RPA2 (replication protein A2, 32kDa)
2010-04-01 Anar KZ Murphy , James A Borowiec AffiliationDept of Biochemistry, New York University Cancer Institute, New York University School of Medicine, New York, New York 10016, USA
DNA/RNA
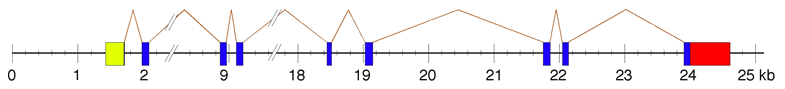
Description
Transcription
Pseudogene
Proteins
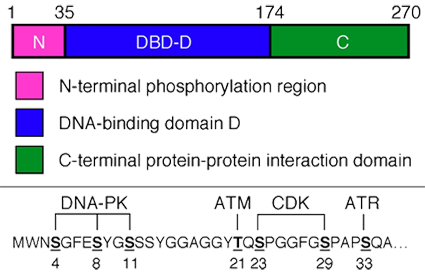
Description
Expression
Localisation
Function
1) RPA2 phosphorylation. The N-terminal 33 residues of RPA2 contain seven phosphorylation sites. In interphase cells, genotoxic stress (e.g., caused by chromosomal double-strand DNA breaks or DNA replication stress) induces RPA2 phosphorylation by members of the phosphatidylinositol 3-kinase-like kinase (PIKK; ATM, ATR, and DNA-PK) and cyclin-dependent kinases (CDK) families (reviewed in Binz et al., 2004). Mutation of particular RPA2 phosphorylation sites causes defects in homologous recombination (Lee et al., 2010), and Rad51 recruitment to nuclear repair foci (Anantha et al., 2008; Lee et al., 2010). Mutation of these sites also causes genomic instability in response to DNA replication stress induced by cellular treatment with hydroxyurea (Vassin et al., 2009). RPA phosphorylation also increases cell viability in response to DNA damage arising during mitosis (Anantha et al., 2008). Modification of sites in the phosphorylation region of RPA2 proceeds in a favored order in response to genotoxic stress (Anantha et al., 2007). The phosphorylation of individual RPA2 residues is dependent on the type of DNA damage or replication stress encountered (Anantha et al., 2007; Vassin et al., 2009). RPA2 is a substrate both for PP2A and PP4 phosphatases (Feng et al., 2009; Lee et al., 2010).
2) Involvement of RPA2 in protein-protein interactions. RPA2 interacts with the nucleotide excision repair factor XPA (He et al., 1995), base excision repair enzyme UNG2 (Mer et al., 2000), homologous recombination (HR) factor Rad52 (Mer et al., 2000), replication checkpoint protein Tipin (Unsal-Kacmaz et al., 2007), and the annealing helicase HARP/SMARCAL1 (Bansbach et al., 2009; Ciccia et al., 2009; Yuan et al., 2009). These interactions likely aid the multiple roles of RPA in facilitating DNA repair.
Homology
Mutations
Note
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18723675 | 2008 | RPA phosphorylation facilitates mitotic exit in response to mitotic DNA damage. | Anantha RW et al |
| 19793861 | 2009 | The annealing helicase SMARCAL1 maintains genome integrity at stalled replication forks. | Bansbach CE et al |
| 11479296 | 2001 | Functional analysis of the four DNA binding domains of replication protein A. The role of RPA2 in ssDNA binding. | Bastin-Shanower SA et al |
| 15279788 | 2004 | Replication protein A phosphorylation and the cellular response to DNA damage. | Binz SK et al |
| 10449415 | 1999 | The crystal structure of the complex of replication protein A subunits RPA32 and RPA14 reveals a mechanism for single-stranded DNA binding. | Bochkarev A et al |
| 11927569 | 2002 | Structure of the RPA trimerization core and its role in the multistep DNA-binding mechanism of RPA. | Bochkareva E et al |
| 19793862 | 2009 | The SIOD disorder protein SMARCAL1 is an RPA-interacting protein involved in replication fork restart. | Ciccia A et al |
| 19704001 | 2009 | Protein phosphatase 2A-dependent dephosphorylation of replication protein A is required for the repair of DNA breaks induced by replication stress. | Feng J et al |
| 7761458 | 1995 | Presence of antibodies to different subunits of replication protein A in autoimmune sera. | Garcia-Lozano R et al |
| 17361204 | 2007 | Replication protein A is an independent prognostic indicator with potential therapeutic implications in colon cancer. | Givalos N et al |
| 19942684 | 2010 | A naturally occurring human RPA subunit homolog does not support DNA replication or cell-cycle progression. | Haring SJ et al |
| 7700386 | 1995 | RPA involvement in the damage-recognition and incision steps of nucleotide excision repair. | He Z et al |
| 11313881 | 2001 | Expression analysis using DNA microarrays demonstrates that E2F-1 up-regulates expression of DNA replication genes including replication protein A2. | Kalma Y et al |
| 1320195 | 1992 | Binding properties of replication protein A from human and yeast cells. | Kim C et al |
| 18706093 | 2008 | Cia5d regulates a new fibroblast-like synoviocyte invasion-associated gene expression signature. | Laragione T et al |
| 20154705 | 2010 | A PP4 phosphatase complex dephosphorylates RPA2 to facilitate DNA repair via homologous recombination. | Lee DH et al |
| 11081631 | 2000 | Structural basis for the recognition of DNA repair proteins UNG2, XPA, and RAD52 by replication factor RPA. | Mer G et al |
| 19834905 | 2009 | Identification of molecular targets for selective elimination of TRAIL-resistant leukemia cells. From spots to in vitro assays using TOP15 charts. | Petrak J et al |
| 8804316 | 1996 | A hierarchy of SSB protomers in replication protein A. | Philipova D et al |
| 11895905 | 2002 | Autoimmunity to the M(r) 32,000 subunit of replication protein A in breast cancer. | Tomkiel JE et al |
| 17296725 | 2007 | The human Tim/Tipin complex coordinates an Intra-S checkpoint response to UV that slows replication fork displacement. | Unsal-Kaçmaz K et al |
| 19843584 | 2009 | Human RPA phosphorylation by ATR stimulates DNA synthesis and prevents ssDNA accumulation during DNA-replication stress. | Vassin VM et al |
| 19793864 | 2009 | The annealing helicase HARP protects stalled replication forks. | Yuan J et al |
Other Information
Locus ID:
NCBI: 6118
MIM: 179836
HGNC: 10290
Ensembl: ENSG00000117748
Variants:
dbSNP: 6118
ClinVar: 6118
TCGA: ENSG00000117748
COSMIC: RPA2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38416570 | 2024 | CSB and SMARCAL1 compete for RPA32 at stalled forks and differentially control the fate of stalled forks in BRCA2-deficient cells. | 0 |
| 38658755 | 2024 | Mechanism of single-stranded DNA annealing by RAD52-RPA complex. | 0 |
| 38416570 | 2024 | CSB and SMARCAL1 compete for RPA32 at stalled forks and differentially control the fate of stalled forks in BRCA2-deficient cells. | 0 |
| 38658755 | 2024 | Mechanism of single-stranded DNA annealing by RAD52-RPA complex. | 0 |
| 37831348 | 2023 | Upregulated RPA2 in endometrial tissues of repeated implantation failure patients impairs the endometrial decidualization. | 0 |
| 37831348 | 2023 | Upregulated RPA2 in endometrial tissues of repeated implantation failure patients impairs the endometrial decidualization. | 0 |
| 33784377 | 2021 | RPA2 winged-helix domain facilitates UNG-mediated removal of uracil from ssDNA; implications for repair of mutagenic uracil at the replication fork. | 5 |
| 34642383 | 2021 | hSSB2 (NABP1) is required for the recruitment of RPA during the cellular response to DNA UV damage. | 4 |
| 33784377 | 2021 | RPA2 winged-helix domain facilitates UNG-mediated removal of uracil from ssDNA; implications for repair of mutagenic uracil at the replication fork. | 5 |
| 34642383 | 2021 | hSSB2 (NABP1) is required for the recruitment of RPA during the cellular response to DNA UV damage. | 4 |
| 32856505 | 2020 | Dynamic elements of replication protein A at the crossroads of DNA replication, recombination, and repair. | 50 |
| 32856505 | 2020 | Dynamic elements of replication protein A at the crossroads of DNA replication, recombination, and repair. | 50 |
| 31582797 | 2019 | HERC2 regulates RPA2 by mediating ATR-induced Ser33 phosphorylation and ubiquitin-dependent degradation. | 11 |
| 31582797 | 2019 | HERC2 regulates RPA2 by mediating ATR-induced Ser33 phosphorylation and ubiquitin-dependent degradation. | 11 |
| 28007956 | 2017 | Upregulation of RPA2 promotes NF-κB activation in breast cancer by relieving the antagonistic function of menin on NF-κB-regulated transcription. | 14 |
Citation
Anar KZ Murphy ; James A Borowiec
RPA2 (replication protein A2, 32kDa)
Atlas Genet Cytogenet Oncol Haematol. 2010-04-01
Online version: http://atlasgeneticsoncology.org/gene/42146/rpa2-(replication-protein-a2-32kda)
