BNIP3 (Bcl-2/adenovirus E1B 19kD-interacting protein 3)
2007-10-01 Sang-Gi Paik , Hayyoung Lee AffiliationDepartment of Biology, School of Biosciences, Biotechnology, Chungnam National University, Daejeon 305-764, Korea.
DNA/RNA
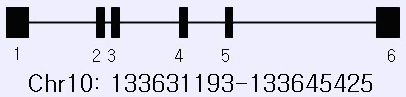
Description
14.23 kb on reverse strand; 6 exons
Transcription
mRNA in MCF-7 cells are 1.7kb (major) and 1.5 kb (minor) and 1.3 kb (minor).
Proteins
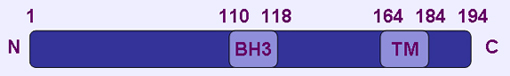
Domain map of BNIP3 protein; BH3 domain (Bcl-2 holomogy 3 domain); TM domain (transmembrane domain)
Description
194 amino acids; 1 BH3 domain and 1 TM domain; BH3 only Bcl2 family member. The TM domain and C-terminal tail are essential for mitochondrial membrane localization and proapoptotic function. The predicted molecular weight is 21.5 kDa. BNIP3 migrates as 30 kDa monomeric form and 60 kDa dimeric form on SDS-PAGE.
Expression
BNIP3 is detected in mouse oviduct, uterus, spleen, lung, stomach, brain, seminal, lacrimal, submaxillary, heart, kidney, liver. It can be detected in cell lines such as HeLa, 293T, RAW264.7 and K562 cells. Its expression can be induced in both normal and cancer tissues that experience hypoxia or hypoxia-like conditions. Other stimuli, such as nitric oxide or arsenic trioxide, are also reported to induce BNIP3 expression.
Localisation
Outer mitochondrial membrane
Function
Proapoptotic protein;
BNIP3 leads to opening of the mitochondrial permeability transition pore (PTP) thereby abolishing the proton electrochemical gradient and this is followed by chromatin condensation and DNA fragmentation. BNIP3 leads necrosis-like apoptosis. Unusually to the other Bcl-2 family proteins, the BNIP3-induced cell death depends not on BH3 domain but on C-terminal TM domain. BNIP3-induced cell death is known to be independent the nuclear translocation of AIF. However, whether caspase activation and cytochrome c release are involved in the cell death remains controversial. BNIP3 can induce autophagy. However whether the consequence of the autophagy is the cell death or survival remains to be established.
Since BNIP3 is induced by hypoxia through transcription factor HIF-1, it was postulated to play a role in hypoxia-induced cell death. Hypoxia-induced acidosis augments the proapoptotic function of BNIP3.
BNIP3 leads to opening of the mitochondrial permeability transition pore (PTP) thereby abolishing the proton electrochemical gradient and this is followed by chromatin condensation and DNA fragmentation. BNIP3 leads necrosis-like apoptosis. Unusually to the other Bcl-2 family proteins, the BNIP3-induced cell death depends not on BH3 domain but on C-terminal TM domain. BNIP3-induced cell death is known to be independent the nuclear translocation of AIF. However, whether caspase activation and cytochrome c release are involved in the cell death remains controversial. BNIP3 can induce autophagy. However whether the consequence of the autophagy is the cell death or survival remains to be established.
Since BNIP3 is induced by hypoxia through transcription factor HIF-1, it was postulated to play a role in hypoxia-induced cell death. Hypoxia-induced acidosis augments the proapoptotic function of BNIP3.
Homology
The close homologue: BNIP3L/ BNIP3a/ Nix/ B5 (8q21)
The BH3-only Bcl2 family members: BBC3/PUMA (19q13), BCL2L11/BIM/BOD (2Q13), BID (22q11), BIK/NBK/BBC1 (22q13), BLK (8q23), BMF (15Q14), HRK/DP5/BID3 (12q24), PMAIP1/NOXA (18q21)
The BH3-only Bcl2 family members: BBC3/PUMA (19q13), BCL2L11/BIM/BOD (2Q13), BID (22q11), BIK/NBK/BBC1 (22q13), BLK (8q23), BMF (15Q14), HRK/DP5/BID3 (12q24), PMAIP1/NOXA (18q21)
Implicated in
Entity name
Pancreatic cancer
Prognosis
Pancreatic adenocarcinoma is highly resistant to chemical and radiation therapy, and has an extremely poor prognosis. Reduced expression of BNIP3 increased resistance to gemcitabine and 5-fluoro-uracil (5-FU) and showed a good correlation with reduced patient survival.
Oncogenesis
In most cases of pancreatic adenocarcinoma, BNIP3 expression was not detected even in response to hypoxia. The promoter of BNIP3 is located within a CpG island and is methylated in most pancreatic cancer cell lines. Restoration of BNIP3 expression by the methyltransferase inhibitor, 5-aza-deoxycytidine, induced death of pancreatic cancer cells in response to hypoxia.
Entity name
Colorectal cancer
Oncogenesis
Methylation of BNIP3 in 66% of primary colorectal cancer
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 15942716 | 2005 | Upregulation of BNIP3 by 5-aza-2'-deoxycytidine sensitizes pancreatic cancer cells to hypoxia-mediated cell death. | Abe T et al |
| 15837765 | 2005 | Intrinsic chemoresistance to gemcitabine is associated with decreased expression of BNIP3 in pancreatic cancer. | Akada M et al |
| 16954213 | 2006 | Activation of Ras up-regulates pro-apoptotic BNIP3 in nitric oxide-induced cell death. | An HJ et al |
| 16799636 | 2007 | Selective silencing of the hypoxia-inducible factor 1 target gene BNIP3 by histone deacetylation and methylation in colorectal cancer. | Bacon AL et al |
| 7954800 | 1994 | Adenovirus E1B 19 kDa and Bcl-2 proteins interact with a common set of cellular proteins. | Boyd JM et al |
| 10922063 | 2000 | Expression of the gene encoding the proapoptotic Nip3 protein is induced by hypoxia. | Bruick RK et al |
| 9396766 | 1997 | The E1B 19K/Bcl-2-binding protein Nip3 is a dimeric mitochondrial protein that activates apoptosis. | Chen G et al |
| 15856026 | 2005 | Loss of BNIP3 expression is a late event in pancreatic cancer contributing to chemoresistance and worsened prognosis. | Erkan M et al |
| 11831844 | 2002 | The carboxy terminal C-tail of BNip3 is crucial in induction of mitochondrial permeability transition in isolated mitochondria. | Kim JY et al |
| 12226479 | 2002 | Hypoxia and acidosis activate cardiac myocyte death through the Bcl-2 family protein BNIP3. | Kubasiak LA et al |
| 16511341 | 2006 | Regulation of BNIP3 in normal and cancer cells. | Lee H et al |
| 15709167 | 2005 | Aberrant methylation and silencing of the BNIP3 gene in colorectal and gastric cancer. | Murai M et al |
| 15289340 | 2004 | Silencing of the hypoxia-inducible cell death protein BNIP3 in pancreatic cancer. | Okami J et al |
| 10625696 | 2000 | BNIP3 heterodimerizes with Bcl-2/Bcl-X(L) and induces cell death independent of a Bcl-2 homology 3 (BH3) domain at both mitochondrial and nonmitochondrial sites. | Ray R et al |
| 10891486 | 2000 | BNIP3 and genetic control of necrosis-like cell death through the mitochondrial permeability transition pore. | Vande Velde C et al |
| 15623420 | 2005 | BNip3 and signal-specific programmed death in the heart. | Webster KA et al |
| 9575197 | 1998 | Adenovirus E1B-19K/BCL-2 interacting protein BNIP3 contains a BH3 domain and a mitochondrial targeting sequence. | Yasuda M et al |
| 15358175 | 2004 | Nitric oxide induces BNIP3 expression that causes cell death in macrophages. | Yook YH et al |
Other Information
Locus ID:
NCBI: 664
MIM: 603293
HGNC: 1084
Ensembl: ENSG00000176171
Variants:
dbSNP: 664
ClinVar: 664
TCGA: ENSG00000176171
COSMIC: BNIP3
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000176171 | ENST00000368636 | Q12983 |
| ENSG00000176171 | ENST00000540159 | F6RP06 |
| ENSG00000176171 | ENST00000633835 | A0A0J9YW18 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37876279 | 2024 | Excessive BNIP3- and BNIP3L-dependent mitophagy underlies the pathogenesis of FBXL4-mutated mitochondrial DNA depletion syndrome. | 0 |
| 38142898 | 2024 | BNIP3 and DAPK1 methylation in peripheral blood leucocytes are noninvasive biomarkers for gastric cancer. | 0 |
| 38199116 | 2024 | Autophagy induced by hypoxia in pulpitis is mediated by HIF-1α/BNIP3. | 1 |
| 38211650 | 2024 | CD30 protects EBV-positive diffuse large B-cell lymphoma cells against mitochondrial dysfunction through BNIP3-mediated mitophagy. | 1 |
| 38443598 | 2024 | The Mst1/2-BNIP3 axis is required for mitophagy induction and neuronal viability under mitochondrial stress. | 1 |
| 38519771 | 2024 | Mitophagy mediated by BNIP3 and NIX protects against ferroptosis by downregulating mitochondrial reactive oxygen species. | 1 |
| 37876279 | 2024 | Excessive BNIP3- and BNIP3L-dependent mitophagy underlies the pathogenesis of FBXL4-mutated mitochondrial DNA depletion syndrome. | 0 |
| 38142898 | 2024 | BNIP3 and DAPK1 methylation in peripheral blood leucocytes are noninvasive biomarkers for gastric cancer. | 0 |
| 38199116 | 2024 | Autophagy induced by hypoxia in pulpitis is mediated by HIF-1α/BNIP3. | 1 |
| 38211650 | 2024 | CD30 protects EBV-positive diffuse large B-cell lymphoma cells against mitochondrial dysfunction through BNIP3-mediated mitophagy. | 1 |
| 38443598 | 2024 | The Mst1/2-BNIP3 axis is required for mitophagy induction and neuronal viability under mitochondrial stress. | 1 |
| 38519771 | 2024 | Mitophagy mediated by BNIP3 and NIX protects against ferroptosis by downregulating mitochondrial reactive oxygen species. | 1 |
| 36628478 | 2023 | The mitophagy receptor BNIP3 is critical for the regulation of metabolic homeostasis and mitochondrial function in the nucleus pulposus cells of the intervertebral disc. | 8 |
| 36795401 | 2023 | The outer mitochondrial membrane protein TMEM11 demarcates spatially restricted BNIP3/BNIP3L-mediated mitophagy. | 10 |
| 36928344 | 2023 | HIF1α-BNIP3-mediated mitophagy protects against renal fibrosis by decreasing ROS and inhibiting activation of the NLRP3 inflammasome. | 8 |
Citation
Sang-Gi Paik ; Hayyoung Lee
BNIP3 (Bcl-2/adenovirus E1B 19kD-interacting protein 3)
Atlas Genet Cytogenet Oncol Haematol. 2007-10-01
Online version: http://atlasgeneticsoncology.org/gene/822/bnip3
