DNA/RNA
Description
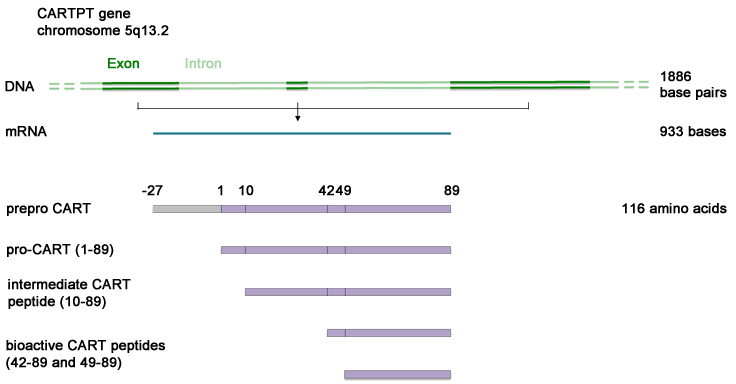
The human CARTPT gene spans 1886 base pairs and includes three exons and two introns (Douglass and Daoud, 1996).
Transcription
Transcription of the 1886 base pair (bp) long CARTPT gene including three exons, yields a 933 bases CARTPT mRNA, which in turn is translated into the 116 amino acid (aa) long prepro-CART peptide. The first 27 amino acid residues serve as a hydrophobic N-terminal signal sequence, which is cleaved, resulting in the 89 aa long pro-CART peptide (Douglass et al., 1995). Pro-CART is subsequently subject to processing by prohormone/proprotein convertases (PCs) during the regulated secretory pathway in the Golgi apparatus, immature secretory granules and finally mature secretory granules (Stein et al., 2006b). PC1/3 is primarily responsible for producing the intermediate peptide 10-89, whereas PC2 and to a lesser extent PC5/6A generate the two bioactive CART peptides (42-89 and 49-89) (Stein et al., 2006a; Stein et al., 2006b). In addition, there are a number of smaller CART peptide fragments of uncertain importance (Kuhar and Yoho, 1999; Dylag et al., 2006; Stein et al., 2006b).
Proteins
Description
Prepro-CART is a 116 aa long peptide with a molecular weight of 12829 Da. As described above, posttranslational modifications yield an 89 aa long pro-CART peptide (28-116) and eventually two bioactive CART peptides (42-89 and 49-89).
Expression
CART peptides have been found in the central, peripheral and enteric nervous systems, and also in endocrine cells in the pancreatic islets (Jensen et al., 1999; Wierup et al., 2004; Wierup and Sundler, 2006), the GI tract mucosa (Ekblad et al., 2003), the thyroid (Wierup et al., 2007), and the adrenal medulla (Dun et al., 2006).
Localisation
CART is localized to the secretory granules of neuroendocrine cells as demonstrated in rat pancreatic β-cells (Wierup et al., 2006).
Function
CART peptide is a brain-gut peptide, acting both as a neurotransmitter and as a hormone. Within the CNS, the spatial distribution of CART peptides together with various experimental studies suggest a role of CART peptides in regulating food intake and body weight (Kuhar et al., 1999; Rogge et al., 2008) with an overall anorexigenic effect via largely unknown mechanisms (Hunter et al., 2004; Rogge et al., 2008). This is also supported by observations that CARTPT null mice and humans carrying a mutated CARTPT gene develop obesity and signs of type 2 diabetes (Asnicar et al., 2001; del Giudice et al., 2001; Guérardel et al., 2005; Wierup et al., 2005). In addition, there is evidence of CART involvement in other brain processes such as mechanisms of reward and stress response (Kuhar et al., 1999; Jaworski et al., 2006; Rogge et al., 2008).
Hormonal expression of CART in pancreatic islets occurs mainly in somatostatin-producing δ-cells (Jensen et al., 1999; Wierup and Sundler, 2006), and the produced CART peptides participate in the regulation of insulin, glucagon, and somatostatin secretion (Wierup and Sundler, 2006; Wierup et al., 2006). Indeed, CART is necessary for normal islet function, since Cart null mice have diminished insulin secretion and reduced glucose elimination (Wierup et al., 2005). Interestingly, there is recent evidence that CART is crucial for pancreatic islet β-cell viability, both by reducing apoptosis and by increasing proliferation via several different pathways (Sathanoori et al., 2013).
Within the GI tract mucosa, CART expression has been identified mainly in gastrin-producing G-cells (Ekblad et al., 2003). The physiological function of CART in the enteric neurons and GI endocrine cells remains poorly elucidated (Ekblad, 2006).
Hormonal expression of CART in pancreatic islets occurs mainly in somatostatin-producing δ-cells (Jensen et al., 1999; Wierup and Sundler, 2006), and the produced CART peptides participate in the regulation of insulin, glucagon, and somatostatin secretion (Wierup and Sundler, 2006; Wierup et al., 2006). Indeed, CART is necessary for normal islet function, since Cart null mice have diminished insulin secretion and reduced glucose elimination (Wierup et al., 2005). Interestingly, there is recent evidence that CART is crucial for pancreatic islet β-cell viability, both by reducing apoptosis and by increasing proliferation via several different pathways (Sathanoori et al., 2013).
Within the GI tract mucosa, CART expression has been identified mainly in gastrin-producing G-cells (Ekblad et al., 2003). The physiological function of CART in the enteric neurons and GI endocrine cells remains poorly elucidated (Ekblad, 2006).
Homology
CART is highly conserved between species. The amino acid sequence is 95% identical between humans, rats and mice (Douglass and Daoud, 1996; Stein et al., 2006b). Goldfish pro-CART peptide is 40-50% homologous to the mammalian counterpart, with 70-80% homology in the biologically active C-terminal portions of the peptide (Volkoff and Peter, 2001).
Mutations
Somatic
A missense mutation resulting in the substitution of Leu with Phe at codon 34, within the NH2-terminal CART region, was identified in an Italian family. Heterozygosity for this mutation was associated with severe obesity within the family and was not found in controls. Resting metabolic rates were lower than expected in subjects with the mutation (del Giudice et al., 2001).
A SNP within the CARTPT gene (SNP-3608T>C (rs7379701/rs4703647)) has been demonstrated to possibly contribute to the genetic risk for obesity (Guérardel et al., 2005).
A SNP within the CARTPT gene (SNP-3608T>C (rs7379701/rs4703647)) has been demonstrated to possibly contribute to the genetic risk for obesity (Guérardel et al., 2005).
Implicated in
Entity name
Neuroendocrine neoplasia (NEN)
Note
CART expression in neoplastic tissue was first demonstrated in rat pancreatic islet tumours (Jensen et al., 1999). CART was later found to co-localize with insulin in human insulin-producing pancreatic neuroendocrine neoplasia (NEN) (Wierup and Sundler, 2006). Next, Bech et al. found raised levels of circulating CART in patients with a wide range of various NENs (Bech et al., 2008). We recently demonstrated presence of CART in tumour tissue of human NENs originating in the stomach, ileum, rectum, pancreas, and thyroid (Landerholm et al., 2011).
Disease
CARTPT is expressed in several types of NENs including gastric NEN, small intestinal NEN (SI-NEN), rectal NEN, pancreatic NEN (P-NEN) and medullary thyroid cancer. CART peptide is not detected in all tumours, and when present levels range from scarce to abundant (Landerholm et al., 2011).
Prognosis
As CART is expressed in all examined types of NEN, it is a tentative diagnostic and/or prognostic biomarker (Landerholm et al., 2011). The only study to date investigating the use of circulating CART found that the presently used Chromogranin A outperforms CART as a general diagnostic NEN biomarker (Bech et al., 2008; Bech et al., 2013). However, the sensitivity of CART was better for P-NENs than other NENs and circulating CART was raised in 95% of progressive P-NENs.
There is also only one study of tumour tissue CART as a biomarker for NENs, demonstrating that increasing CART levels are associated with worse survival in SI-NENs (see below) (Landerholm et al., 2012).
There is also only one study of tumour tissue CART as a biomarker for NENs, demonstrating that increasing CART levels are associated with worse survival in SI-NENs (see below) (Landerholm et al., 2012).
Entity name
Small intestinal neuroendocrine tumours (SI-NETs)
Disease
Neuroendocrine tumours (NETs) correspond to grade 1 and 2 NENs, as opposed to grade 3 neuroendocrine carcinomas (NECs) (Rindi et al., 2007; Sobin et al., 2009). SI-NETs arise from enteroendocrine cells or their precursors in the small intestinal mucosa. At presentation, a majority of SI-NET patients already have metastases to regional mesenteric lymph nodes or to distant sites, in particular the liver. SI-NETs are typically slow-growing and produce and secrete several bioactive substances including serotonin, tachykinins and growth factors. These substances cause several symptoms that are characteristical of SI-NETs; in particular marked fibrosis of the mesentery, diarrhoea, flushing, and tricuspid valvular lesions. CART peptide has been detected in varying levels in SI-NET primary tumours and metastases (Landerholm et al., 2011), but no association was found between tumour CART levels and the abovementioned symptoms (Landerholm et al., 2012).
Prognosis
The only study to investigate CART expression as a prognostic factor in SI-NET found that increasing CART peptide levels in the tumour was independently associated with worse survival (Landerholm et al., 2012).
Oncogenesis
Addition of CART peptide in vitro caused an increase in viability of cell lines GLUTag (murine enteroendocrine cancer cell line) and HCT-116 (human colon cancer cell line) via enhanced proliferation (Landerholm et al., 2012).
Entity name
Breast cancer
Note
CART peptide is expressed in tumour cells in some primary breast cancer tumours and, when present, also in their corresponding lymph node metastases. CART peptide has not been found in normal breast tissue (Brennan et al., 2012).
Prognosis
High CART was an independent poor prognostic factor for overall survival in lymph node negative breast cancer. CART was found to induce transcriptional activity of estrogen receptor-α (ERα) in a ligand-independent manner. In vitro, CART protected breast cancer cells from tamoxifen-mediated cell death and in three breast cancer cohorts high CART expression was predictive of poor outcome in tamoxifen-treated patients (Brennan et al., 2012).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 11564703 | 2001 | Absence of cocaine- and amphetamine-regulated transcript results in obesity in mice fed a high caloric diet. | Asnicar MA et al |
| 18211969 | 2008 | Elevated cocaine- and amphetamine-regulated transcript immunoreactivity in the circulation of patients with neuroendocrine malignancy. | Bech P et al |
| 23939195 | 2014 | The biochemical utility of chromogranin A, chromogranin B and cocaine- and amphetamine-regulated transcript for neuroendocrine neoplasia. | Bech PR et al |
| 22139072 | 2012 | The cocaine- and amphetamine-regulated transcript mediates ligand-independent activation of ERα, and is an independent prognostic factor in node-negative breast cancer. | Brennan DJ et al |
| 8647455 | 1996 | Characterization of the human cDNA and genomic DNA encoding CART: a cocaine- and amphetamine-regulated transcript. | Douglass J et al |
| 16707193 | 2006 | Cocaine- and amphetamine-regulated transcript peptide and sympatho-adrenal axis. | Dun SL et al |
| 16730858 | 2006 | The activity of CART peptide fragments. | Dylag T et al |
| 14507354 | 2003 | Cocaine- and amphetamine-regulated transcript: distribution and function in rat gastrointestinal tract. | Ekblad E et al |
| 16759747 | 2006 | CART in the enteric nervous system. | Ekblad E et al |
| 15823203 | 2005 | Analysis of sequence variability in the CART gene in relation to obesity in a Caucasian population. | Guérardel A et al |
| 15519893 | 2004 | CART in feeding and obesity. | Hunter RG et al |
| 16766084 | 2006 | The role of CART in the reward/reinforcing properties of psychostimulants. | Jaworski JN et al |
| 10214934 | 1999 | The hypothalamic satiety peptide CART is expressed in anorectic and non-anorectic pancreatic islet tumors and in the normal islet of Langerhans. | Jensen PB et al |
| 10370256 | 1999 | CART peptides: novel addiction- and feeding-related neuropeptides. | Kuhar MJ et al |
| 10420164 | 1999 | CART peptide analysis by Western blotting. | Kuhar MJ et al |
| 22553341 | 2012 | Expression of cocaine- and amphetamine-regulated transcript is associated with worse survival in small bowel carcinoid tumors. | Landerholm K et al |
| 21855138 | 2011 | CART peptide stimulation of G protein-mediated signaling in differentiated PC12 cells: identification of PACAP 6-38 as a CART receptor antagonist. | Lin Y et al |
| 17674042 | 2007 | TNM staging of midgut and hindgut (neuro) endocrine tumors: a consensus proposal including a grading system. | Rindi G et al |
| 18802445 | 2008 | CART peptides: regulators of body weight, reward and other functions. | Rogge G et al |
| 23250745 | 2013 | Cocaine- and amphetamine-regulated transcript (CART) protects beta cells against glucotoxicity and increases cell proliferation. | Sathanoori R et al |
| 16800814 | 2006 | RNAi-mediated silencing of prohormone convertase (PC) 5/6 expression leads to impairment in processing of cocaine- and amphetamine-regulated transcript (CART) precursor. | Stein J et al |
| 16784796 | 2006 | Processing of cocaine- and amphetamine-regulated transcript (CART) precursor proteins by prohormone convertases (PCs) and its implications. | Stein J et al |
| 11713200 | 2001 | Characterization of two forms of cocaine- and amphetamine-regulated transcript (CART) peptide precursors in goldfish: molecular cloning and distribution, modulation of expression by nutritional status, and interactions with leptin. | Volkoff H et al |
| 16443761 | 2006 | CART regulates islet hormone secretion and is expressed in the beta-cells of type 2 diabetic rats. | Wierup N et al |
| 17625001 | 2007 | Characterisation of CART-containing neurons and cells in the porcine pancreas, gastro-intestinal tract, adrenal and thyroid glands. | Wierup N et al |
| 14729868 | 2004 | Cocaine- and amphetamine-regulated transcript (CART) is expressed in several islet cell types during rat development. | Wierup N et al |
| 15927717 | 2005 | CART knock out mice have impaired insulin secretion and glucose intolerance, altered beta cell morphology and increased body weight. | Wierup N et al |
| 16842887 | 2006 | CART is a novel islet regulatory peptide. | Wierup N et al |
| 11522684 | 2001 | Mutational screening of the CART gene in obese children: identifying a mutation (Leu34Phe) associated with reduced resting energy expenditure and cosegregating with obesity phenotype in a large family. | del Giudice EM et al |
Other Information
Locus ID:
NCBI: 9607
MIM: 602606
HGNC: 24323
Ensembl: ENSG00000164326
Variants:
dbSNP: 9607
ClinVar: 9607
TCGA: ENSG00000164326
COSMIC: CARTPT
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000164326 | ENST00000296777 | Q16568 |
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37615074 | 2023 | Cocaine and amphetamine regulated transcript (CART): a newly characterized neuropeptide in human prostate. | 0 |
| 37615074 | 2023 | Cocaine and amphetamine regulated transcript (CART): a newly characterized neuropeptide in human prostate. | 0 |
| 36404325 | 2022 | Dietary glycemic index and glycemic load mediate the effect of CARTPT rs2239670 gene polymorphism on metabolic syndrome and metabolic risk factors among adults with obesity. | 0 |
| 36404325 | 2022 | Dietary glycemic index and glycemic load mediate the effect of CARTPT rs2239670 gene polymorphism on metabolic syndrome and metabolic risk factors among adults with obesity. | 0 |
| 33757831 | 2021 | Demystifying functional role of cocaine- and amphetamine-related transcript (CART) peptide in control of energy homeostasis: A twenty-five year expedition. | 10 |
| 34043767 | 2021 | Overcoming Stress, Hunger, and Pain: Cocaine- and Amphetamine-Regulated Transcript Peptide's Promise. | 2 |
| 34876220 | 2021 | Hypothalamic-pituitary hormones will be affected by the interaction between 5q13-14-rs2239670 (CARTPT) gene variants and diet in different obesity phenotypes. | 1 |
| 33757831 | 2021 | Demystifying functional role of cocaine- and amphetamine-related transcript (CART) peptide in control of energy homeostasis: A twenty-five year expedition. | 10 |
| 34043767 | 2021 | Overcoming Stress, Hunger, and Pain: Cocaine- and Amphetamine-Regulated Transcript Peptide's Promise. | 2 |
| 34876220 | 2021 | Hypothalamic-pituitary hormones will be affected by the interaction between 5q13-14-rs2239670 (CARTPT) gene variants and diet in different obesity phenotypes. | 1 |
| 31918705 | 2020 | Cocaine and amphetamine-regulated transcript prepropeptide gene (CARTPT) polymorphism interacts with Diet Quality Index-International (DQI-I) and Healthy Eating Index (HEI) to affect hypothalamic hormones and cardio-metabolic risk factors among obese individuals. | 5 |
| 32902020 | 2020 | Nucleobindin 2/nesfatin-1 expression and colocalisation with neuropeptide Y and cocaine- and amphetamine-regulated transcript in the human brainstem. | 3 |
| 31918705 | 2020 | Cocaine and amphetamine-regulated transcript prepropeptide gene (CARTPT) polymorphism interacts with Diet Quality Index-International (DQI-I) and Healthy Eating Index (HEI) to affect hypothalamic hormones and cardio-metabolic risk factors among obese individuals. | 5 |
| 32902020 | 2020 | Nucleobindin 2/nesfatin-1 expression and colocalisation with neuropeptide Y and cocaine- and amphetamine-regulated transcript in the human brainstem. | 3 |
| 29275211 | 2018 | Up-regulation of POMC and CART mRNAs by intermittent hypoxia via GATA transcription factors in human neuronal cells. | 16 |
Citation
Kalle Landerholm ; Nils Wierup
CARTPT (CART Prepropeptide)
Atlas Genet Cytogenet Oncol Haematol. 2013-11-01
Online version: http://atlasgeneticsoncology.org/gene/52655/cartpt-(cart-prepropeptide)
