NAPA (N-ethylmaleimide-sensitive factor attachment protein, alpha)
2013-09-01 Nayden G Naydenov , Andrei I Ivanov AffiliationDNA/RNA
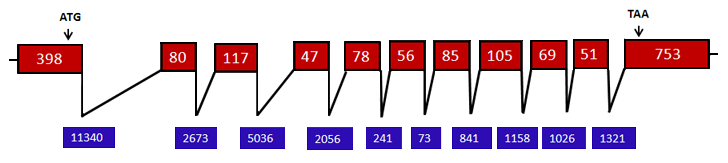
Figure 1. Schematic representation of the genomic structure of human NAPA gene-transcript variant 1, (ENST00000263354). Exons are represented by red boxes while introns appear as waved black lines. The length of exons in (bp) is shown in the red boxes, and that of the introns is presented at the bottom blue boxes (not to scale). The relative position of ATG start and TAA stop codons are indicated with black arrows.
Description
The NAPA gene spans 27,61 kb on chromosome 19q13 and consists of 11 exons and 10 introns (fig. 1) with an open reading frame of 1839 bp. The NAPA promoter has not been functionally explored.
Transcription
The predominant transcript variant (1) of the NAPA gene encodes a functional protein of 295 amino acids. The second transcript of 1521 bp is predicted to encode a 256 amino acid protein. This variant (2) uses an alternate splice site in the 3 terminal exon, compared to variant 1. This variant is represented as non-coding because the use of the alternate splice site renders the resulting transcript a candidate for nonsense-mediated mRNA decay (NMD). The third transcript, variant (3), is 1668 bp. Variant (3) lacks two consecutive internal exons, compared to variant 1. This variant is also represented as non-coding because the use of the 5-most expected translational start codon, as used in variant 1, also makes this transcript a candidate for NMD. Additionally, 2 alternative transcribed variants are predicted to exist via the automatic annotation program, Havana. However, these predicted transcripts need to be experimentally confirmed.
Pseudogene
None discovered.
Proteins
Description
The sequence of the predicted 295-amino acid; 33233 Da human protein encoded by NAPA shares 37%, 60%, and 81% identity with sequences in yeast (Sacharomyces cerevisiae), Drosophila melanogaster, and zebrafish (Danio rerio). NAPA belongs to a protein family comprised of, in higher eukaryotes, three homologues named NAPA, soluble NSF attachment protein beta (NAPB), and soluble NSF attachment protein gamma (NAPG) (Clary et al., 1990). Amino acid sequence comparison of bovine proteins show that NAPA and NAPB are most related to each other with 83% amino acid identity (Whiteheart et al., 1993). In contrast, NAPA and NAPG have only about 25% sequence identity. Structural information is available for the yeast homolog of NAPA, Sec17 (Rice and Brunger, 1999), and zebrafish NAPG (Bitto et al., 2008). Both homologs represent elongated proteins comprised of an extended twisted sheet of α-helical hairpins and a helical-bundle domain at the carboxy-terminal end.
Expression
NAPA is ubiquitously expressed in various mammalian cells and tissues. In mouse tissues, its expression level is highest in the brain, spleen, and testis (Whiteheart et al., 1993). Data from the human gene atlas has shown that NAPA is expressed at the highest level in heart, liver, lung, and placenta. Regulation of NAPA expression remains unexplored. One recent report demonstrated decreased protein levels of NAPA in brain synaptosomes of rats subjected to oxidative stress (Kaneai et al., 2013).
Localisation
In polarized epithelial cells such as alveolar type II cells, T84, and SK-CO15 human colonic epithelial cells NAPA predominantly localizes in the plasma membrane with significant enrichment at the apical junctional complex (Abonyo et al., 2003; Naydenov et al., 2012a). By contrast, in contact-naïve epithelial cells, NAPA accumulates in the perinuclear intracellular compartment that resembles the Golgi complex (Naydenov et al., 2012a).
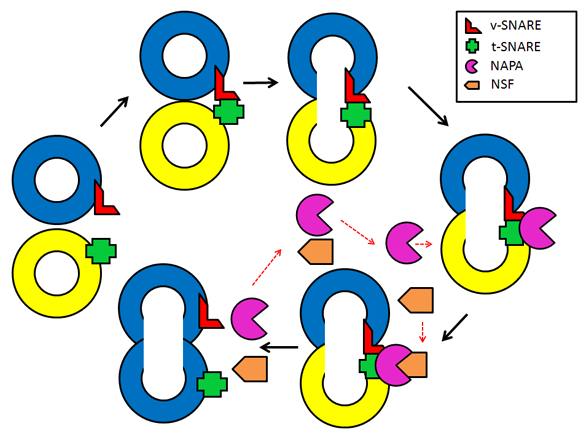
Figure 2. A model depicting vesicle fusion events according to the SNARE hypothesis. During the initial docking step, v-SNARE and t-SNARE bind creating a complex that drives pore formation and fusion of the two opposing membranes. After the fusion step, NAPA is recruited to the cys-SNARE complex allowing NSF binding. Next the ATPase activity of NSF and conformational changes of both NSF and NAPA disassemble the cys-SNARE complex, and release its components for recycling in subsequent fusion events.
Function
One of the most-studied intracellular functions of NAPA involves regulation of NSF-dependent vesicle fusion (Andreeva et al., 2006). According to the so-called "SNARE (soluble NSF attachment protein receptor) hypothesis" vesicle fusion is driven by specific associations of complementary SNARE proteins residing on the vesicle (v-SNAREs) and target (t-SNAREs) membranes (Chen and Scheller, 2001). These proteins form a core SNARE complex consisting of a parallel four-helix bundle. This complex drives the opposing membranes to a close apposition, and subsequently to a complete fusion (fig. 2). Eukaryotic cells use an evolutionarily-conserved mechanism to disassemble and recycle the cys-SNARE complex that is formed after the fusion of two membranes (Barnard et al., 1997). This mechanism involves a hexameric ATPase, NSF, and its adaptor protein, NAPA (Vivona et al., 2013; Wilson et al., 1992; Winter et al., 2009). NAPA interacts with the SNARE complex, recruits NSF, stimulates NSF activity, and transduces a conformational change of NSF to drive SNARE disassembly (fig. 2). NAPA was implicated in the regulation of numerous trafficking/fusion events in different cellular compartments (Andreeva et al., 2006). Examples of this regulation include ER-Golgi transport, intra-Golgi vesicle fusion, trafficking from the trans-Golgi network to the plasma membrane, neuromediator exocytosis, and synaptic vesicle fusion (Barnard et al., 1997; Burgalossi et al., 2010; Clary et al., 1990; Low et al., 1998; Peter et al., 1998; Xu et al., 2002). Interestingly, recent studies discovered several NSF-independent activities of NAPA. These activities involve assembly of epithelial junctions (Andreeva et al., 2005; Naydenov et al., 2012a), suppression of cell apoptosis (Naydenov et al., 2012b; Wu and Chao, 2010), and autophagy (Naydenov et al., 2012c), as well as regulation of store-operated calcium entry (Miao et al., 2013). Such NSF-independent functions are likely to depend on NAPA interactions with different binding partners. For example, NAPA may suppress apoptosis due to its binding to the ER-resident, pro-apoptotic protein BNIP1 (Nakajima et al., 2004). Furthermore, NAPA-dependent regulation of autophagy can be mediated by its ability to interact and dephosphorylate AMP-activated protein kinase (Wang and Brautigan, 2013).
Mutations
Note
A missense G to A transition that leads to a change from methionine to isoleucine (M105I) has been detected in mice (Chae et al., 2004; Hong et al., 2004). This mutation leads to the appearance of the so-called hyh (hydrocephalus with hop gait) phenotype and also impairs acrosomal exocytosis in sperm (Bátiz et al., 2009). Other mutations have not been found associated with this gene potentially because of the multiple roles of NAPA in critical cellular functions which would prevent survival of cells with dysfunctional NAPA mutations.
Implicated in
Entity name
Colorectal cancer
Note
A study utilizing patients with small undifferentiated colorectal cancer revealed a significant increase of NAPA immunoreactivity in cancer cells that correlated with a more aggressive course of the disease (Grabowski et al., 2002).
Entity name
Down syndrome
Note
One study has analyzed expression of NAPA protein in fetal cortex samples of patients with Down Syndrome. The study reported a significant decrease in NAPA expression compared to control samples that correlated with deterioration of the neuronal dendritic tree (Weitzdoerfer et al., 2001). Another study demonstrated a loss of NAPA homolog, NAPB, in brain specimens of adult patients with Down syndrome (Yoo et al., 2001).
Entity name
Huntingtons disease
Note
Marked elevation of NAPA expression was observed by Western blotting of hippocampus samples from patients with Huntingtons diseases as compared to age-matched controls (Morton et al., 2001).
Entity name
Atopic dermatitis
Note
A proteomic analysis of peripheral blood leukocytes demonstrated significant downregulation of NAPA expression in patients with atopic dermatitis as compared to control subjects (Kim et al., 2008). This decrease was validated by Western blotting analysis and NAPA level was suggested as a possible biomarker for the disease.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12663329 | 2003 | Characterization of alpha-soluble N-ethylmaleimide-sensitive fusion attachment protein in alveolar type II cells: implications in lung surfactant secretion. | Abonyo BO et al |
| 16981829 | 2006 | A ubiquitous membrane fusion protein alpha SNAP: a potential therapeutic target for cancer, diabetes and neurological disorders? | Andreeva AV et al |
| 9362506 | 1997 | Stimulation of NSF ATPase activity by alpha-SNAP is required for SNARE complex disassembly and exocytosis. | Barnard RJ et al |
| 19305511 | 2009 | Sperm from hyh mice carrying a point mutation in alphaSNAP have a defect in acrosome reaction. | Bátiz LF et al |
| 17634982 | 2008 | Structure and dynamics of gamma-SNAP: insight into flexibility of proteins from the SNAP family. | Bitto E et al |
| 21040848 | 2010 | SNARE protein recycling by αSNAP and βSNAP supports synaptic vesicle priming. | Burgalossi A et al |
| 14758363 | 2004 | The hyh mutation uncovers roles for alpha Snap in apical protein localization and control of neural cell fate. | Chae TH et al |
| 11252968 | 2001 | SNARE-mediated membrane fusion. | Chen YA et al |
| 2111733 | 1990 | SNAPs, a family of NSF attachment proteins involved in intracellular membrane fusion in animals and yeast. | Clary DO et al |
| 12242522 | 2002 | Expression of neuroendocrine markers: a signature of human undifferentiated carcinoma of the colon and rectum. | Grabowski P et al |
| 14755058 | 2004 | The gene for soluble N-ethylmaleimide sensitive factor attachment protein alpha is mutated in hydrocephaly with hop gait (hyh) mice. | Hong HK et al |
| 23995663 | 2013 | Vitamin E prevents hyperoxia-induced loss of soluble N-ethylmaleimide-sensitive fusion protein attachment protein receptor proteins in the rat neuronal cytoplasm. | Kaneai N et al |
| 18755076 | 2008 | Comparative proteomic analysis of peripheral blood mononuclear cells from atopic dermatitis patients and healthy donors. | Kim WK et al |
| 9647644 | 1998 | The SNARE machinery is involved in apical plasma membrane trafficking in MDCK cells. | Low SH et al |
| 23878724 | 2013 | An essential and NSF independent role for α-SNAP in store-operated calcium entry. | Miao Y et al |
| 11704347 | 2001 | Abnormalities in the synaptic vesicle fusion machinery in Huntington's disease. | Morton AJ et al |
| 15272311 | 2004 | Involvement of BNIP1 in apoptosis and endoplasmic reticulum membrane fusion. | Nakajima K et al |
| 23187805 | 2012 | Loss of a membrane trafficking protein αSNAP induces non-canonical autophagy in human epithelia. | Naydenov NG et al |
| 9701561 | 1998 | Alpha-SNAP but not gamma-SNAP is required for ER-Golgi transport after vesicle budding and the Rab1-requiring step but before the EGTA-sensitive step. | Peter F et al |
| 10445030 | 1999 | Crystal structure of the vesicular transport protein Sec17: implications for SNAP function in SNARE complex disassembly. | Rice LM et al |
| 23836889 | 2013 | Disassembly of all SNARE complexes by N-ethylmaleimide-sensitive factor (NSF) is initiated by a conserved 1:1 interaction between α-soluble NSF attachment protein (SNAP) and SNARE complex. | Vivona S et al |
| 23463002 | 2013 | α-SNAP inhibits AMPK signaling to reduce mitochondrial biogenesis and dephosphorylates Thr172 in AMPKα in vitro. | Wang L et al |
| 11771761 | 2001 | Fetal life in Down syndrome starts with normal neuronal density but impaired dendritic spines and synaptosomal structure. | Weitzdoerfer R et al |
| 8455721 | 1993 | SNAP family of NSF attachment proteins includes a brain-specific isoform. | Whiteheart SW et al |
| 1315316 | 1992 | A multisubunit particle implicated in membrane fusion. | Wilson DW et al |
| 19762473 | 2009 | A conserved membrane attachment site in alpha-SNAP facilitates N-ethylmaleimide-sensitive factor (NSF)-driven SNARE complex disassembly. | Winter U et al |
| 20653109 | 2010 | Knockdown of NAPA using short-hairpin RNA sensitizes cancer cells to cisplatin: implications to overcome chemoresistance. | Wu ZZ et al |
| 11756488 | 2002 | Differential regulation of exocytosis by alpha- and beta-SNAPs. | Xu J et al |
| 11244216 | 2001 | Synaptosomal proteins, beta-soluble N-ethylmaleimide-sensitive factor attachment protein (beta-SNAP), gamma-SNAP and synaptotagmin I in brain of patients with Down syndrome and Alzheimer's disease. | Yoo BC et al |
Other Information
Locus ID:
NCBI: 8775
MIM: 603215
HGNC: 7641
Ensembl: ENSG00000105402
Variants:
dbSNP: 8775
ClinVar: 8775
TCGA: ENSG00000105402
COSMIC: NAPA
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 34653219 | 2021 | High affinity associations with α-SNAP enable calcium entry via Orai1 channels. | 1 |
| 34653219 | 2021 | High affinity associations with α-SNAP enable calcium entry via Orai1 channels. | 1 |
| 26156199 | 2015 | Association of Alpha-Soluble NSF Attachment Protein with Epileptic Seizure. | 3 |
| 26156199 | 2015 | Association of Alpha-Soluble NSF Attachment Protein with Epileptic Seizure. | 3 |
| 24311785 | 2014 | N-ethylmaleimide-sensitive factor attachment protein α (αSNAP) regulates matrix adhesion and integrin processing in human epithelial cells. | 14 |
| 24311785 | 2014 | N-ethylmaleimide-sensitive factor attachment protein α (αSNAP) regulates matrix adhesion and integrin processing in human epithelial cells. | 14 |
| 22194596 | 2012 | Loss of soluble N-ethylmaleimide-sensitive factor attachment protein α (αSNAP) induces epithelial cell apoptosis via down-regulation of Bcl-2 expression and disruption of the Golgi. | 22 |
| 22485163 | 2012 | A membrane fusion protein αSNAP is a novel regulator of epithelial apical junctions. | 22 |
| 23187805 | 2012 | Loss of a membrane trafficking protein αSNAP induces non-canonical autophagy in human epithelia. | 28 |
| 22194596 | 2012 | Loss of soluble N-ethylmaleimide-sensitive factor attachment protein α (αSNAP) induces epithelial cell apoptosis via down-regulation of Bcl-2 expression and disruption of the Golgi. | 22 |
| 22485163 | 2012 | A membrane fusion protein αSNAP is a novel regulator of epithelial apical junctions. | 22 |
| 23187805 | 2012 | Loss of a membrane trafficking protein αSNAP induces non-canonical autophagy in human epithelia. | 28 |
| 21931730 | 2011 | BKV agnoprotein interacts with α-soluble N-ethylmaleimide-sensitive fusion attachment protein, and negatively influences transport of VSVG-EGFP. | 14 |
| 21931730 | 2011 | BKV agnoprotein interacts with α-soluble N-ethylmaleimide-sensitive fusion attachment protein, and negatively influences transport of VSVG-EGFP. | 14 |
| 20089838 | 2010 | Combinational soluble N-ethylmaleimide-sensitive factor attachment protein receptor proteins VAMP8 and Vti1b mediate fusion of antimicrobial and canonical autophagosomes with lysosomes. | 94 |
Citation
Nayden G Naydenov ; Andrei I Ivanov
NAPA (N-ethylmaleimide-sensitive factor attachment protein, alpha)
Atlas Genet Cytogenet Oncol Haematol. 2013-09-01
Online version: http://atlasgeneticsoncology.org/gene/44025/napa-(n-ethylmaleimide-sensitive-factor-attachment-protein-alpha)
