USP7 (ubiquitin specific peptidase 7 (herpes virus-associated))
2008-09-01 Kwang-Hyun Baek , Suresh Ramakrishna AffiliationLaboratory of Molecular Signal Transduction, Graduate School of Life Science, Biotechnology, Cell, Gene Therapy Research Institute, Pochon CHA university, CHA General Hospital, Seoul, Korea
Identity
HGNC
LOCATION
16p13.2
LOCUSID
ALIAS
HAFOUS,HAUSP,TEF1
FUSION GENES
DNA/RNA
Description
Consists of 31 Exons with a total transcription length of 4,013bps.
Transcription
The coding region of the gene starts from exon 1 to exon 31 (200th bps to 3508th bps). The length of the transcript is 3308 bps.
Pseudogene
None
Proteins
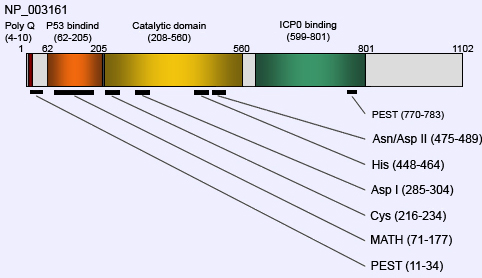
Description
HAUSP encodes for 1102 amino acids and its molecular weight is approximately 135kDa. MALDI-TOF/MS analysis has revealed four structural domains which are mainly involved in protein-protein interaction and deubiquitination activity.
N-terminal MATH (TRAF-like) domain (62-205aa) represented in brown colour is responsible for interaction with p53, MDM2 and EBNA1. N-terminal domain of USP7 complexed with Mdm2 at peptide 147-150 and with p53 at position 359-362 and 364-367 respectively.
Ubiquitin processing protease domain represented in yellow colour is a large family of cysteine proteases responsible for the cleavage of ubiquitin conjugates. Catalytic domain consists of approximately 350 amino acids, comprising three conserved domain architectures Finger, Palm, and Thumb. It has highly conserved Cys, Asp(I), His, and Asn/Asp(II) domains, which are responsible for deubiquitination activities.
ICP0 binding domain represented in green colour is located in the C-terminal region at position 599-801 amino acids. N-terminal polyglutamine (poly Q) region at position 4-10 amino acids which is conserved among mouse, rat and human.
N-terminal MATH (TRAF-like) domain (62-205aa) represented in brown colour is responsible for interaction with p53, MDM2 and EBNA1. N-terminal domain of USP7 complexed with Mdm2 at peptide 147-150 and with p53 at position 359-362 and 364-367 respectively.
Ubiquitin processing protease domain represented in yellow colour is a large family of cysteine proteases responsible for the cleavage of ubiquitin conjugates. Catalytic domain consists of approximately 350 amino acids, comprising three conserved domain architectures Finger, Palm, and Thumb. It has highly conserved Cys, Asp(I), His, and Asn/Asp(II) domains, which are responsible for deubiquitination activities.
ICP0 binding domain represented in green colour is located in the C-terminal region at position 599-801 amino acids. N-terminal polyglutamine (poly Q) region at position 4-10 amino acids which is conserved among mouse, rat and human.
Expression
HAUSP is expressed in wide variety of cell types including brain, liver, placenta, lung, ovary and melanocytes.
Localisation
HAUSP primarily localized in Nucleus.
Function
Herpesvirus-associated ubiquitin-specific protease was identified as a novel p53-interacting protein. HAUSP binds and stabilizes p53 through deubiquitination. It also strongly interacts with MDM2, hence playing an important role in the p53-MDM2 pathway resulting in p53-dependent cell growth repression and apoptosis. The tumor suppressor p53 protein is a transcription factor that responds to many cellular stress signals and is regulated primarily through ubiquitination and subsequent degradation. HAUSP contains an N-terminal TRAF-like domain in which p53 and MDM2 binds at the same site implied that HAUSP may function as a tumor suppressor by stabilizing p53.
HAUSP also interacts with the Epstein-Barr nuclear antigen 1 (EBNA1) protein of the Epstein-Barr virus (EBV), which is responsible for EBV latent infection and cellular transformation. Interaction of EBNA1 with USP7 occurs at same N-terminal TRAF-like domain at which p53 also binds to USP7. Through interactions with p53, MDM2 and EBNA1, HAUSP plays a role in cell proliferation, apoptosis and EBV-mediated immortalization.
HAUSP also interacts with the Epstein-Barr nuclear antigen 1 (EBNA1) protein of the Epstein-Barr virus (EBV), which is responsible for EBV latent infection and cellular transformation. Interaction of EBNA1 with USP7 occurs at same N-terminal TRAF-like domain at which p53 also binds to USP7. Through interactions with p53, MDM2 and EBNA1, HAUSP plays a role in cell proliferation, apoptosis and EBV-mediated immortalization.
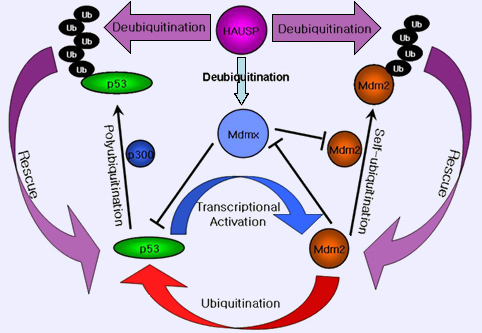
A regulatory model controlling the stability of p53 and Mdm2 by HAUSP.
Note: HAUSP can deubiquitinate p53, Mdm2, and Mdmx. The selfubiquitination activity of Mdm2 is important to regulate both Mdm2 and p53 at opposite levels. Mdmx stabilizes Mdm2 by inhibiting self-ubiquitination. HAUSP plays a crucial role for regulating the levels of p53, Mdm2, and Mdmx.
Homology
Human HAUSP shows 98.6% amino acid homology with both rat HAUSP and mouse HAUSP.
Implicated in
Entity name
Leukemia
Disease
Several studies implicated that ubiquitin proteasome pathway plays a critical role in thymocyte apoptosis (Beyette et al., 1998). Upon induction of apoptosis in murine thymocytes, USP7 specifically processes dexamethasone and gamma irradiation induced cell death (Vugmeyster et al., 2002). High expression was found in thymus, spleen and brain, organs which rely on apoptosis for development. A similar observation was not observed in caspase 3-deficient thymocytes or thymocytes treated with general caspase inhibitors indicating caspase involvement in the process of apoptosis.
Entity name
Herpes simplex
Disease
Herpes simplex virus type 1 immediate-early protein Vmw110 is a non-specific activator of gene expression and it is involved in the initiation of the viral lytic cycle. It has been demonstrated that USP7 interacts with Vmw 110 and its expression level is high during early infection (Everett et al., 1997). USP7 stabilizes herpes simplex virus type 1 regulatory protein ICP0 by its interaction during productive HSV-1 infection (Boutell et al., 2005).
Entity name
Cervical carcinoma
Disease
A chemistry-based functional proteomics approach to identify individual USPs in human papillomavirus (HPV) carrying cervical carcinoma and adjacent normal tissue by biopsies showed high expression of USP7. Upregulation of USP7 in cervical carcinoma suggests its role in growth transformation (Rolen et al., 2006).
Entity name
Tumor
Disease
USP7 was upregulated by mitogen activation or virus infection in normal T and B lymphocytes. USP7 expression was revealed by chemistry based functional proteomics approach in virus infected and tumor derived human cells (Ovaa et al., 2004). Holowaty and colleagues (2003) showed that USP7 interacts with Epstein-Barr nuclear antigen-1 (EBNA1) and involved in the regulation of EBNA1 replication activity. This findings suggests that USP7 has a critical role in EBV induced immortalization and tumorigenesis.
Entity name
Non-small cell lung cancers and adenocarcinomas
Note
Most non-small cell lung cancers (NSCLCs) shows a reduced herpesvirus-associated ubiquitin-specific protease expression. Therefore, the HAUSP gene might play an important role in carcinogenesis.
Disease
Quantitative reverse-transcription polymerase chain reaction (RT-PCR) and immunohistochemistry were performed to evaluate the protein expression of HAUSP in several patients with non-small cell lung cancer (NSCLC) (Masuya et al., 2006). Fifty-nine carcinomas (45.0%) showed reduced expression of HAUSP and HAUSP mRNA expression was significantly lower in adenocarcinomas and squamous cell carcinomas. In total, 93 carcinomas (71.0%) showed either mutant p53 or reduced HAUSP expression. The down-regulation of USP7 affects the p53 protein expression which in turn leads to tumors. These data show the importance of USP7 expression in NSCLC carcinogenesis, especially in adenocarcinomas.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16160161 | 2005 | Reciprocal activities between herpes simplex virus type 1 regulatory protein ICP0, a ubiquitin E3 ligase, and ubiquitin-specific protease USP7. | Boutell C et al |
| 15247261 | 2004 | A RING finger ubiquitin ligase is protected from autocatalyzed ubiquitination and degradation by binding to ubiquitin-specific protease USP7. | Canning M et al |
| 16596237 | 2006 | HAUSP as a therapeutic target for hematopoietic tumors (review). | Cheon KW et al |
| 9034339 | 1997 | A novel ubiquitin-specific protease is dynamically associated with the PML nuclear domain and binds to a herpesvirus regulatory protein. | Everett RD et al |
| 17651432 | 2007 | Biochemical characterization of USP7 reveals post-translational modification sites and structural requirements for substrate processing and subcellular localization. | Fernández-Montalván A et al |
| 15494000 | 2004 | HAUSP/USP7 as an Epstein-Barr virus target. | Holowaty MN et al |
| 12093161 | 2002 | USP7, a ubiquitin-specific protease, interacts with ataxin-1, the SCA1 gene product. | Hong S et al |
| 16402859 | 2006 | Structural basis of competitive recognition of p53 and MDM2 by HAUSP/USP7: implications for the regulation of the p53-MDM2 pathway. | Hu M et al |
| 12507430 | 2002 | Crystal structure of a UBP-family deubiquitinating enzyme in isolation and in complex with ubiquitin aldehyde. | Hu M et al |
| 17927229 | 2007 | Proteome changes induced by knock-down of the deubiquitylating enzyme HAUSP/USP7. | Kessler BM et al |
| 15053880 | 2004 | A dynamic role of HAUSP in the p53-Mdm2 pathway. | Li M et al |
| 11923872 | 2002 | Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization. | Li M et al |
| 16450335 | 2006 | The HAUSP gene plays an important role in non-small cell lung carcinogenesis through p53-dependent pathways. | Masuya D et al |
| 15916963 | 2005 | Loss of HAUSP-mediated deubiquitination contributes to DNA damage-induced destabilization of Hdmx and Hdm2. | Meulmeester E et al |
| 14982996 | 2004 | Activity-based ubiquitin-specific protease (USP) profiling of virus-infected and malignant human cells. | Ovaa H et al |
| 9925944 | 1998 | Assignment1 of herpesvirus-associated ubiquitin-specific protease gene HAUSP to human chromosome band 16p13.3 by in situ hybridization. | Robinson PA et al |
| 16402389 | 2006 | Activity profiling of deubiquitinating enzymes in cervical carcinoma biopsies and cell lines. | Rolén U et al |
| 15808506 | 2005 | Structure of the p53 binding domain of HAUSP/USP7 bound to Epstein-Barr nuclear antigen 1 implications for EBV-mediated immortalization. | Saridakis V et al |
| 16474402 | 2006 | Molecular recognition of p53 and MDM2 by USP7/HAUSP. | Sheng Y et al |
| 12413694 | 2002 | The ubiquitin-proteasome pathway in thymocyte apoptosis: caspase-dependent processing of the deubiquitinating enzyme USP7 (HAUSP). | Vugmeyster Y et al |
| 15942648 | 2005 | Expression and functional analyses of mHAUSP regulating apoptosis of cervical adenocarcinoma cells. | Yoo KJ et al |
Other Information
Locus ID:
NCBI: 7874
MIM: 602519
HGNC: 12630
Ensembl: ENSG00000187555
Variants:
dbSNP: 7874
ClinVar: 7874
TCGA: ENSG00000187555
COSMIC: USP7
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37867415 | 2024 | USP7 reduces the level of nuclear DICER, impairing DNA damage response and promoting cancer progression. | 1 |
| 37890652 | 2024 | USP7 promotes decidualization of ESCs by STAT3/PR axis during early pregnancy. | 0 |
| 38097136 | 2024 | USP7-mediated ERβ stabilization mitigates ROS accumulation and promotes osimertinib resistance by suppressing PRDX3 SUMOylation in non-small cell lung carcinoma. | 0 |
| 38126281 | 2024 | DNA methylation episignature, extension of the clinical features, and comparative epigenomic profiling of Hao-Fountain syndrome caused by variants in USP7. | 0 |
| 38254206 | 2024 | Stabilization of KPNB1 by deubiquitinase USP7 promotes glioblastoma progression through the YBX1-NLGN3 axis. | 0 |
| 38279869 | 2024 | SP1-activated USP27X-AS1 promotes hepatocellular carcinoma progression via USP7-mediated AKT stabilisation. | 0 |
| 38287116 | 2024 | Deubiquitinase USP7 stabilizes KDM5B and promotes tumor progression and cisplatin resistance in nasopharyngeal carcinoma through the ZBTB16/TOP2A axis. | 2 |
| 38340226 | 2024 | Ubiquitination-specific protease 7 enhances stemness of hepatocellular carcinoma by stabilizing basic transcription factor 3. | 0 |
| 38472728 | 2024 | Backbone (1)H, (13)C and (15)N resonance assignment of the ubiquitin specific protease 7 catalytic domain (residues 208-554) in complex with a small molecule ligand. | 0 |
| 38555840 | 2024 | USP7 interacts with and destabilizes oncoprotein SET. | 0 |
| 38625581 | 2024 | USP7 promotes cervical cancer progression by stabilizing MTDH expression through deubiquitination. | 0 |
| 38702792 | 2024 | The deubiquitinase USP7 and E3 ligase TRIM21 regulate vasculogenic mimicry and malignant progression of RMS by balancing SNAI2 homeostasis. | 0 |
| 38752725 | 2024 | Biologically significant interaction of human herpesvirus 8 viral interferon regulatory factor 4 with ubiquitin-specific protease 7. | 0 |
| 37867415 | 2024 | USP7 reduces the level of nuclear DICER, impairing DNA damage response and promoting cancer progression. | 1 |
| 37890652 | 2024 | USP7 promotes decidualization of ESCs by STAT3/PR axis during early pregnancy. | 0 |
Citation
Kwang-Hyun Baek ; Suresh Ramakrishna
USP7 (ubiquitin specific peptidase 7 (herpes virus-associated))
Atlas Genet Cytogenet Oncol Haematol. 2008-09-01
Online version: http://atlasgeneticsoncology.org/gene/42773/usp7-(ubiquitin-specific-peptidase-7-(herpes-virus-associated))
