CD2AP (CD2 associated protein)
2017-11-01 Simona Panni AffiliationDiBEST Department (Biologia, Ecologia, Scienze della Terra), via Pietro Bucci, Cubo 6C 87036 Arcavacata di Rende, CS (Italy). [email protected]
Identity

Abstract
CD2AP gene encodes for an adaptor protein with high homology to Cin85. It was originally cloned as CD2 interacting protein and, in an independent study, as p130-Cas interacting protein. It contains three SH3 domains, a proline-rich motif and a coiled-coil domain that allow it to form complexes with numerous proteins and to participate in different physiological processes. Several evidences suggest that it links cell surface proteins and specialized junctions with the actin cytoskeleton and that it regulates the assembly of actin filaments to guide cell shape and movements.
DNA/RNA

Description
Transcription
Pseudogene
Proteins
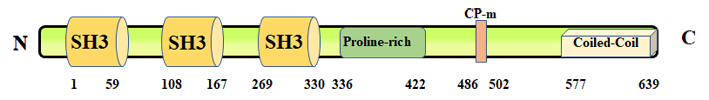
Description
Expression
Localisation
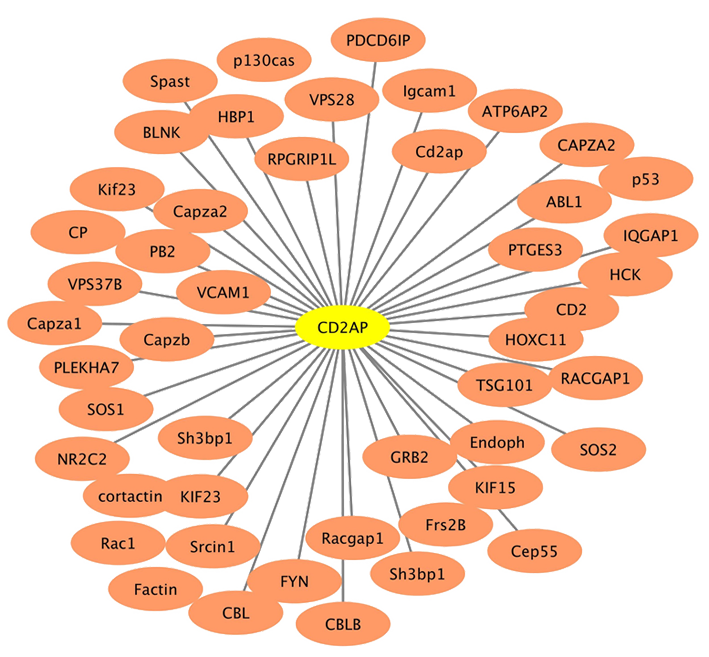
Function

Homology
Mutations
Note
Two mutations of the splicing acceptor site of exon seven, that result in a lower expression of the gene, have been reported from FSGS patients (Kim et al. 2003).
Implicated in
It has been proposed as a promising target for ErbB2 overexpressing tumors since it participates in the inhibition of activated ErbB2 (Mineghishi et al. 2013).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12057195 | 2002 | Accessory protein recruitment motifs in clathrin-mediated endocytosis. | Brett TJ et al |
| 16707503 | 2006 | Identification of a novel inhibitory actin-capping protein binding motif in CD2-associated protein. | Bruck S et al |
| 26156092 | 2015 | Focal segmental glomerulosclerosis: molecular genetics and targeted therapies. | Chen YM et al |
| 26358779 | 2015 | The Alzheimer's disease risk factor CD2AP maintains blood-brain barrier integrity. | Cochran JN et al |
| 12354621 | 2002 | CIN85/CMS family of adaptor molecules. | Dikic I et al |
| 9741631 | 1998 | A novel adaptor protein orchestrates receptor patterning and cytoskeletal polarity in T-cell contacts. | Dustin ML et al |
| 27899578 | 2017 | COSMIC: somatic cancer genetics at high-resolution. | Forbes SA et al |
| 17606992 | 2007 | Structure and function analysis of the CMS/CIN85 protein family identifies actin-bundling properties and heterotypic-complex formation. | Gaidos G et al |
| 21460840 | 2011 | Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer's disease. | Hollingworth P et al |
| 23027549 | 2012 | Interactions between Drosophila IgCAM adhesion receptors and cindr, the Cd2ap/Cin85 ortholog. | Johnson RI et al |
| 24951455 | 2015 | Alzheimer's disease risk genes and mechanisms of disease pathogenesis. | Karch CM et al |
| 12764198 | 2003 | CD2-associated protein haploinsufficiency is linked to glomerular disease susceptibility. | Kim JM et al |
| 11067845 | 2001 | The adapter type protein CMS/CD2AP binds to the proto-oncogenic protein c-Cbl through a tyrosine phosphorylation-regulated Src homology 3 domain interaction. | Kirsch KH et al |
| 12217865 | 2002 | CD2-associated protein directly interacts with the actin cytoskeleton. | Lehtonen S et al |
| 25887956 | 2015 | Effects of CD2-associated protein deficiency on amyloid-β in neuroblastoma cells and in an APP transgenic mouse model. | Liao F et al |
| 17713465 | 2007 | Focal segmental glomerulosclerosis in a patient homozygous for a CD2AP mutation. | Löwik MM et al |
| 12672817 | 2003 | A Cortactin-CD2-associated protein (CD2AP) complex provides a novel link between epidermal growth factor receptor endocytosis and the actin cytoskeleton. | Lynch DK et al |
| 18218851 | 2008 | Novel markers of normal and neoplastic human plasmacytoid dendritic cells. | Marafioti T et al |
| 23279575 | 2013 | Adaptor protein complex of FRS2β and CIN85/CD2AP provides a novel mechanism for ErbB2/HER2 protein downregulation. | Minegishi Y et al |
| 17020880 | 2006 | Atypical polyproline recognition by the CMS N-terminal Src homology 3 domain. | Moncalián G et al |
| 28664408 | 2017 | Focal segmental glomerulosclerosis; why does it occur segmentally? | Nagata M et al |
| 21460841 | 2011 | Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer's disease. | Naj AC et al |
| 24234451 | 2014 | The MIntAct project--IntAct as a common curation platform for 11 molecular interaction databases. | Orchard S et al |
| 25261582 | 2015 | The adapter protein CD2AP binds to p53 protein in the cytoplasm and can discriminate its polymorphic variants P72R. | Panni S et al |
| 25310209 | 2014 | Blastic plasmacytoid dendritic cell neoplasm: update on molecular biology, diagnosis, and therapy. | Riaz W et al |
| 22953501 | 2012 | Expression of the CD2AP adaptor molecule in normal, reactive and neoplastic human tissue. | Rizvi H et al |
| 24829845 | 2014 | Late-Onset Alzheimer's Disease Genes and the Potentially Implicated Pathways. | Rosenthal SL et al |
| 14597658 | 2003 | Cytoscape: a software environment for integrated models of biomolecular interaction networks. | Shannon P et al |
| 11733379 | 2001 | CD2AP localizes to the slit diaphragm and binds to nephrin via a novel C-terminal domain. | Shih NY et al |
| 20625546 | 2010 | Two distinct mechanisms for actin capping protein regulation--steric and allosteric inhibition. | Takeda S et al |
| 24322428 | 2013 | FSGS3/CD2AP is a barbed-end capping protein that stabilizes actin and strengthens adherens junctions. | Tang VW et al |
| 17188587 | 2007 | Solution structure of the second SH3 domain of human CMS and a newly identified binding site at the C-terminus of c-Cbl. | Yao B et al |
| 11912254 | 2002 | Nephrin dissociates from actin, and its expression is reduced in early experimental membranous nephropathy. | Yuan H et al |
| 23090967 | 2013 | CD2AP links cortactin and capping protein at the cell periphery to facilitate formation of lamellipodia. | Zhao J et al |
| 20404345 | 2010 | Rac1 recruits the adapter protein CMS/CD2AP to cell-cell contacts. | van Duijn TJ et al |
Citation
Simona Panni
CD2AP (CD2 associated protein)
Atlas Genet Cytogenet Oncol Haematol. 2017-11-01
Online version: http://atlasgeneticsoncology.org/gene/54896/cd2ap-(cd2-associated-protein)
