigenetics
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16432179 | 2006 | Cancer-specific mutations in PIK3CA are oncogenic in vivo. | Bader AG et al |
| 17877460 | 2008 | Effects of oncogenic p110alpha subunit mutations on the lipid kinase activity of phosphoinositide 3-kinase. | Carson JD et al |
| 26619011 | 2016 | Identifying recurrent mutations in cancer reveals widespread lineage diversity and mutational specificity. | Chang MT et al |
| 18418043 | 2008 | Insights into the oncogenic effects of PIK3CA mutations from the structure of p110alpha/p85alpha. | Huang CH et al |
| 21216929 | 2011 | PIK3CA mutations in patients with advanced cancers treated with PI3K/AKT/mTOR axis inhibitors. | Janku F et al |
| 29508857 | 2018 | Targeting the PI3K pathway in cancer: are we making headway? | Janku F et al |
| 29401002 | 2018 | Phosphatidylinositol 3-Kinase α-Selective Inhibition With Alpelisib (BYL719) in PIK3CA-Altered Solid Tumors: Results From the First-in-Human Study. | Juric D et al |
| 16764926 | 2007 | Rare mutations of the PIK3CA gene in malignancies of the hematopoietic system as well as endometrium, ovary, prostate and osteosarcomas, and discovery of a PIK3CA pseudogene. | Müller CI et al |
| 23246288 | 2013 | Germline PIK3CA and AKT1 mutations in Cowden and Cowden-like syndromes. | Orloff MS et al |
| 26712086 | 2016 | PIK3CA Mutation, Aspirin Use after Diagnosis and Survival of Colorectal Cancer. A Systematic Review and Meta-analysis of Epidemiological Studies. | Paleari L et al |
| 19621391 | 2010 | Cyclooxygenase-2 inhibition inhibits PI3K/AKT kinase activity in epithelial ovarian cancer. | Uddin S et al |
Other Information
Locus ID:
NCBI: 5290
MIM: 171834
HGNC: 8975
Ensembl: ENSG00000121879
Variants:
dbSNP: 5290
ClinVar: 5290
TCGA: ENSG00000121879
COSMIC: PIK3CA
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000121879 | ENST00000263967 | P42336 |
| ENSG00000121879 | ENST00000468036 | C9JAM9 |
| ENSG00000121879 | ENST00000477735 | C9J951 |
| ENSG00000121879 | ENST00000643187 | A0A2R8Y2F6 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA152241907 | lapatinib | Chemical | MultilinkAnnotation | associated | 25199759 | ||
| PA164722345 | MIR10A | Gene | Literature, MultilinkAnnotation | associated | 24522205 | ||
| PA166184523 | alpelisib | Chemical | LabelAnnotation | associated | |||
| PA24684 | AKT1 | Gene | Pathway | associated | 28362716 | ||
| PA27844 | ERBB2 | Gene | Pathway | associated | |||
| PA27846 | ERBB3 | Gene | Pathway | associated | |||
| PA27847 | ERBB4 | Gene | Pathway | associated | |||
| PA28180 | FLT1 | Gene | Pathway | associated | 28362716 | ||
| PA28183 | FLT4 | Gene | Pathway | associated | 28362716 | ||
| PA30086 | KDR | Gene | Pathway | associated | 28362716 | ||
| PA33312 | PIK3R1 | Gene | MultilinkAnnotation | associated | 26807692 | ||
| PA443622 | Carcinoma, Non-Small-Cell Lung | Disease | ClinicalAnnotation | associated | PK | PD | 20157331 |
| PA448803 | carboplatin | Chemical | ClinicalAnnotation | associated | PD | ||
| PA449014 | cisplatin | Chemical | ClinicalAnnotation | associated | PD | ||
| PA449383 | docetaxel | Chemical | ClinicalAnnotation | associated | PK | PD | 20157331 |
| PA451743 | trastuzumab | Chemical | MultilinkAnnotation | associated | 25199759 | ||
| PA7360 | EGFR | Gene | Pathway | associated |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 35951471 | 2024 | Hippo-YAP signaling activation and cross-talk with PI3K in oral cancer: A retrospective cohort study. | 1 |
| 37705207 | 2024 | Microcystic lymphatic malformations in Turner syndrome are due to somatic mosaicism of PIK3CA. | 0 |
| 37916958 | 2024 | Allosteric PI3Kα Inhibition Overcomes On-target Resistance to Orthosteric Inhibitors Mediated by Secondary PIK3CA Mutations. | 4 |
| 37949664 | 2024 | Molecular characterization of 13 patients with PIK3CA-related overgrowth spectrum using a targeted deep sequencing approach. | 0 |
| 37968472 | 2024 | APC/PIK3CA mutations and β-catenin status predict tankyrase inhibitor sensitivity of patient-derived colorectal cancer cells. | 0 |
| 37983855 | 2024 | Mucinous cystadenocarcinoma of the breast harbours TRPS1 expressions and PIK3CA alterations. | 0 |
| 38194422 | 2024 | PIK3CA regulates development of diabetes retinopathy through the PI3K/Akt/mTOR pathway. | 1 |
| 38238254 | 2024 | PIK3CA mutation status in apocrine carcinoma arising in apocrine gland hyperplasia/apocrine nevus: A study of four cases. | 0 |
| 38432445 | 2024 | PIK3CA mutation as an acquired resistance driver to EGFR-TKIs in non-small cell lung cancer: Clinical challenges and opportunities. | 1 |
| 38466449 | 2024 | PIK3CA mutation-driven immune signature as a prognostic marker for evaluating the tumor immune microenvironment and therapeutic response in breast cancer. | 0 |
| 38822093 | 2024 | PIK3CA mutations in endocrine-resistant breast cancer. | 1 |
| 38891994 | 2024 | Genetic Insights into Colorectal Cancer: Evaluating PI3K/AKT Signaling Pathway Genes Expression. | 0 |
| 35951471 | 2024 | Hippo-YAP signaling activation and cross-talk with PI3K in oral cancer: A retrospective cohort study. | 1 |
| 37705207 | 2024 | Microcystic lymphatic malformations in Turner syndrome are due to somatic mosaicism of PIK3CA. | 0 |
| 37916958 | 2024 | Allosteric PI3Kα Inhibition Overcomes On-target Resistance to Orthosteric Inhibitors Mediated by Secondary PIK3CA Mutations. | 4 |
Citation
Julia Phillips ; Enric Domingo
PIK3CA (phosphoinositide-3-kinase, catalytic, alpha polypeptide)
Atlas Genet Cytogenet Oncol Haematol. 2018-06-01
Online version: http://atlasgeneticsoncology.org/gene/415/pik3ca-(phosphoinositide-3-kinase-catalytic-alpha-polypeptide)
Historical Card
2009-04-01 PIK3CA (phosphoinositide-3-kinase, catalytic, alpha polypeptide) by Montserrat Sanchez-Cespedes Affiliation
