Clinics and Pathology
Note
Intraocular tumors vary significantly in epidemiology, etiology, pathology, and treatment methods. As a result, the major tumors are discussed separately under each heading.
Etiology
Traditionally, retinoblastoma was believed to come in a germinal and non-germinal form, both of which result from a mutation or loss of both alleles of the retinoblastoma gene (
RB1). In germinal cases, the first mutation is in the germline and the second is somatic. In the non-germinal form, both mutations are somatic resulting in unilateral and unifocal lesions. Recent data suggests that all patients with retinoblastoma express a degree of mosaicism. (Sippel et al., 1998).
Epidemiology
With a lifetime incidence rate ranging between one in 18000 to 30000 live births, the most common primary ocular malignancy of childhood is a rare tumor. The etiology of this tumor seems to be minimally affected by environment, which is shown by the surprisingly similar incidence rates among several populations of the world. The median age of diagnosis in the United States is 18 months, with the median age for unilateral cases being 24 months and the median age for bilateral cases being 12 months. (Augsburger et al., 1995).
Clinics
The most common presenting symptom of patients with retinoblastoma in developed nations is leukocoria, which is a reflection of incoming light off of the tumor. Retinoblastoma is diagnosed ophthalmoscopically, but ultrasound and fundus photography should also be done to confirm the diagnosis. Ultrasound typically demonstrates a mass with high reflectivity and intralesional calcium causing shadowing behind the tumor.
Staging: The Reese-Ellsworth classification has been the predominant system used since its development in the 1960s as a way to predict prognosis in patients treated with external beam radiation (Table 1) (Reese, 1976). Because of the decreasing use of external beam radiation for intraocular retinoblastoma, a new international classification system has been developed with subgroups ranging from disease that is most easily treated by chemotherapy and focal treatments to disease that is least easily treated by these methods (Table 2) (Murphree, 2005). The exclusive use of the new classification system makes comparisons with older series more difficult. Consequently, many authors will continue to include the Reese-Ellsworth classifications.
| Group I |
It most commonly presents as a gray-white lesion of the anterior chamber angle, but can present as a diffuse mass causing leukocoria (Broughton and Zimmerman, 1978). Neovascular glaucoma in a child with a normal posterior segment, iris notching, and an unexplained cyclitic membrane are features that may assist in diagnosis. While clinical examination may be sufficient to diagnose medulloepithelioma, ultrasound can be a useful adjunct as it demonstrates cystic spaces and a lack of calcifications. (Foster et al., 2000).| | b. Multiple tumors, none over 4 disc diameters in size, all at or behind the equator |
| Group II |
| | a. Solitary tumor, 4 to 10 disc diameters in size, at or behind the equator |
| | b. Multiple tumors, 4 to 10 disc diameters in size, behind the equator |
| Group III |
| | a. Any lesion anterior to the equator |
| | b. Solitary tumors larger than 10 disc diameters behind the equator |
| Group IV |
| | a. Multiple tumors, some larger than 10 disc diameters |
Because CHRPE are most commonly found in the peripheral retina, patients are commonly asymptomatic and present with round, darkly pigmented, flat lesions that can be surrounded by a hypopigmented halo (Lloyd et al., 1990). Lacunae, atrophied window-like defects, are present in about 50% of CHRPE lesions (Shields et al., 2003). Fundoscopic examination showing classical features of the lesion are sufficient for diagnosis, and no further ancillary studies are needed.| Group V |
| | a. Massive tumors involving over half the retina |
| | b. Vitreous seeding |
Table 1: Reese-Ellsworth scheme for intraocular retinoblastoma
| Group A | Rb ≤ 3 mm in basal dimension or thickness |
| Group B | Rb > 3 mm or with one or more of the following: |
Classically, combined hamartomas present as unilateral, dark, solitary lesions that are slightly elevated with varying amounts of retinal and epiretinal tissue centrally causing progressive traction and vascular tortuosity (Font et al., 1989). They are located in the macular and extramacular region at equal frequencies (Shields et al., 2008). Indirect ophthalmoscopy is often sufficient for diagnosis, but fluorescein angiography (FA) is a useful adjunct as it shows blocking of choroidal perfusion and progressive hyperfluorescence in the late phase (Schachat et al., 1984).| | - Juxtapapillary location (≤ 1.5 mm to optic nerve) |
| | - Additional subretinal fluid (≤ 3 mm from margin) |
| Group C | Retinoblastoma tumor with one of the following: |
| | - Subretinal seeds ≤ 3 mm |
| | - Vitreous seeds ≤ 3 mm |
| | - Both subretinal and vitreous seeds ≤ 3 mm |
| Group D | Retinoblastoma tumor with one of the following: |
| | - Subretinal seeds > 3 mm |
Characterized by melanotic pigmentation of the iris, patches of gray-brown scleral pigmentation, and ipsilateral choroidal hyperpigmentation (Rahman et al., 2008). There may or may not be involvement of the periorbital facial tissues in the distribution of the trigeminal nerve. Diagnosis is based on presentation with classic findings noted during slit-lamp examination.| | - Both subretinal and vitreous seeds > 3 mm |
| Group E | Extensive retinoblastoma occupying > 50% of globe or any of the following: |
| | - Neovascular glaucoma |
| | - Opaque media from vitreous hemorrhage in anterior chamber, vitreous, or subretinal space |
| | - Invasion of postlaminar optic nerve, choroid (> 2 mm), sclera, orbit, or anterior chamber |
Table 2: New international classification for retinoblastoma
Pathology
Retinoblastomas are characterized histopathologically by basophilic cells with minimal cytoplasm surrounding a lumen in a rosette formation or radially arranged around a central tangle of fibrils in a pseudorosette formation. Often times, necrosis and hemorrhage are present within the tumors, as they tend to outgrow their vascular supplies.
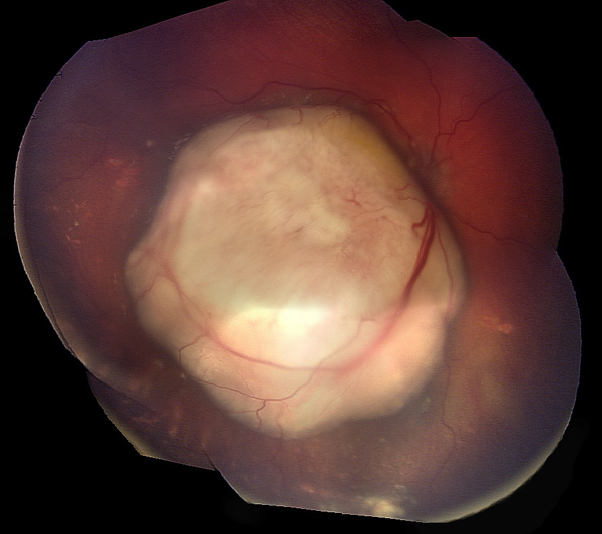
Endophytic retinoblastoma lesion in the right eye obscuring the view of the macula and optic disc.
Cytogenetics
Linkage analysis and deletion techniques discovered the RB1 to be localized to chromosome 13q14 (Yunis and Ramsay, 1978). The gene spans 180 kb, is composed of 27 exons, and encodes a 4.8 kb mRNA transcript that is expressed in all human tissues. The resulting protein product is a 110 kD nuclear phosphoprotein, consisting of 928 amino acids.
Genes
The RB1 gene encodes a protein that is a regulator at the major checkpoint of the cell cycle, between the G1 and S-phase. In its normal non-phosphorylated form, the retinoblastoma protein (pRB) binds to transcriptional factors, like E2F, to prevent entry into the S-phase. The phosphorylated form of pRB dissociates from E2F, which allows the transcription factor to bind DNA and promote progression through the cell cycle. Abnormal RB1 function allows for continuous entry into the S-phase and rapid cell division, which causes tumor formation. (Hernando et al., 2004).
Treatment
Treatment of retinoblastoma is often performed in a multi-modal approach including enucleation, external beam radiation, chemotherapy, transpupillary thermotherapy, cryotherapy, and brachytherapy. Chemotherapy has been the most widely used treatment for retinoblastoma since the mid-1990s when extensive research showed an increased risk in secondary tumors in survivors of germinal retinoblastoma treated with external beam radiation. The dosing regimens, schedules, and chemotherapeutic agents vary significantly among oncology centers, but the most common includes carboplatin, etoposide and vincristine. Most centers use chemotherapy to reduce the tumors, which allows focal treatment like laser and cryotherapy to be more effective. Enucleation is still used in very advanced cases. Some centers use periocular carboplatin with systemic chemotherapy to salvage globes with advanced disease. Anti-angiogenic agents and intra-arterial chemotherapy infusions are also currently being investigated.
Prognosis
The most significant factor leading to death is extraocular invasion by the tumor, with a considerable delay in initial diagnosis also contributing to a reduced likelihood of survival. The overall 5 to 10 year survival rate in developed nations is often listed to be around 92%.
Etiology
Medulloepithelioma is a tumor that arises from the epithelium of the medullary tube, most often the ciliary body. It can take a teratoid or nonteratoid form and usually presents itself as a unilateral congenital disease, although bilateral, juvenile, and adult-onset cases have also been reported. (Broughton and Zimmerman, 1978).
Epidemiology
Medulloepithelioma is a rare tumor with an incidence of 1 case per 450000 - 1000000 people (Augsburger and Schneider, 2004). Medulloepithelioma has no racial and gender predilection, has no clear pattern of inheritance, and no identifiable risk factors. The mean age at diagnosis is 4 - 5 years.
Pathology
Histopathologic analysis reveals a tumor composed of epithelium that can be arranged in cords and sheets separated by cystic spaces containing proteinaceous material. Teratoid forms contain heterotopic elements including skeletal muscle and cartilage. Malignant tumors often have areas consisting of poorly differentiated neuroblastic cells, increased mitotic activity, sarcomatous areas, and tumor invasion of other ocular tissue, regardless of extraocular extension. (Broughton and Zimmerman, 1978).
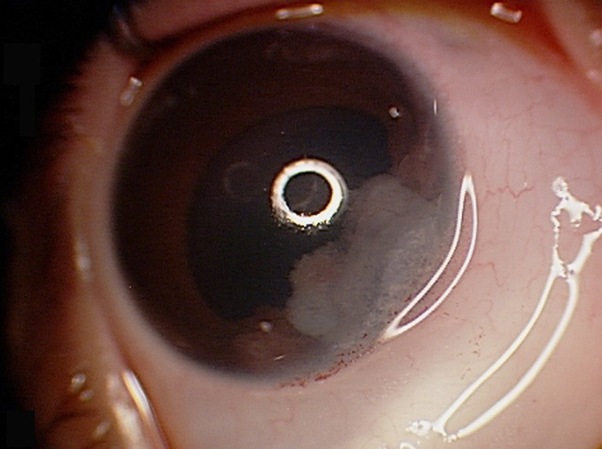
Medulloepithelioma with opaque white lesion.
Treatment
Currently, there is not a definitive treatment for medulloepithelioma. Observation is often recommended for smaller tumors without sequellae. Primary enucleation is recommended if the tumor is large, there is extrascleral extension, the eye is blind and painful, or there is neovascular glaucoma. Local resection and invasive diagnostic procedures should be avoided as they may lead to recurrence, direct seeding of the orbit, or creation of a tract for subsequent tumor migration from the globe and into the orbit. (Shields et al., 1996).
Prognosis
The natural history of untreated medulloepitheliomas is essentially unknown. Metastasis in medulloepithelioma is very rare, but portends a more negative prognosis when present. Only lesions with malignant features metastasize, but neither teratoid features nor malignant morphology predict mortality. While there has been no reported deaths or metastases in patients who undergo definitive enucleation without prior diagnostic invasive procedures, patients who have had prior invasive procedures are thought to have a higher mortality rate. (Canning et al., 1988).
Note
An association in the literature has been made between
familial adenomatous polyposis (FAP) and CHRPE-like lesions. FAP is a syndrome with an autosomal dominant mode of inheritance that causes the development of hundreds of pre-malignant colonic polyps and is caused by a mutation in the
APC gene located on chromosome 5q21. The lesions have also been described in instances of Gardners and Turcots syndrome. Gardners syndrome consists of FAP associated with soft-tissue tumors and skeletal growths, while Turcots syndrome consists of APC associated with tumors of the central nervous system. The ocular manifestations associated with FAP, Gardners, and Turcots syndrome have been labeled as pigmented ocular fundus lesions (POFLs) in order to differentiate them from CHRPE lesions. Not only are POFLs bilateral, numerous, and pisciform in shape, but they also differ from CHRPE lesions histopathologically as they are shown to have hamartomatous changes in addition to RPE hypertrophy and hyperpigmentation. (Parsons et al., 2005).
Etiology
Congenital hypertrophy of the retinal pigment epithelium (CHRPE) is an isolated sporadic congenital lesion with no known underlying genetic basis.
Epidemiology
While the prevalence of CHRPE is unknown because it usually presents asymptomatically, one study demonstrated a prevalence of 1.2% (Coleman and Barnard, 2007). Age is not a relevant factor in the development of CHRPE because it is a congenital lesion, but studies have shown a median age of diagnosis to be 45 (Shields et al., 2003). Studies suggest Caucasians are more likely to demonstrate CHRPE (Shields et al., 2003).
Pathology
Histopathologically, CHRPE lesions show hypertrophied RPE cells that contain excessive pigment granules resembling melanin without lipofuscin (Parsons et al., 2005). The overlying photoreceptor layer may be atrophic, while the underlying choroid and choriocapillaris are usually normal. There may be RPE dropout or reduced pigmentation in areas of lacunae, and glial cells are present between Bruchs membrane and the RPE in these areas (Parsons et al., 2005).
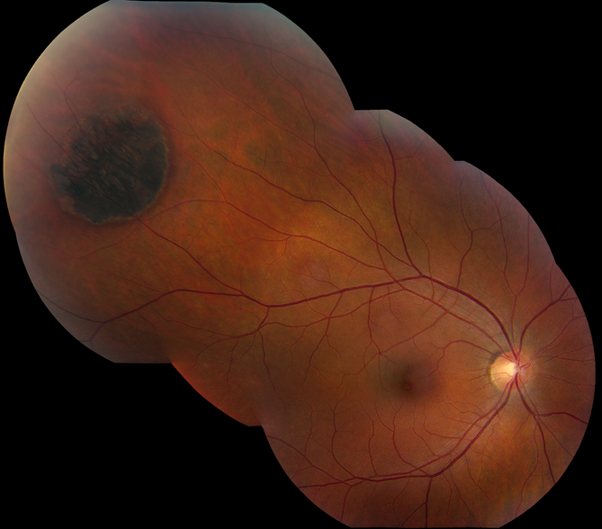
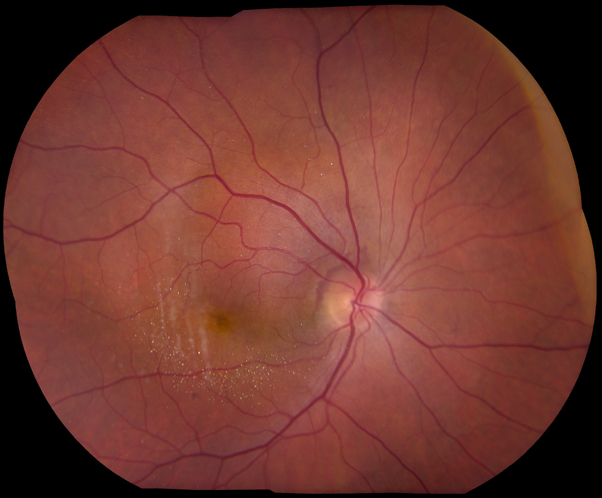
Congenital hypertrophy of the retinal pigment epithelium located in the superotemporal quadrant of the right eye. Note the flat, round, and darkly pigmented classical appearance of the lesion.
Treatment
Treatment is usually unnecessary except for the rare instance in which neovascularization presents at the periphery of the lesion.
Prognosis
CHRPE are benign lesions that show minimal growth in up to 80% of cases, and usually do not cause visual disturbances (Shields et al., 2003). On a rare occasion, CHRPE may transform to malignant adenocarcinoma, but the etiology and most appropriate management has yet to be determined (Shields et al., 2009).
Etiology
Combined hamartoma of the retina and retinal pigment epithelium (RPE) is a rare developmental disorder caused by benign proliferations of both the retina and RPE. There may be a systemic association with
neurofibromatosis type I and
neurofibromatosis type II (Palmer et al., 1990; Kaye et al., 1992), but the mechanistic relationship has not yet been established. They are considered to be congenital lesions in most instances but acquired lesions have been reported infrequently (Ticho et al., 1998).
Epidemiology
The prevalence of this occurrence has not been established, but whites have been shown to be more frequently affected (Shields et al., 2008). Two major studies have shown a mean age of diagnosis 15-18 years (Font et al., 1989; Schachat et al., 1984), and there may be a slight predilection for males (Font et al., 1989).
Pathology
Histopathologically, combined hamartomas demonstrate infiltration of hyperplastic RPE into the retina and inner retinal surface. Gliosis is significant and is responsible for the tractional changes and vascular tortuosity. (Vogel et al., 1969).
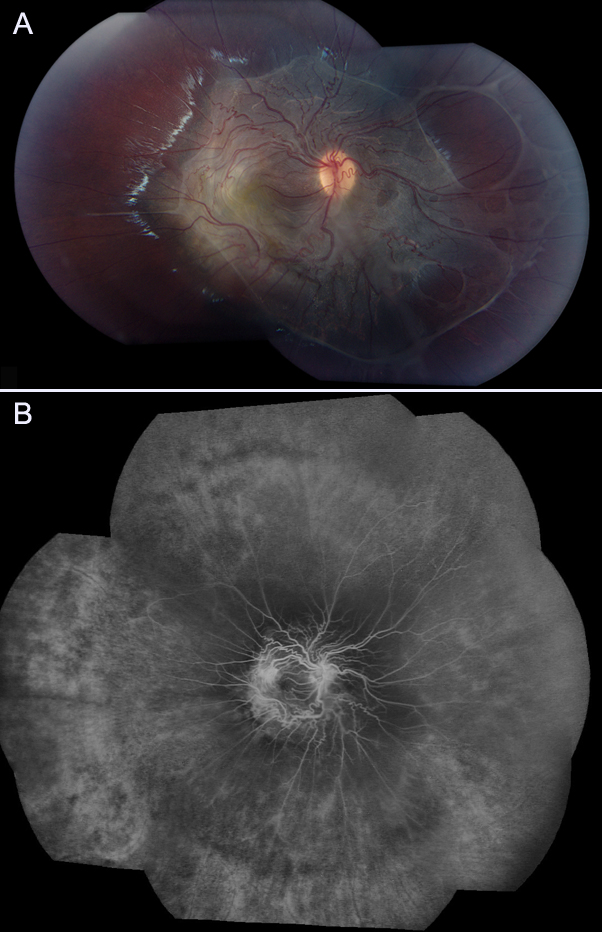
Combined hamartoma of the retina and retinal pigment epithelium positioned on the optic disk and adjacent retina with a predominantly glial component. A, Color fundus photography with peripheral areas of traction corresponding to the posterior hyaloid face. B, Fluorescein angiography of the lesion in the same patient demonstrating the striking vascular abnormalities.
Treatment
While most cases of combined hamartoma are isolated from systemic findings, patients who have been diagnosed should undergo evaluation to exclude neurofibromatosis. Amblyopia therapy has been shown to improve vision in some patients with combined hamartoma (Schachat et al., 1984). Vitrectomy and membrane removal has been performed, but the visual acuity does not always improve significantly and membranes may recur (Shields et al., 2008). Consequently, the role of vitrectomy in managing combined hamartoma has not been fully established. Rarely, choroidal neovascularization may occur as a complication and can be treated with laser.
Prognosis
Combined hamartoma can cause significant visual loss, with a visual acuity
Etiology
Ocular melanocytosis is a congenital hyperpigmentation of the globe caused by increased numbers of melanocytes. One study shows that the hyperpigmentation of ocular melanocytosis is primarily congenital as it was documented in 85% of patients at birth (Teekhasaenee et al., 1990). There is often dermal involvement due to the failure of melanocytes of neural crest cell origin to reach the intended surface positions, which gives rise to hamartomatous nests in the distribution of the first and second branches of the trigeminal nerve (Zaihosseini et al., 2008).
Epidemiology
Ocular melanocytosis is an uncommon condition with a prevalence rate of 0.038% in the white population, 0.014% in the black population, and between 0.4 and 0.84% in the Asian population (Gonder et al., 1982). There are no differences in frequency based on laterality or gender.
Pathology
Histopathologically, ocular melanocytosis is characterized by the presence of dendritic melanocytes in the areas of hyperpigmentation.
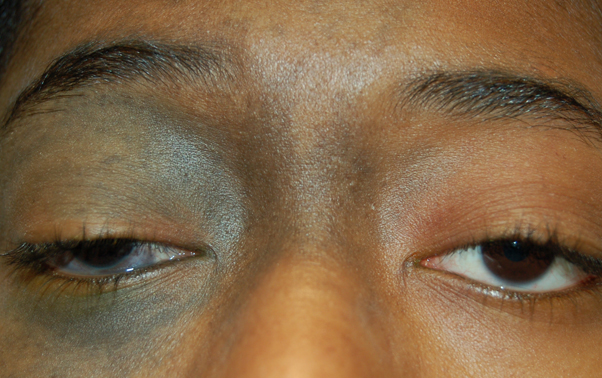
Congenital melanocytosis revealing gray-brown hyperpigmentation of the sclera and periorbital area of the right eye.
Treatment
In and of itself, congenital ocular melanocytosis is a benign condition that does not require treatment. However, in the white population there has been shown to be a lifetime risk of developing uveal melanoma of 1 in 400, which is significantly greater than the 1 in 13000 risk observed without underlying congenital melanocytosis (Shields et al., 1991). Because of the association with uveal melanoma, annual ophthalmic follow-up is recommended for all patients with ocular melanocytosis (Rahman et al., 2008).
Prognosis
Visual impairments that arise in the context of ocular melanocytosis are due to the development of complications including uveitis, glaucoma, and cataract. Additionally, the association with an increased frequency of uveal melanoma in the affected eye, has further implications on morbidity and mortality. (Gonder et al., 1982).
Etiology
Uveal nevi are stromal, hamartomatous clusters consisting of atypical melanocytes of neural crest origin much like cutaneous melanocytes. They have been described predominantly in three locations including the iris, ciliary body, and choroid.
Epidemiology
Iris nevi are found in about 4 to 6% of the population (Harbour et al., 2004), while ciliary body nevi are infrequently reported. Choroidal nevi have a reported prevalence rate that ranges from 0.2% to 30% depending on the population (Shields et al., 1995) and are rarely found in blacks. They often become pigmented or develop within the first three decades of life, and there is no conclusive data to show an association with gender.
Clinics
Iris nevi are typically solitary, circumscribed lesions located in the lower quadrants of the iris, ranging from tan to dark brown. Ciliary body nevi present as dome-shaped masses without intrinsic vascularity. Diagnosis of both iris and ciliary body lesions can often be made based on anterior segment evaluation with gonioscopy. Ultrasound biomicroscopy assists in diagnosis by determining size, extent and solid or cystic consistency (Conway et al., 2005). Choroidal nevi usually do not cause symptoms and present as grayish brown lesions with minimal thickness, and diagnosis can be made by ophthalmoscopy.
Pathology
Nevi are known to consist of four different cell types including plump polyhedral, slender spindle, intermediate, and balloon cells. Posteriorly, choroidal nevi have been shown to involve full thickness of the choroid with sparing of the choriocapillaris. (Naumann et al., 1966).
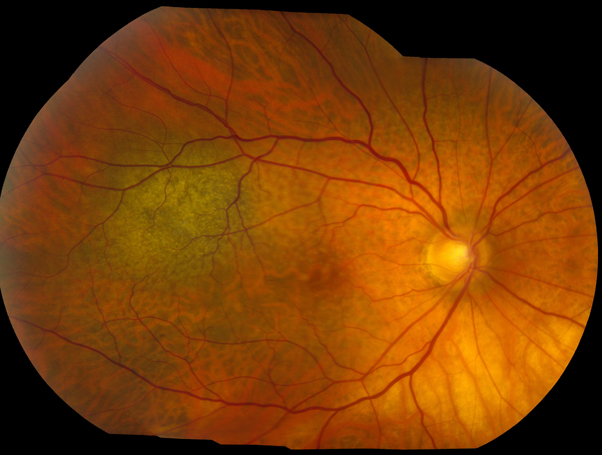
Pigmented choroidal nevus situated temporal to the macula with overlying drusen.
Treatment
Periodic ophthalmic examination for nevi to check for growth or malignant progression is recommended for nevi in all locations. For choroidal nevi, follow-up is especially important if the nevi cause decreased vision or visual field defects or have high-risk characteristics such as thickness greater than 2mm, posterior location, orange pigment, subretinal fluid, and absence of drusen (Singh et al., 2005; Singh et al., 2006).
Prognosis
The prognosis for all forms of nevi tends to be good with iris nevi showing enlargement in less than 5% of cases (Territo et al., 1988). For choroidal nevi, the rate of transformation to melanoma is significantly less than 1%, which means that the majority of patients with nevi can be advised that their condition is most likely benign.
Etiology
Uveal melanoma is the most common primary intraocular malignant tumor, and they originate from the iris, ciliary body, or choroid. The majority of these tumors arise in the choroid, and the predisposing factors include family history of choroidal melanoma,
dysplastic nevus syndrome,
xeroderma pigmentosum, and congenital ocular melanocytosis. While some arise de novo (Sahel et al., 1988), there is clinical and histopathologic evidence that many originate from pre-existing, benign nevi (Naumann et al., 1966). Exposure to sunlight has been postulated to have an effect, but there is no convincing evidence of a causative relationship (Shah et al., 2005).
Epidemiology
Choroidal melanomas are found most often in countries with large populations of people of northern European decent. Caucasians are 19 times more likely to have choroidal melanoma than African Americans and 16 times more likely than Asians (Hu et al., 2005). While there is wide age variation in melanoma patients, 65% are over the age of 50. Men and women are equally affected, and there is no predilection for either eye.
Clinics
Patients with both iris and ciliary body melanomas may present without symptoms, or may have glaucomatous changes or cataract progression as a result of the tumor. Diagnosis can often be made from slit-lamp examination and gonioscopy, but ultrasound biomicroscopy is useful to guide therapy. Choroidal melanomas are also usually found on routine examinations in asymptomatic patients, but they may cause decreased visual field and acuity. Ophthalmoscopy is typically adequate for diagnosis, as the lesions tend to have a dome or mushroom shaped appearance that often protrudes into the vitreous. Ultrasound and FA can be used to aid in diagnosis, with ultrasound classically showing an elevated solid tumor with low to medium internal reflectivity and significant vascularity. (Coleman et al., 1974).
Clinics
Staging: While melanomas lack discrete clinical and pathological stages, new evidence indicates that they can be classified into distinct molecular classes that are strongly associated with metastatic risk. These classes are based on gene expression profile with class 1 representing low-grade and class 2 representing high-grade tumors. Class 2 tumors demonstrate down-regulated gene clusters on chromosome 3 and up-regulated gene clusters on chromosome 8q. A Kaplan-Meier based analysis showed survival prediction of 95% in class 1 and 31% in class 2 at 92 months. (Onken et al., 2004).
Pathology
Spindle A, spindle B and epithelioid are the three cell types found in melanomas. The Callender classification divides these tumors by histopathologic type into spindle cell, mixed-cell and epithelioid cell tumors, with a poorer prognosis for survival in tumors that have a higher proportion of epithelioid cells (Mclean et al., 1983). However, this classification can only be used in cases in which the eye has been removed.
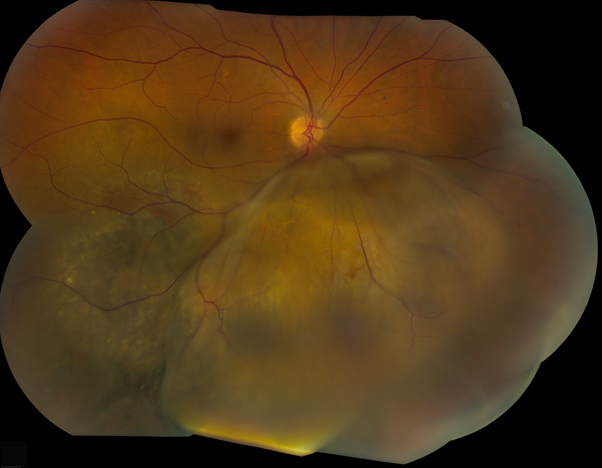
Large, elevated inferior choroidal melanoma with both melanotic and amelanotic components. Note the overlying retinal detachment located in the inferonasal quadrant.
Cytogenetics
Multiple chromosomal abnormalities have been detected in uveal melanoma tissues and associated with metastasis, including gain or loss of chromosomal material in chromosomes 3, 6, and 8 (Sisley et al., 1990). Monosomy 3 is a predictor of increased risk of relapse and mortality (Prescher et al., 1996). Studies have shown as many as 57% of patients with monosomy 3 developed metastasis, while no patients with disomy 3 developed metastatic disease within 3 years (Prescher et al., 1996).
Treatment
The Collaborative Ocular Melanoma Study (COMS) divided choroidal melanomas into small, medium and large tumors in order to identify the best treatment modality for each (Karlsson et al., 1989). Observation is often the primary management for small tumors, particularly if they do not show high-risk characteristics, but they can be treated with transpupillary thermotherapy (TTT) or brachytherapy (Robertson et al., 1999; Sobrin et al., 2005). Medium sized tumors are treated by brachytherapy, which shows equivalent survival rates as enucleation (Diener-West et al., 2001). Large tumors are most often treated with enucleation alone, but treatment with brachytherapy may be reasonable as recent studies suggest similar survival rates to those in patients treated with enucleation.
Prognosis
Analysis of choroidal melanoma has determined various mortality rates based primarily on tumor size and treatment used. The COMS showed a 1% melanoma-specific mortality rate for small tumors (COMS group, 1997), but it is important to note that this study included a large number of suspected tumors that did not grow and were never treated. Medium-sized melanomas have similar 5-year melanoma-specific mortality rates after brachytherapy or enucleation (19 and 18% respectively) (Diener-West et al., 2001). Due to complications after treatment with brachytherapy, 43% of patients have poor vision, 20/200 or worse, by 3 years of follow-up (Melia et al., 2006). Large melanomas treated with enucleation alone have a 43% all-cause and 27% melanoma-specific 5-year mortality rate (COMS group, 1998).
Etiology
Fuchs adenoma is benign, acquired tumor that arises from pars plicata of the ciliary body and seems to be age-related (Bateman and Foos, 1979). This lesion has also been labeled coronal adenoma and age-related hyperplasia of nonpigmented ciliary epithelium.
Epidemiology
These lesions are not uncommon as they have been shown to be found in 14-18% of eyes at autopsy (Zaidman et al., 1983). This tumor has an increased frequency in older patients, generally found from 50-60 years.
Clinics
Fuchs adenoma predominantly presents as an asymptomatic opaque white mass that can be solitary or multiple, unilateral or bilateral and is usually confined to 1 ciliary process (Shields et al., 2009). Clinical observation is often enough for diagnosis, but these lesions are often misdiagnosed as malignant neoplasms. Careful observation is initially important to determine the growth pattern, which assists in diagnosis.
Pathology
Histopathologically, Fuchs adenoma are characterized by a proliferation of cords of nonpigmented epithelial cells surrounded by amorphous periodic acid-Schiff-positive extracellular material (Zaidman et al., 1983).
Fuchs adenoma located in the anterior chamber angle.
Treatment
No treatment is recommended in most cases of Fuchs adenoma as they are benign and nonprogressive. Because these lesions may precipitate cataract formation, cataract extraction surgery may be required (Zaidman et al., 1983).
Prognosis
Most often the tumors are found incidentally because of small size and location (Bateman and Foos, 1979). As a result, they tend not to impair visual acuity except in the rare instance in which they cause cataract progression.
Etiology
Metastasis to the eye most commonly arises from primary lung and breast cancer, with many patients having no previously diagnosed primary cancer (American Academy of Ophthalmology, 2004).
Epidemiology
It is estimated that 30000 to 100000 patients with cancer develop metastasis to the eye each year, which is significantly more than the 350 and 1500 yearly cases of retinoblastoma and choroidal melanoma respectively (Shields et al., 1997).
Clinics
The uveal tract is the most common location for metastatic disease in the eye, with choroidal metastasis being more common than iris or ciliary body metastasis. Ophthalmoscopic examination demonstrates classic findings including multiple, bilateral, minimally elevated, and amelanotic lesions. Ultrasound is often used to confirm diagnosis, showing high internal reflectivity.
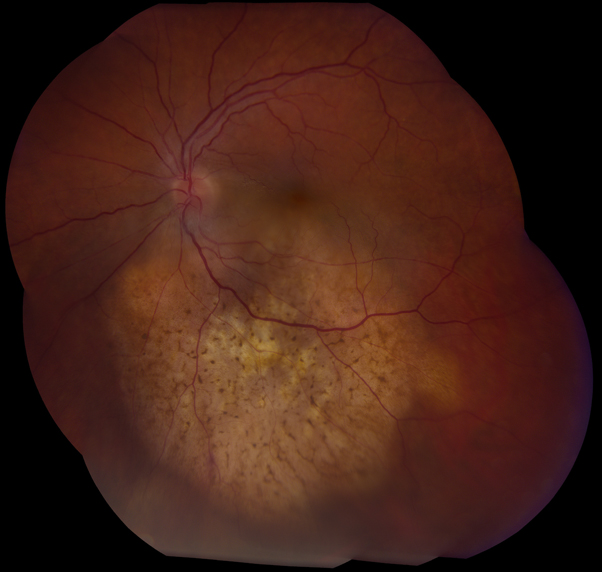
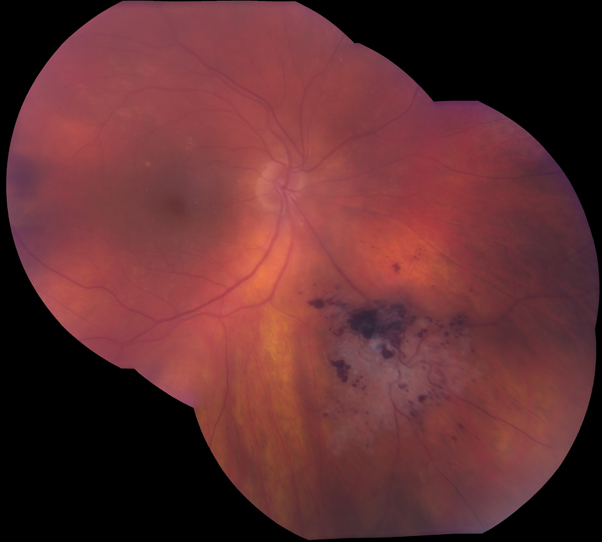
Large amelanotic choroidal metastasis from a lung adenocarcinoma in a patient whose vision recovered to 20/30 in this eye after external beam radiation. The patient died 21 months after diagnosis of the lung cancer, which was discovered due to ocular symptoms.
Treatment
Treatment of ocular lesions rarely has an impact on survival, but may improve the quality of life. It is therefore considered in response to decreased vision, pain, or diplopia. Treatment options include chemotherapy, hormonal manipulation, and external beam radiation.
Prognosis
With the exception of breast and carcinoid cancer, median survival in patients with metastatic choroidal lesions is just over 6 months, reflecting the overall mortality patterns due to the primary lesion.
Etiology
Retinal capillary hemangioma (RCH) is the earliest and most frequent manifestation of
von Hippel-Lindau (VHL) disease, which is a cancer syndrome with an autosomal-dominant mode of inheritance that results from mutations in the
VHL gene. Between 49-85% of people with VHL have a RCH (Singh et al., 2001a), which usually presents as a solitary lesion. However, there are multiple RCHs in about one-third of patients and bilateral involvement in up to half of patients, which are not associated with VHL disease.
Epidemiology
Incidence of RCH is estimated to occur 1 in 36000 to 1 in 53000 people. Mean age of VHL diagnosis was 25 in patients with retinal involvement (Singh et al., 2001a). Non-VHL-associated hemangiomas have a later onset, with the mean age of diagnosis being 35 (Singh et al., 2001b). The accumulation of the tumor phenotype may be age dependent.
Clinics
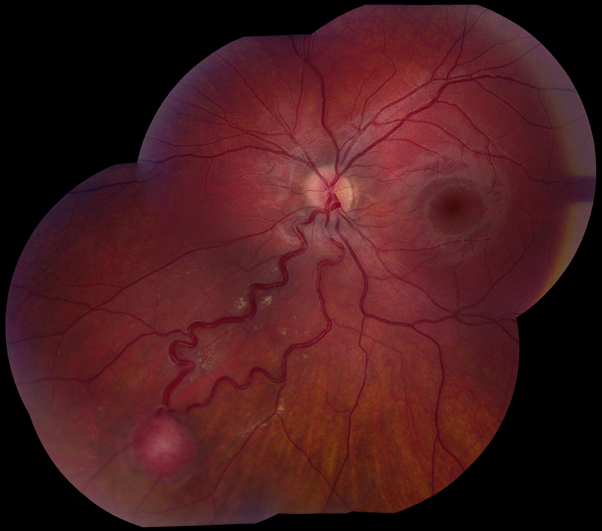
The classic manifestation of RCH is a round, circumscribed, red-orange colored vascular lesion that is supplied by prominent feeder vessels. The lesion is most often located in the peripheral retina but may also be seen in the juxtapapillary retina or both locations (Wong et al., 2008). Ophthalmoscopic evaluation is usually sufficient for diagnosis, but the use of FA is particularly helpful because of the vascular nature of the tumor. The FA classically shows filling the dilated feeder arteriole in the arterial phase followed by filling of the draining vein in the venous phase, and the lesion itself shows progressive hyperfluorescence with late leakage (Singh et al., 2001a).
Pathology
Histopathologically, RCHs demonstrate vascular channels that are lined by pericytes and endothelial cells and are separated by stromal cells. Of the three cells associated with the tumor, the stromal cells are thought to be the neoplastic component (Singh et al., 2001a).
Retinal capillary hemangioma presenting with the classic appearance of a dilated artery feeding the vascular lesion and engorged draining vein.
Cytogenetics
The VHL gene is located on chromosome 3p25-26 and is represented in 3 exons contained in a 20 kb region (Singh et al., 2001a). The mutation in the VHL gene varies significantly, ranging from a substitution of a single amino acid to the deletion of the entire gene (Wong and Chew, 2008). The somatic cells in patients with inherited VHL have a single mutated copy of the VHL gene, which is a tumor suppressor gene. When a retinal cell acquires a mutation in the remaining VHL gene, there is subsequent transformation into the tumor phenotype (Wong et al., 2008).
Genes
The VHL protein is responsible for degrading hypoxia inducible factors (HIFs), which are produced in response to hypoxic conditions and upregulate the release of
vascular endothelial growth factor and platelet-derived growth factor. The increased levels of HIFs that occur in the absence of VHL protein function subsequently lead to elevated levels of vascular endothelial derived growth factor and platelet derived growth factor, which contribute to tumor formation. (Wong and Chew, 2008; Kaelin, 2002).
Treatment
Multiple treatment modalities are used in the management of RCH. Observation is often the initial management for juxtapapillary hemangiomas as treatment tends to damage the optic nerve and the major retinal vessels leading to permanent visual loss. Laser is usually indicated for small lesions located in the periphery, while cryotherapy is preferred when the tumor is anteriorly located, has a diameter larger than 3 mm, and associated subretinal fluid large enough to cause decreased laser uptake. Transpupillary thermotherapy and plaque brachytherapy have also been used, but their roles are still undefined. Vitrectomy and enucleation are useful adjuvant therapies used in the management of RCHs, particularly in treating complex complications including rhegmatogenous or tractional retinal detachment, phthisis bulbi, neovascular glaucoma, and painful blind eye. Anti-angiogenic agents have been proposed as alternative treatments and a few studies have demonstrated positive responses. (Wong and Chew, 2008).
Prognosis
Because most RCHs progressively enlarge, early diagnosis and treatment are associated with better visual outcomes. Regardless, even in the setting of optimal treatment, more than 25% of affected patients show permanent visual loss with a best corrected visual acuity of less than 20/40 in at least one eye and about 20% have vision less than 20/100 in at least one eye (Webster et al., 1999). The likelihood of visual loss increases with age, secondary to the development of complications over time (Wong et al., 2008). A worse visual prognosis associated with juxtapapillary as compared to peripheral lesions. There is also an association of worse vision with increased number of peripheral lesions.
Etiology
Cavernous hemangioma of the retina are rare forms of congenital hamartomas that appear as solitary vascular tumors about 1 to 2 disc diameters in size and are usually in the midperiphery (Hewick et al., 2004). They are composed of thin-walled vascular channels with surface gliosis lined by non-fenestrated endothelium. Though sometimes transmitted in an autosomal dominant form, these tumors are usually sporadic (Sarraf et al., 2000). These may be included in the neuro-oculo-cutaneous (phakomatosis) syndromes, but the association with cerebral and cutaneous lesions is inconsistent (Gass, 1971).
Epidemiology
Although the incidence of cavernous hemangiomas of the retina cannot be determined because they are asymptomatic (Pancurak et al., 1985), women are more commonly affected (Patikulsila et al., 2007). The average age of presentation is 23 years, and the lesions are usually unilateral.
Clinics
Cavernous hemangiomas of the retina classically present as saccular, grapelike aneurysms that are filled with blood and protrude into the vitreous. Layering of the plasma overlying erythrocytes in the dependant part of some of the larger aneurysms can often be seen (Gass, 1971). While the presence of characteristic findings on fundoscopy is sufficient for diagnosis in most cases, flourescein angiography (FA) is the most common ancillary examination used to assist in diagnosis. The classic findings on FA are delayed perfusion of the tumor with normal retinal perfusion, lack of feeder or draining vessels and a lack of exudates (Chen, 2008).
Pathology
Cavernous hemangiomas of the retina are identified histopathologically by saccular aneurysmal dilatations that exhibit the anatomy of normal vessels. Nonfenestrated endothelial cells line the vessels, and the basal membrane is surrounded by basement membrane encased pericytes, which is why there are no intraretinal exudates. (Messmer et al., 1984).
Retinal cavernous hemagioma showing characteristic cluster-of-grapes appearance.
Cytogenetics
The causative gene of cerebral cavernous hemangiomas has been localized to 7q11-22, but it is not known if a gene in this region is responsible for retinal lesions.
Treatment
Because cavernous hemangiomas of the retina are usually non-progressive, observation is the best management. No treatment has been shown to be effective or necessary, but photocoagulation has been used in some cases (Gass, 1971). The association of retinal cavernous hemangiomas with cerebral cavernous malformations in an autosomal dominant syndrome with variable expressivity and high penetrance indicates neuroimaging may be reasonable for patients and family members in addition to dilated funduscopic exams (Pancurak et al., 1985).
Prognosis
Most patients with localized cavernous hemangioma of the retina retain good vision. Rarely, visual loss secondary to vitreous hemorrhage or contraction of the preretinal membrane overlying the tumor may occur. (Hewick et al., 2004).
Etiology
Choroidal hemangioma is a benign vascular hamartoma that occurs most commonly in a solitary circumscribed form but may also present as a diffuse form (Witschel and Font, 1976). The cause and pathogenesis of circumscribed choroidal hemangioma (CCH) remains unclear, while diffuse choroidal hemangioma (DCH) is congenital and associated with
Sturge-Weber syndrome. Sturge-Weber syndrome is a sporadic neurocutaneous disorder characterized by facial capillary malformation, leptomeningial angioma, and vascular ocular abnormalities (Baselga, 2004).
Epidemiology
Both DCH and CCH most commonly present in the white population, and age at diagnosis is 8 and 39, respectively (Witschel and Font, 1976). There is no predilection based on gender or laterality.
Clinics
CCH often present as a dome-shaped, orange-red mass that may be confused with malignant lesions ophthalmoscopically, which increases the necessity of FA, ICG, and ultrasonography to aid in diagnosis. Ultrasonography demonstrates high internal reflectivity and acoustic solidity, FA is characterized by progressive hyperflourescence in all stages, and ICG transitions from an early hyperfluorescence to late moderate hypofluorescence (Shields et al., 2001). The vast majority of patients with DCH have a marked exudative detachment, hyperopia, and involvement of more than half of the retina (Witschel and Fond, 1976; Heimann and Damato, 2009). Ophthalmoscopically, DCH may be more difficult to identify, making ancillary studies crucial for diagnosis. Patients with DCH have similar FA and ICG findings as those with CCH, but ultrasonography demonstrates diffuse thickening of the choroid with acoustic solidity.
Pathology
Histopathologically, choroidal hemagiomas have a complete lack of cellular proliferation of the elements of the vessel wall, indicating the nonproliferative nature of these lesions (Witschel and Font, 1976). CCH tend to have well-demarcated peripheral margins with a layer of compressed melanocytes and choroidal lamellae separating the lesion from the uninvolved choroid, while the DCH does not have clear margins separating it from the normal choroid (Witschel and Font, 1976).
Typical presentation of a choroidal hemangioma with an orange elevated lesion just superotemporal to the optic disk.
Treatment
Observation is recommended for patients with CCH who are asymptomatic, while photodynamic therapy has become the treatment of choice for patients who are symptomatic. Because of the association with Sturge-Weber syndrome, all patients with port-wine stains should be evaluated for DCH with a fundus examination, ultrasonography, and measurement of intraocular pressure (Baselga, 2004). Neurological evaluation should be performed in patients with DCH because of the high association of leptomeningeal angiomatous lesions in patients with DCH (Baselga, 2004). Treatment for DCH consists of external or proton beam radiotherapy, but photodynamic therapy has been shown to be an effective treatment more recently (Bains et al., 2004).
Prognosis
The visual loss and visual field defects in CCH are due to the location of the hemagioma and retinal complications including subretinal fluid, macular edema, and macular fibrosis and atrophy. Visual prognosis has been poor, with greater than 50% of eyes demonstrating a visual acuity of 20/200 or worse (Shields et al., 2001). DCH also has a poor visual prognosis as a result of the extensive choroidal involvement and secondary glaucoma (Grant et al., 2008).