CRK (v-crk sarcoma virus CT10 oncogene homolog (avian))
2012-03-01 Shinya Tanaka AffiliationLaboratory of Cancer Research, Department of Pathology, Hokkaido University Graduate School of Medicine, Sapporo, Japan
DNA/RNA
Note
Crk belongs to an adaptor family of protein mostly composed of SH2 (src homology 2) and SH3 domains.
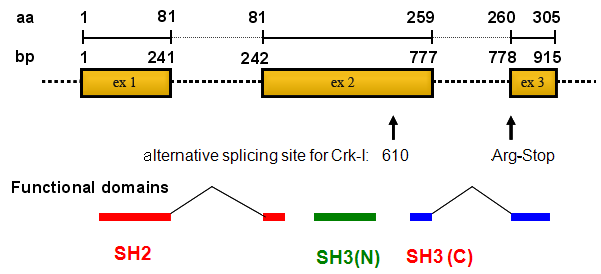
Crk-II gene comprises of three exons spanning on chromosome 17p13.3.
Description
The human genomic DNA sequence contains 3 exons.
Transcription
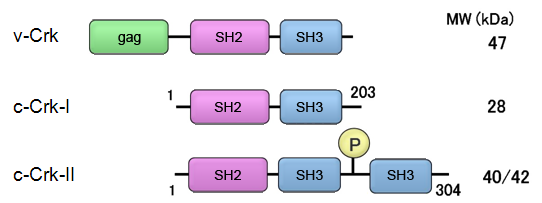
The length of open reading frame of transcript is 912 mer and translated to a 304 residues protein referred as c-Crk-II. By alternative splicing, 610-778 were deleted and truncated protein encoding 203 amino acids protein as c-Crk-I was generated. Recently, Crk-III which comprises 283 amino acids was reported.
Pseudogene
Not identified.
Proteins
Note
Crk was originally isolated as avian retrovirus (CT10, chicken tumor No. 10) encoding oncoprotein as v-Crk (viral Crk, CT10 regulator of kinase).
Crk-II is composed of SH2-SH3(N)-SH3(C) and alternative splicing product Crk-I which deleted SH3(C) as SH2-SH3(N).
Crk-II is composed of SH2-SH3(N)-SH3(C) and alternative splicing product Crk-I which deleted SH3(C) as SH2-SH3(N).
Tyrosine Y221 (human) of CrkII can be tyrosine phosphorylated and SH2 domain of CrkII binds to pY221 intramolecularly.
Description
NMR analysis of Crk-I, Crk-II, and phosphorylated form of Crk-II (aa 1-231). Crk-I has flexible structure of the linker region, thus individual SH2 or SH3 domain freely access to the targets (data not shown). Unphosphorylated form of Crk-II exhibits compact structure, and each SH domains were relatively fixed around inter SH3 region (designated as ISC: inter SH3 core; yellow in the figure below). In this form, interaction surface of SH3(N) is in the semi-closed status. Upon tyrosine phosphorylation of Y221, SH2 binds to pY221 and structure is dramatically altered and SH3(N) surface is completely blocked by internal structure. Thus, pCrk-II is signaling-OFF form.
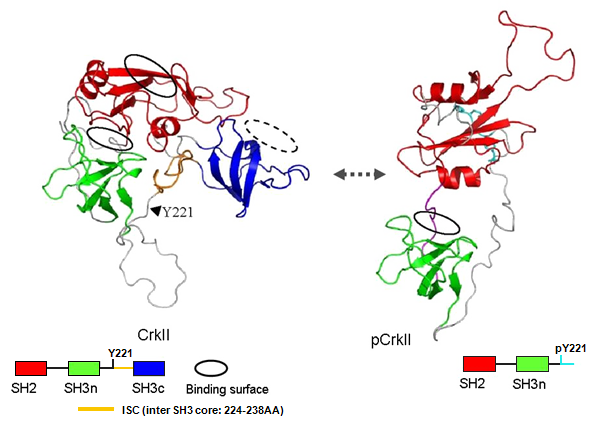
Structure of signaling adaptor protein CrkII. Adapted from Kobashigawa et al. (2007).
Expression
Ubiquitous.
Localisation
Mostly cytoplasm.
Function
Signaling adaptor protein which links tyrosine kinases and small G proteins.
Crk transmits signals from extracellular stimuli such as growth factors and extracellular matrices. SH2 domain of Crk bound to tyrosine phosphorylated peptide motif as YXXP. In focal adhesion complex, Crk binds to p130Cas which possesses six Crk binding consensus sequences and to paxillin. Crk activates Fak and transmit signals to PI3 kinase. Crk also binds to growth factor receptors such as EGFR or FGF, in addition to downstream signaling scaffold protein Gab1 which is mainly activated by HGF. CRK transmits signals to small G proteins through its SH3 domain binding target C3G and DOCK180 those can activate Rap/R-Ras and Rac, respectively, and regulates cell growth, adhesion, and motility.
Crk plays various roles including cell growth, differentiation, phagocytosis, glucose transport, and infectious diseases.
Crk transmits signals from extracellular stimuli such as growth factors and extracellular matrices. SH2 domain of Crk bound to tyrosine phosphorylated peptide motif as YXXP. In focal adhesion complex, Crk binds to p130Cas which possesses six Crk binding consensus sequences and to paxillin. Crk activates Fak and transmit signals to PI3 kinase. Crk also binds to growth factor receptors such as EGFR or FGF, in addition to downstream signaling scaffold protein Gab1 which is mainly activated by HGF. CRK transmits signals to small G proteins through its SH3 domain binding target C3G and DOCK180 those can activate Rap/R-Ras and Rac, respectively, and regulates cell growth, adhesion, and motility.
Crk plays various roles including cell growth, differentiation, phagocytosis, glucose transport, and infectious diseases.
Homology
CrkL (Crk-like).
Implicated in
Entity name
Various diseases
Note
Crk is overexpressed in various human cancers especially in lung adenocarcinoma and in brain tumor. Crk knockdown study demonstrates the essential roles for Crk in malignant potentials of various human cancers including ovarian cancer, sarcoma, and brain tumor. Recently, microRNA-126 was shown to suppress lung cancer growth by the suppression of Crk expression. Crk has been reported to bind to NS1 protein of influenza virus type H5N1, CagA protein of Helicobacter pylori, and also known to regulate the infection of Pseudomonas aeruginosa and Shigella flexneri. Therefore, Crk can be a therapeutic target molecule for cancer and infectious diseases.
Entity name
Lung cancer
Note
96 cases of lung adenocarcinoma were analyzed by using Gene chip and in the cases of stage III, mRNA levels of Crk were increased comparing to those of in stage I (Beer et al., 2002).
Affymetrix oligonucleotide arrays were used to analyze 86 lung adenocarcinomas and 10 uninvolved lung tissues, and Crk mRNA expression was increased in more advanced (stage III versus stage I), larger (T2-4 versus T1), and poorly differentiated tumors and in tumors from patients demonstrating poor survival (Miller et al., 2003).
Immunohistochemical analysis of Crk-II demonstrated that the levels of Crk-II were significantly elevated in most of the tumors, particularly in the colon and lung cancers. Furthermore, immunoblot analysis using human lung cancer cell lines revealed that the expression levels of Crk-II were correlated to growth rates of cells (Nishihara et al., 2002).
Affymetrix oligonucleotide arrays were used to analyze 86 lung adenocarcinomas and 10 uninvolved lung tissues, and Crk mRNA expression was increased in more advanced (stage III versus stage I), larger (T2-4 versus T1), and poorly differentiated tumors and in tumors from patients demonstrating poor survival (Miller et al., 2003).
Immunohistochemical analysis of Crk-II demonstrated that the levels of Crk-II were significantly elevated in most of the tumors, particularly in the colon and lung cancers. Furthermore, immunoblot analysis using human lung cancer cell lines revealed that the expression levels of Crk-II were correlated to growth rates of cells (Nishihara et al., 2002).
Prognosis
Expression of Crk mRNA was increased in patients with poor prognosis (P=0.00034) (Miller et al., 2003).
Entity name
Brain tumor
Note
CrkII mRNA was detected both in normal brain and glioblastoma tissues, whereas crkI mRNA levels were quite low in normal brain and up-regulated in glioblastoma tissues. Expression of CrkI but not CrkII in glioblastoma U87MG cells induced transformation that stimulated cell migration and invasion concomitant with tyrosine phosphorylation of p130 Cas (Takino et al., 2003).
Crk-knockdown cell lines of glioblastoma KMG4 was established by siRNA, and early phase of cell adhesion to laminin was found to be suppressed. Wound healing assay revealed the decreased cell motility in Crk knockdown cells, and suppression of both anchorage-dependent and -independent growth were demonstrated in these cells. Furthermore, in vivo tumor forming potential was also markedly suppressed (Wang et al., 2007).
Crk-knockdown cell lines of glioblastoma KMG4 was established by siRNA, and early phase of cell adhesion to laminin was found to be suppressed. Wound healing assay revealed the decreased cell motility in Crk knockdown cells, and suppression of both anchorage-dependent and -independent growth were demonstrated in these cells. Furthermore, in vivo tumor forming potential was also markedly suppressed (Wang et al., 2007).
Entity name
Ovarian cancer
Note
Crk expression was targeted in the human ovarian cancer cell line MCAS through RNA interference, resulting in disorganized actin fibers, reduced number of focal adhesions, and decreased Rac activity in association with suppression of cell motility and invasion, and anchorage-dependent growth in soft agar. Tumor forming potential in nude mice was attenuated and intraperitoneal dissemination was not observed when Crk knockdown cells were injected into the peritoneal cavity (Linghu et al., 2006).
Entity name
Breast cancer
Note
CrkI/II knockdown resulted in a significant decrease in migration and invasion of multiple malignant breast and other human cancer cell lines (MDA-231, MDA-435s, H1299, KB, and HeLa). Moreover, CrkI/II knockdown decreased cell spreading on extracellular matrix and led to a decrease in actin stress fibers and the formation of mature focal adhesions. Using immunohistochemistry, we show elevated CrkI/II protein levels in patients with breast adenocarcinoma (Rodrigues et al., 2005).
Entity name
Note
Crk adaptor protein is required for the sustained phosphorylation of Gab1 in response to HGF, leading to the enhanced cell motility of human synovial sarcoma cell lines SYO-1, HS-SY-II, and Fuji. Crk knockdown by RNA interference disturbed this HGF-induced tyrosine phosphorylation of Gab1. The elimination of Crk in these cells induced the disorganization of actin cytoskeleton and complete abolishment of HGF-mediated Rac1 activation and cell motility. Furthermore, the depletion of Crk remarkably inhibited the tumor formation and its invasive growth in vivo (Watanabe et al., 2006).
Entity name
Oral cancer
Note
The expression of CRKII in 71 primary oral squamous cell carcinomas and 10 normal oral mucosal specimens was determined immunohistochemically, and the correlation of CRKII overexpression with clinicopathological factors was evaluated. Overexpression of CRKII was detected in 41 of 70 oral squamous cell carcinomas, the frequency being more significant than in normal oral mucosa. In addition, CRKII overexpression was more frequent in higher-grade cancers according to the T classification, N classification, and invasive pattern. Moreover, RNAi-mediated suppression of CRKII expression reduced the migration and invasion potential of an oral squamous cell carcinoma cell line, OSC20 (Yamada et al., 2011).
Entity name
Endometriosis
Note
The expression levels of miR-126 and and its putative target Crk mRNA were quantified using real time PCR in ectopic endometrium and eutopic endometrium in patients with endometriosis. The expression level of miR-126 was significantly downregulated in ectopic endometrium whereas the protein levels of Crk was increased. Thus, miR-126 may play an initial role in the development and progression of endometriosis (Liu et al., 2012).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18165234 | 2008 | Avian and 1918 Spanish influenza a virus NS1 proteins bind to Crk/CrkL Src homology 3 domains to activate host cell signaling. | Heikkinen LS et al |
| 10852971 | 2000 | v-Crk activates the phosphoinositide 3-kinase/AKT pathway in transformation. | Akagi T et al |
| 12118244 | 2002 | Gene-expression profiles predict survival of patients with lung adenocarcinoma. | Beer DG et al |
| 8194526 | 1994 | c-Abl kinase regulates the protein binding activity of c-Crk. | Feller SM et al |
| 11607838 | 2001 | Crk family adaptors-signalling complex formation and biological roles. | Feller SM et al |
| 15598735 | 2004 | CrkII regulates focal adhesion kinase activation by making a complex with Crk-associated substrate, p130Cas. | Iwahara T et al |
| 9808620 | 1998 | Activation of Rac1 by a Crk SH3-binding protein, DOCK180. | Kiyokawa E et al |
| 17515907 | 2007 | Structural basis for the transforming activity of human cancer-related signaling adaptor protein CRK. | Kobashigawa Y et al |
| 16491127 | 2006 | Involvement of adaptor protein Crk in malignant feature of human ovarian cancer cell line MCAS. | Linghu H et al |
| 22012249 | 2012 | Expression of miR-126 and Crk in endometriosis: miR-126 may affect the progression of endometriosis by regulating Crk expression. | Liu S et al |
| 16495483 | 2006 | Elmo1 inhibits ubiquitylation of Dock180. | Makino Y et al |
| 1630456 | 1992 | Two species of human CRK cDNA encode proteins with distinct biological activities. | Matsuda M et al |
| 2450282 | 1988 | A novel viral oncogene with structural similarity to phospholipase C. | Mayer BJ et al |
| 12970743 | 2003 | Increased C-CRK proto-oncogene expression is associated with an aggressive phenotype in lung adenocarcinomas. | Miller CT et al |
| 11911970 | 2002 | Molecular and immunohistochemical analysis of signaling adaptor protein Crk in human cancers. | Nishihara H et al |
| 12894221 | 2003 | CrkIII: a novel and biologically distinct member of the Crk family of adaptor proteins. | Prosser S et al |
| 10850658 | 2000 | Sustained formation of focal adhesions with paxillin in morphological differentiation of PC12 cells. | Rhee S et al |
| 15831672 | 2005 | CrkI and CrkII function as key signaling integrators for migration and invasion of cancer cells. | Rodrigues SP et al |
| 17289588 | 2007 | Proline cis-trans isomerization controls autoinhibition of a signaling protein. | Sarkar P et al |
| 7537852 | 1995 | pp125FAK-dependent tyrosine phosphorylation of paxillin creates a high-affinity binding site for Crk. | Schaller MD et al |
| 9627109 | 1998 | Crk protein binds to PDGF receptor and insulin receptor substrate-1 with different modulating effects on PDGF- and insulin-dependent signaling pathways. | Sorokin A et al |
| 12727859 | 2003 | CrkI adapter protein modulates cell migration and invasion in glioblastoma. | Takino T et al |
| 8321240 | 1993 | Both the SH2 and SH3 domains of human CRK protein are required for neuronal differentiation of PC12 cells. | Tanaka S et al |
| 7512734 | 1994 | C3G, a guanine nucleotide-releasing protein expressed ubiquitously, binds to the Src homology 3 domains of CRK and GRB2/ASH proteins. | Tanaka S et al |
| 9122199 | 1997 | Downstream of Crk adaptor signaling pathway: activation of Jun kinase by v-Crk through the guanine nucleotide exchange protein C3G. | Tanaka S et al |
| 11959814 | 2002 | Signaling adaptor protein v-Crk activates Rho and regulates cell motility in 3Y1 rat fibroblast cell line. | Tsuda M et al |
| 17825249 | 2007 | Signaling adaptor protein Crk is indispensable for malignant feature of glioblastoma cell line KMG4. | Wang L et al |
| 16849525 | 2006 | Adaptor molecule Crk is required for sustained phosphorylation of Grb2-associated binder 1 and hepatocyte growth factor-induced cell motility of human synovial sarcoma cell lines. | Watanabe T et al |
| 21339045 | 2011 | Overexpression of CRKII increases migration and invasive potential in oral squamous cell carcinoma. | Yamada S et al |
| 8361759 | 1993 | Isolation and chromosomal localization of CRKL, a human crk-like gene. | ten Hoeve J et al |
Other Information
Locus ID:
NCBI: 1398
MIM: 164762
HGNC: 2362
Ensembl: ENSG00000167193
Variants:
dbSNP: 1398
ClinVar: 1398
TCGA: ENSG00000167193
COSMIC: CRK
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA134941638 | FGFR1OP | Gene | Pathway | associated | 20124951 | ||
| PA134972108 | FGFR1OP2 | Gene | Pathway | associated | 20124951 | ||
| PA28127 | FGFR1 | Gene | Pathway | associated | 20124951 | ||
| PA28128 | FGFR2 | Gene | Pathway | associated | 20124951 | ||
| PA28129 | FGFR3 | Gene | Pathway | associated | 20124951 | ||
| PA28130 | FGFR4 | Gene | Pathway | associated | 20124951 | ||
| PA28131 | FGFR3P1 | Gene | Pathway | associated | 20124951 | ||
| PA28132 | FGFRL1 | Gene | Pathway | associated | 20124951 | ||
| PA29444 | HRAS | Gene | Pathway | associated | 20124951 | ||
| PA30196 | KRAS | Gene | Pathway | associated | 20124951 | ||
| PA31768 | NRAS | Gene | Pathway | associated | 20124951 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38683131 | 2024 | CRKL but not CRKII contributes to hemin-induced erythroid differentiation of CML. | 0 |
| 38683131 | 2024 | CRKL but not CRKII contributes to hemin-induced erythroid differentiation of CML. | 0 |
| 36737758 | 2023 | Crk proteins activate the Rap1 guanine nucleotide exchange factor C3G by segregated adaptor-dependent and -independent mechanisms. | 3 |
| 37085900 | 2023 | Gab2 plays a carcinogenic role in ovarian cancer by regulating CrkII. | 0 |
| 36737758 | 2023 | Crk proteins activate the Rap1 guanine nucleotide exchange factor C3G by segregated adaptor-dependent and -independent mechanisms. | 3 |
| 37085900 | 2023 | Gab2 plays a carcinogenic role in ovarian cancer by regulating CrkII. | 0 |
| 33026665 | 2021 | Prenatal diagnosis of Miller-Dieker syndrome by chromosomal microarray. | 1 |
| 33561443 | 2021 | Quantitative assessment of glioblastoma phenotypes in vitro establishes cell migration as a robust readout of Crk and CrkL activity. | 1 |
| 34384283 | 2021 | MicroRNA-215-5p Inhibits the Proliferation and Migration of Wilm's Tumor Cells by Targeting CRK. | 0 |
| 33026665 | 2021 | Prenatal diagnosis of Miller-Dieker syndrome by chromosomal microarray. | 1 |
| 33561443 | 2021 | Quantitative assessment of glioblastoma phenotypes in vitro establishes cell migration as a robust readout of Crk and CrkL activity. | 1 |
| 34384283 | 2021 | MicroRNA-215-5p Inhibits the Proliferation and Migration of Wilm's Tumor Cells by Targeting CRK. | 0 |
| 32005519 | 2020 | Signaling adaptor protein Crk is involved in malignant feature of pancreatic cancer associated with phosphorylation of c-Met. | 4 |
| 32147820 | 2020 | Novel role for CRK adaptor proteins as essential components of SRC/FAK signaling for epithelial-mesenchymal transition and colorectal cancer aggressiveness. | 9 |
| 32321766 | 2020 | Cyclophilin A Inhibitor Debio-025 Targets Crk, Reduces Metastasis, and Induces Tumor Immunogenicity in Breast Cancer. | 11 |
Citation
Shinya Tanaka
CRK (v-crk sarcoma virus CT10 oncogene homolog (avian))
Atlas Genet Cytogenet Oncol Haematol. 2012-03-01
Online version: http://atlasgeneticsoncology.org/gene/40149/crk
