PAEP (progestagen-associated endometrial protein)
2010-11-01 Hannu Koistinen , Markku Seppälä AffiliationDepartment of Clinical Chemistry, Helsinki University Central Hospital, University of Helsinki, Helsinki, Finland
Identity
HGNC
LOCATION
9q34.3
IMAGE

LEGEND
Chromosomal location and gene structure of PAEP. Promoter region shows some of the potential regulatory elements. After translation- initiating codon (ATG) exons of the major transcript are shown in black. Some splicing variants contain also parts outside of these exons. PRE: glucocorticoid/progesterone response element; CRE: cAMP responsive element; Sp1: Sp1 transcription factor binding site; AP-1: activator protein-1 element.
LOCUSID
ALIAS
GD,GdA,GdF,GdS,PAEG,PEP,PP14,ZIF-1
FUSION GENES
DNA/RNA
Note
Many other lipocalin genes have similar exon/intron organization.
Description
Maps to chromosome 9: 138453602-138458801 on forward (plus) strand (5200 bases). Gene consists of 7 exons. Promoter region contains, by sequence similarity, 2 forward and two reverse Sp1-like binding sites, four putative glucocorticoid/progesterone response elements (PREs), cAMP responsive element (CRE) and activator protein-1 (AP-1) element.
Transcription
PAEP mRNA (NM_001018049) has 857 bp. Several alternatively spliced mRNA forms have been described, but for most of these evidence for the corresponding protein lacks. Alternative Splicing and Transcript Diversity database (ASTD) reports 16 different transcripts.
Pseudogene
Not known.
Proteins
Note
Some of the localization studies have employed antibodies, the specificity of which is questionable. Some of the biological studies have utilized short peptides derived from PAEP sequence. It is unclear whether such peptides are present in vivo. Glycosylation plays an important part in modulating/dictating the activity of PAEP. In the literature, PAEP is widely referred to as PP14 and glycodelin.
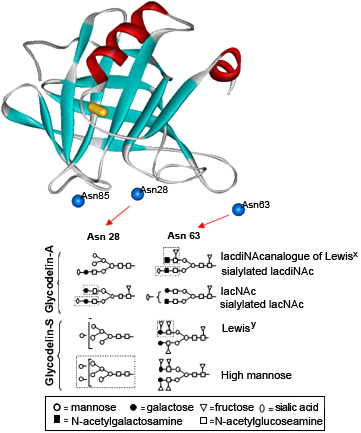
Swiss model-deduced tertiary structure of the PAEP monomer. The S-S bridge is shown as cylinder and side chain nitrogen atoms of asparagines of potential glycosylation sites are shown as balls. Below are representative examples of the major complex-type glycans present at the N-glycosylation sites Asn 28 and Asn 63 of amniotic fluid glycodelin-isoform (glycodelin-A) and seminal plasma glycodelin-isoform (glycodelin-S). Some of the characteristic epitopes are marked by broken line.
Description
PAEP (180 amino acids, of which 18 corresponds to signal sequence) is a 28 kDa secreted glycoprotein, belonging to the kernel lipocalin family. Most family members share three conserved sequence motifs. Although sequence similarity between the family members is low, their three dimensional structures are similar. Lipocalins are small extracellular proteins, many of which bind small hydrophobic molecules, such as retinol and steroids. There is no evidence that PAEP exhibits similar binding properties. PAEP is a glycoprotein with three potential glycosylation sites. Two of them are glycosylated. Many differentially glycosylated forms have been characterized in these sites. Glycosylation modulates/dictates the biological activity of PAEP. Some of the alternatively spliced mRNAs lack the sequences encoding glycosylation sites and/or the lipocalin signature sequence.
Expression
The expression of PAEP is highly regulated in a spatiotemporal fashion. In the female, PAEP is mainly expressed in secretory/decidualized endometrial glands after progesterone exposure. In secretory endometrium, expression becomes detectable four days after ovulation and reaches maximum at the end of the menstrual cycle unless pregnancy ensues. PAEP is one of the major proteins in endometrial secretions. In the male, the highest expression has been reported in seminal vesicles. PAEP is also expressed in other epithelial cells of reproductive tissues, such as fallopian tubes, ovary and the breast. In addition, other secretory epithelia, such as eccrine sweat glands and the bronchus epithelium express PAEP. It is also expressed in differentiated areas of breast cancer, ovarian tumors, endometrial adenocarcinoma, and synovial sarcoma. In addition to epithelial tissues, PAEP has been found in megakaryocytes and erythroid precursor cells. Experimental evidence suggests that PAEP expression is regulated by progesterone/progestins, relaxin, and histone deacetylase inhibitors.
Localisation
PAEP is mostly found in exocrine epithelial cells, from which it is secreted into the gland lumen. In breast cancer, PAEP has been found also in paranuclear vacuoles of lobular carcinoma cells.
Function
PAEP/PP14/glycodelin regulates the functions of spermatozoa during fertilization in a glycosylation dependent manner. The various glycoforms of PAEP have different, sometimes even opposite, biological actions at different phases of the fertilization process. Seminal fluid glycodelin-S binds to the sperm head and inhibits premature capacitation. In the female reproductive tract, spermatozoa come into contact with various PAEP glycoforms, that modulate sperm function, e.g., by preventing premature, progesterone-induced acrosome reaction (glycodelin-F). Glycodelin-A inhibits binding of spermatozoa to the zona pellucida, whereas another glycoform (glycodelin-C) stimulates the same. All these actions are glycosylation-dependent.
PAEP also regulates immune cell functions, which too are, at least in part, regulated by glycosylation. Different PAEP glycoforms contain diverse bi-, tri-, and tetra-antennary complex-type glycans with varying levels of fucose and sialic acid substitution. Glycodelin-A and -F are the most heavily sialylated and inhibit cell proliferation, induce cell death, and suppress interleukin-2 secretion of Jurkat cells and peripheral blood mononuclear cells. No such immunosuppressive effect has been observed for glycodelin-C and -S carrying less or no sialic acids, or for desialylated glycodelin-A and -F. By its immunosuppressive properties one of the PAEP glycoforms (glycodelin-A) may contribute to immunotolerance at the fetomaternal interface and prevent rejection of the fetal semi-allograft.
In early pregnancy, glycodelin-A restrains inappropriate invasion of extravillous cytotrophoblasts by suppressing activity of some key metalloproteinases. In breast and endometrial cancer cell lines, PAEP has been found to revert the malignant phenotype in vitro by inducing morphological differentiation and specific gene expression changes. In a preclinical mouse model, transgenic PAEP expression in breast cancer cells has reduced tumor growth.
PAEP also regulates immune cell functions, which too are, at least in part, regulated by glycosylation. Different PAEP glycoforms contain diverse bi-, tri-, and tetra-antennary complex-type glycans with varying levels of fucose and sialic acid substitution. Glycodelin-A and -F are the most heavily sialylated and inhibit cell proliferation, induce cell death, and suppress interleukin-2 secretion of Jurkat cells and peripheral blood mononuclear cells. No such immunosuppressive effect has been observed for glycodelin-C and -S carrying less or no sialic acids, or for desialylated glycodelin-A and -F. By its immunosuppressive properties one of the PAEP glycoforms (glycodelin-A) may contribute to immunotolerance at the fetomaternal interface and prevent rejection of the fetal semi-allograft.
In early pregnancy, glycodelin-A restrains inappropriate invasion of extravillous cytotrophoblasts by suppressing activity of some key metalloproteinases. In breast and endometrial cancer cell lines, PAEP has been found to revert the malignant phenotype in vitro by inducing morphological differentiation and specific gene expression changes. In a preclinical mouse model, transgenic PAEP expression in breast cancer cells has reduced tumor growth.
Homology
Most lipocalins do not share high sequence similarity, but they are likely to be homologous.
Functional PAEP gene has been found in higher primates. Beta-lactoglobulins represent orthologs of PAEP, but they are likely to be functionally different from human PAEP, not least because of their differences in glycosylation. No convincing evidence of a PAEP ortholog in mouse or rat has been reported.
Functional PAEP gene has been found in higher primates. Beta-lactoglobulins represent orthologs of PAEP, but they are likely to be functionally different from human PAEP, not least because of their differences in glycosylation. No convincing evidence of a PAEP ortholog in mouse or rat has been reported.
Mutations
Note
NCBI SNP database reports 128 PAEP SNPs (Homo sapiens, 13 September 2010). Also HinfI restriction enzyme polymorphism has been reported in Finnish population with 5% frequency for allele A1 and 95% frequency for allele A2. No disease associations for mutations have been described.
Implicated in
Entity name
Ovarian carcinoma
Note
PAEP is expressed in both normal and malignant ovarian tissue. PAEP has been localized to the cytoplasm of tumor cells and its staining is more frequent in well-differentiated than in poorly differentiated carcinomas. Nuclear progesterone receptors (PRA and PRB) are often coexpressed with cytoplasmic PAEP.
Disease
In 2002, ovarian cancer was the 6th most common cancer in women, and 7th most common cause of cancer death. Most malignant neoplasms of the ovary originate from the coelomic epithelium.
Prognosis
In ovarian serous carcinoma, PAEP expression is associated with a more favorable prognosis, even in patients with the same tumor grade and clinical stage.
Entity name
Breast cancer
Note
In breast cancer tissue, PAEP staining has been found in both estrogen and progesterone receptor negative and positive cancers. PAEP is also present in normal breast tissue. Transfection of PAEP in MCF-7 breast cancer cells reverted the malignant phenotype of the cells by inducing morphological differentiation and specific gene expression changes. Furthermore, these cells showed reduced tumor growth in a preclinical xenograft tumor mouse model.
Disease
Breast cancer is the most common cancer among women worldwide. Although the prognosis has improved following improved diagnosis and therapies, breast cancer remains an important cause of death among women. Most of the neoplasms of the breast originate from the ductal epithelium, while a minority originates from the lobular epithelium. Family history of breast cancer is associated with a 2-3-fold higher risk of the disease.
Prognosis
Entity name
Reproductive failure
Note
During the period of endometrial receptivity for implantation, reduced PAEP secretion/serum levels have been observed in reproductive failure, e.g. in unexplained infertility or recurrent early pregnancy loss.
Disease
Unexplained infertility or recurrent miscarriage may result from inadequate implantation and/or placentation.
Entity name
Polycystic ovary syndrome (PCOS)
Note
Pregnant women with PCOS who subsequently miscarry show subnormal rise of PAEP serum concentration during the first trimester.
Disease
PCOS is a common endocrine disorder in fertile-aged women. It is associated with ovulatory disturbance, insulin resistance and androgen excess, and is a frequent cause of menstrual disorders and infertility in women.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 11058742 | 2000 | Lipocalins: unity in diversity. | Akerstrom B et al |
| 19896207 | 2009 | The role of glycodelin as an immune-modulating agent at the feto-maternal interface. | Alok A et al |
| 2881133 | 1987 | Identification of placental protein 14 as an immunosuppressive factor in human reproduction. | Bolton AE et al |
| 17192260 | 2007 | Cumulus oophorus-associated glycodelin-C displaces sperm-bound glycodelin-A and -F and stimulates spermatozoa-zona pellucida binding. | Chiu PC et al |
| 19625311 | 2009 | Cumulus-associated alpha2-macroglobulin derivative retains proconceptive glycodelin-C in the human cumulus matrix. | Chung MK et al |
| 8671249 | 1996 | A role for glycoconjugates in human development: the human feto-embryonic defence system hypothesis. | Clark GF et al |
| 7592613 | 1995 | Structural analysis of the oligosaccharides derived from glycodelin, a human glycoprotein with potent immunosuppressive and contraceptive activities. | Dell A et al |
| 10051679 | 1999 | Modulation of the baboon (Papio anubis) uterine endometrium by chorionic gonadotrophin during the period of uterine receptivity. | Fazleabas AT et al |
| 11058743 | 2000 | The lipocalin protein family: structural and sequence overview. | Flower DR et al |
| 8761444 | 1996 | The lipocalin protein family: structure and function. | Flower DR et al |
| 11369448 | 2001 | Ligand activated hPR modulates the glycodelin promoter activity through the Sp1 sites in human endometrial adenocarcinoma cells. | Gao J et al |
| 2006183 | 1991 | Multiple forms of mRNA encoding human pregnancy-associated endometrial alpha 2-globulin, a beta-lactoglobulin homologue. | Garde J et al |
| 11058745 | 2000 | Evolution of the lipocalin family as inferred from a protein sequence phylogeny. | Gutiérrez G et al |
| 11058757 | 2000 | Glycodelin: a reproduction-related lipocalin. | Halttunen M et al |
| 20676758 | 2011 | Glycodelin expression associates with differential tumour phenotype and outcome in sporadic and familial non-BRCA1/2 breast cancer patients. | Hautala LC et al |
| 16269626 | 2006 | alpha2,6-Sialylation promotes binding of placental protein 14 via its Ca2+-dependent lectin activity: insights into differential effects on CD45RO and CD45RA T cells. | Ish-Shalom E et al |
| 14764802 | 2004 | Reduced serum glycodelin and insulin-like growth factor-binding protein-1 in women with polycystic ovary syndrome during first trimester of pregnancy. | Jakubowicz DJ et al |
| 14679202 | 2004 | Analysis of the role of oligosaccharides in the apoptotic activity of glycodelin A. | Jayachandran R et al |
| 6776278 | 1980 | A progestagen-dependent endometrial protein in human amniotic fluid. | Joshi SG et al |
| 3194393 | 1988 | Complete amino acid sequence of human placental protein 14: a progesterone-regulated uterine protein homologous to beta-lactoglobulins. | Julkunen M et al |
| 10597188 | 1999 | Expression of glycodelin in human breast and breast cancer. | Kämäräinen M et al |
| 19551557 | 2009 | The role of glycodelin in cell differentiation and tumor growth. | Koistinen H et al |
| 19520712 | 2009 | Glycodelin-A as a modulator of trophoblast invasion. | Lam KK et al |
| 19945098 | 2010 | Glycodelin-A modulates cytokine production of peripheral blood natural killer cells. | Lee CL et al |
| 19240032 | 2009 | Effects of differential glycosylation of glycodelins on lymphocyte survival. | Lee CL et al |
| 14559812 | 2003 | Glycodelin in ovarian serous carcinoma: association with differentiation and survival. | Mandelin E et al |
| 15494501 | 2004 | Differential regulation of Th1/Th2 cytokine responses by placental protein 14. | Mishan-Eisenberg G et al |
| 8943270 | 1996 | Gender-specific glycosylation of human glycodelin affects its contraceptive activity. | Morris HR et al |
| 7992851 | 1994 | Hematopoietic placental protein 14. An immunosuppressive factor in cells of the megakaryocytic lineage. | Morrow DM et al |
| 11325960 | 2001 | Placental protein 14 induces apoptosis in T cells but not in monocytes. | Mukhopadhyay D et al |
| 7531163 | 1995 | Factors affecting fertilization: endometrial placental protein 14 reduces the capacity of human spermatozoa to bind to the human zona pellucida. | Oehninger S et al |
| 18178606 | 2008 | Glycodelin blocks progression to S phase and inhibits cell growth: a possible progesterone-induced regulator for endometrial epithelial cell growth. | Ohta K et al |
| 1840727 | 1991 | Suppression by human placental protein 14 of natural killer cell activity. | Okamoto N et al |
| 17716661 | 2007 | Differential sialylation regulates the apoptotic activity of glycodelin A. | Poornima BL et al |
| 12556471 | 2003 | Negative regulation of T cell activation by placental protein 14 is mediated by the tyrosine phosphatase receptor CD45. | Rachmilewitz J et al |
| 11023837 | 2000 | alpha2-macroglobulin modulates the immunoregulatory function of the lipocalin placental protein 14. | Riely GJ et al |
| 20592348 | 2010 | Glycodelin expression in correlation to grading, nodal involvement and steroid receptor expression in human breast cancer patients. | Scholz C et al |
| 9591048 | 1998 | Glycodelins. | Seppälä M et al |
| 19039086 | 2009 | Glycodelin in reproductive endocrinology and hormone-related cancer. | Seppälä M et al |
| 12202458 | 2002 | Glycodelin: a major lipocalin protein of the reproductive axis with diverse actions in cell recognition and differentiation. | Seppälä M et al |
| 9062493 | 1997 | The role of relaxin in glycodelin secretion. | Stewart DR et al |
| 9814484 | 1998 | Promegestone (R5020) and mifepristone (RU486) both function as progestational agonists of human glycodelin gene expression in isolated human epithelial cells. | Taylor RN et al |
| 10321810 | 1999 | Relaxin stimulates glycodelin mRNA and protein concentrations in human endometrial glandular epithelial cells. | Tseng L et al |
| 20592354 | 2010 | Determination of glycodelin-A expression correlated to grading and staging in ovarian carcinoma tissue. | Tsviliana A et al |
| 17720699 | 2007 | Histone deacetylase inhibitor-induced glycodelin enhances the initial step of implantation. | Uchida H et al |
| 2206398 | 1990 | Human placental protein 14 gene: sequence and characterization of a short duplication. | Vaisse C et al |
| 2016092 | 1991 | The human placental protein 14 (PP14) gene is localized on chromosome 9q34. | Van Cong N et al |
Other Information
Locus ID:
NCBI: 5047
MIM: 173310
HGNC: 8573
Ensembl: ENSG00000122133
Variants:
dbSNP: 5047
ClinVar: 5047
TCGA: ENSG00000122133
COSMIC: PAEP
RNA/Proteins
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 33388716 | 2021 | Pregnancy Zone Protein (PZP) is significantly upregulated in the decidua of recurrent and spontaneous miscarriage and negatively correlated to Glycodelin A (GdA). | 6 |
| 33388716 | 2021 | Pregnancy Zone Protein (PZP) is significantly upregulated in the decidua of recurrent and spontaneous miscarriage and negatively correlated to Glycodelin A (GdA). | 6 |
| 31730930 | 2020 | Glycodelin regulates the numbers and function of peripheral natural killer cells. | 5 |
| 32120159 | 2020 | Glycodelin is internalized by peripheral monocytes. | 1 |
| 32205858 | 2020 | Altered glycosylation of glycodelin in endometrial carcinoma. | 10 |
| 31730930 | 2020 | Glycodelin regulates the numbers and function of peripheral natural killer cells. | 5 |
| 32120159 | 2020 | Glycodelin is internalized by peripheral monocytes. | 1 |
| 32205858 | 2020 | Altered glycosylation of glycodelin in endometrial carcinoma. | 10 |
| 30535430 | 2019 | Pathways regulating the expression of the immunomodulatory protein glycodelin in non‑small cell lung cancer. | 6 |
| 30535430 | 2019 | Pathways regulating the expression of the immunomodulatory protein glycodelin in non‑small cell lung cancer. | 6 |
| 28683601 | 2018 | Dysregulation of GdA Expression in Endometrium of Women With Endometriosis: Implication for Endometrial Receptivity. | 15 |
| 28738719 | 2018 | Maternal serum glycodelin levels in preeclampsia and its relationship with the severity of the disease. | 3 |
| 29749501 | 2018 | Placental protein 14 as a potential biomarker for diagnosis of preterm premature rupture of membranes. | 6 |
| 28683601 | 2018 | Dysregulation of GdA Expression in Endometrium of Women With Endometriosis: Implication for Endometrial Receptivity. | 15 |
| 28738719 | 2018 | Maternal serum glycodelin levels in preeclampsia and its relationship with the severity of the disease. | 3 |
Citation
Hannu Koistinen ; Markku Seppälä
PAEP (progestagen-associated endometrial protein)
Atlas Genet Cytogenet Oncol Haematol. 2010-11-01
Online version: http://atlasgeneticsoncology.org/gene/46067/paep
