DDIT4 (DNA-damage-inducible transcript 4)
2012-12-01 Silvia Vega-Rubin-de-Celis , James Brugarolas AffiliationDepartments of Internal Medicine, Developmental Biology, Simmons Comprehensive Cancer Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA
Identity
DNA/RNA
Description
Transcription
Proteins
Note
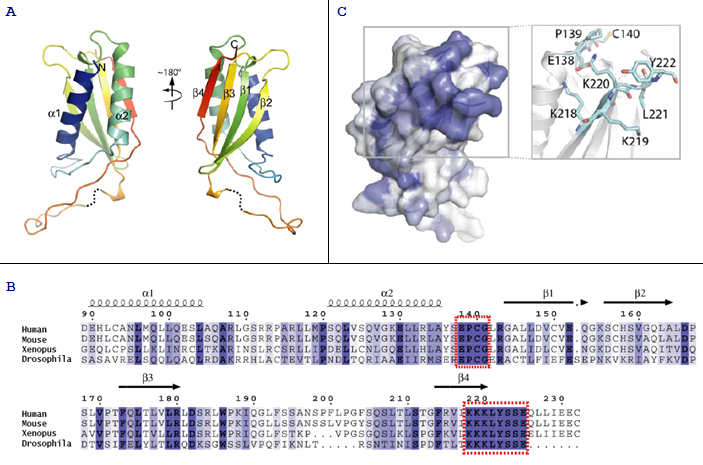
Description
A conserved surface patch that is essential for REDD1 function has been identified. This patch is formed by two regions: a loop between helix α2 and strand β1 (residues 138-141) and residues 218-225, which encompass the C-terminal portion of the strand β4.
REDD1 is an unstable protein, with an estimated half-life of 5-10 min (Kimball et al., 2008; Katiyar et al., 2009). REDD1 has been shown to be phosphorylated at T23 and T25 and phosphorylation at these sites has been postulated to regulate REDD1 half-life. These sites are in an N-terminal region that is dispensable for mTORC1 inhibition (Vega-Rubin-de-Celis et al., 2010). Based on in vitro kinase assays, it was postulated that the kinase implicated in REDD1 phophorylation is Glycogen Sinthase Kinase 3β (GSK3β). HA-GSK3β isolated from transfected HEK293 cells can phosphorylate GST-REDD1 (Katiyar et al., 2009). However, whether GSK3β phosphorylates endogenous REDD1 is still unknown. How these phosphorylation events are implicated in REDD1 stability is also unclear. Katiyar et al. reported a modest increase in REDD1 half-life upon treatment with LiCl, a GSK3β inhibitor, in HEK293 cells. In addition, REDD1 T23A/T25A mutants transfected in HEK293 cells are more stable than wild-type REDD1. However, 35S Methionine/Cysteine pulse-chase experiments in HeLa cells suggested that REDD1 wild-type and the T23A/T25A mutants have a similar half-life and LiCl had no effect on the half-life of endogenous REDD1 in these cells (Vega-Rubin-de-Celis and Brugarolas, unpublished results).
REDD1 half-life has been proposed to be regulated by the CUL4A-DDB1-ROC1-βTRCP-E3 ubiquitin ligase complex (Katiyar et al., 2009). In overexpression experiments in HEK293 cells, Flag-REDD1 was found to interact with the complex components Myc-CUL4A, AU1-DDB1 and HA-βTRCP. In addition, endogenous REDD1 was shown to bind to DDB1 and βTRCP in MCF-7 cells exposed to hypoxia and the proteasome inhibitor MG132 (Katiyar et al., 2009). Furthermore, knock-down of βTRCP by siRNA in MCF-7 cells increased the REDD1 half-life to 25 minutes (Katiyar et al., 2009). Knock-down of other complex components (CUL4A and DDB1) also increased REDD1 half-life, but to a lesser extent (10-15 minutes) (Katiyar et al., 2009).
Several large-scale proteomic studies showed that REDD1 is ubiquitinated at K129 (Meierhofer et al., 2008; Xu et al., 2010; Danielsen et al., 2011; Kim et al., 2011; Lee et al., 2011; Shi et al., 2011; Wagner et al., 2011). Whether this modification affects REDD1 stability (or function) is unknown and K129 is not conserved across species.
Thioredoxin-Interacting protein (TXNIP) has been shown to bind to REDD1 and TXNIP overexpression in HeLa and H1299 cells increased REDD1 levels (Jin et al., 2011). However, these experiments were performed with ectopically expressed protein and whether the endogenous proteins interact and whether TXNIP regulates REDD1 stability remains to be determined.
A yeast-two hybrid screen of a human leukocyte library reported several putative binding proteins (Gery et al., 2007), and other studies identified other REDD1 interacting proteins including TXNIP (DeYoung et al., 2008; Jin et al., 2011). However, none have been validated with endogenous REDD1 protein.
Expression
REDD1 mRNA is broadly induced in response to hypoxia (Shoshani et al., 2002; Wolff et al., 2011). Using mice with a gene trap reporter (β-geo) inserted in Redd1 intron 2, Wolff et al. showed that Redd1 is induced after 1h of hypoxia in most of the tissues analyzed, with the exception of cardiac and skeletal muscle. Redd1 was broadly induced in endothelial cells as well as in cells in the crypts of the small bowel, bronchial epithelial cells, cells in zone 3 in the liver, cells lining the collecting ducts of the kidney, red pulp and histiocytes in the spleen and Purkinje cells in the cerebellum (Wolff et al., 2011).
REDD1 is a hypoxia inducible factor (HIF) target (Shoshani et al., 2002). HIF-1 and -2 are heterodimeric transcription factors containing an α-subunit that is regulated by oxygen levels through hydroxylation (Ivan et al., 2001; Jaakkola et al., 2001; Masson et al., 2001; Yu et al., 2001) and a β-subunit (also called aryl hydrocarbon receptor nuclear translocator, ARNT) that is stable (Semenza, 2006). Hydroxylated HIFα is recognized by an ubiquitin ligase complex containing the von Hippel-Lindau (pVHL) tumor suppressor protein (Cockman et al., 2000; Kamura et al., 2000; Ohh et al., 2000; Tanimoto et al., 2000). It has been shown in cell lines and mouse models that REDD1 induction is VHL and HIF-dependent (Kucejova et al., 2011). REDD1 is a target of both HIF1α and HIF2α, as assessed by ChIP assays and siRNA experiments in clear cell renal cell carcinoma (ccRCC) cell lines 786-O, A498 and Caki-2 (Kucejova et al., 2011).
MEFs deficient for ataxia telangiectasia mutated (ATM) are unable to induce HIF1α (and REDD1) in response to hypoxia suggesting that, at least in some contexts, ATM may be required for HIFα stabilization (Cam et al., 2010).
Besides hypoxia, REDD1 is also induced in response to a variety of other stress conditions, including ER stress (Wang et al., 2003; Whitney et al., 2009), osmotic stress (Wang et al., 2003) and in response to DNA-damaging agents (Ellisen et al., 2002; Lin et al., 2005a). Other REDD1 inducers include glucocorticoids (Wang et al., 2003; Boldizsar et al., 2006) and all-trans-retinoic acid (ATRA) (Gery et al., 2007).
Apart from HIF1 and 2, other transcription factors have been implicated in REDD1 induction:
- ATF4 (activating transcription factor 4): REDD1 is induced under ER stress conditions in an ATF4-dependent manner (Whitney et al., 2009). Experiments performed in HepG2 cells showed REDD1 induction with the ER stress inducers Thapsigargin and Tunicamycin. This induction was shown to be protein kinase RNA-like ER kinase (PERK) and ATF4-dependent based on studies on PERK or ATF4-deficient MEFs. In addition, ectopically overexpressed ATF4 was sufficient to induce REDD1 expression in HEK293.
- CREBP (CCAAT/enhancer-binding protein): DNA-damaging agent MMS induces REDD1 mRNA in HaCaT cells in a CREBP-dependent manner (Lin et al., 2005a). Luciferase reporter assays identified a MMS-responsive region within -1057/-981 (Lin et al., 2005a) which included a putative CREBP site at -1009/-999, and CREBP was found to bind to this sequence in EMSA.
- Elk1: REDD1 mRNA is induced in HaCaT cells exposed to arsenite, in a Elk1 and CCAAT/enhancer-binding protein (CREBP)-dependent manner (Lin et al., 2005b). Luciferase reporter assays in HaCaT cells mapped the arsenite-responsive region to the -1057/-981 (Lin et al., 2005b), and EMSA assays showed CREBP binding to that region.
- NFATc3 (nuclear factor of activated T-cell): in intestinal cells REDD1 is induced by the NFAT activators PMA and A23187, and siRNA knockdown of NFATc3 but not other family members compromised PMA/A23187-induced REDD1 expression (Zhou et al., 2012). Luciferase-reporter assays showed that NFATc3 was sufficient to activate expression of REDD1 promoter sequences and ChIP assays in HT29 cells showed binding of NFATc3 (-2931/-97) (Zhou et al., 2012).
- p53: MEFs treated with ionizing radiation induced REDD1 in a p53-dependent manner, and overexpression of p53 in SAOS and U2OS cells induced REDD1 (Ellisen et al., 2002). Luciferase reporter assays in U2OS cells identified a p53-responsive element at -600 bp (Ellisen et al., 2002).
- Sp1: REDD1 levels are increased in high cell densities in a Sp-1 dependent manner (Jin et al., 2007). Sp1 siRNA in HeLa cells abolished REDD1 induction by high cell density. Sp1 can also contribute to REDD1 induction in hypoxia or in response to hypoxia mymetics (Jin et al., 2007), and siRNA experiments compromised CoCl2-dependent REDD1 induction. The putative Sp1 binding site was mapped to -476/-446 of the REDD1 promoter by luciferase-reporter assays in HeLa cells (Jin et al., 2007).
The REDD1 promoter region contains putative target sequences for other transcription factors, including NFkB and hepatocyte-nuclear factor 4 (HNF4) (Lin et al., 2005b).
Localisation
Function
REDD1 has a paralogue, REDD2 (DDIT4-like (DDIT4-L) or RTP801-like (RTP801-L)). REDD1 and REDD2 appear to be have originated from a gene duplication event that occurred independently in humans and insects (Vega-Rubin-de-Celis et al., 2010). However, unlike Redd1, Redd2 is not induced by hypoxia, at least in MEFs (Wolff et al., 2011). Nevertheless, like REDD1, REDD2 overexpression is sufficient to inhibit mTORC1 (Brugarolas et al., 2004).
The Drosophila orthologues, scylla and charybdis, were identified in a screen to isolate inhibitors of an increased cell size phenotype generated by simultaneously expressing PDK1 and Akt (Reiling and Hafen, 2004). Both genes contain consensus hypoxia-response elements (HRE) sequences (Reiling and Hafen, 2004). Scylla expression is also induced in response to hypoxia in the larval fat body and the gut. Charybdis mRNA was not induced in the fat body and only slightly in the midgut (Reiling and Hafen, 2004). In addition, charybdis is not induced through overexpression of constitutively active Drosophila HIFα (sima) or Hifβ (tgo) (Reiling and Hafen, 2004).
Even though REDD1 induction broadly inhibits mTORC1, under some conditions (and for reasons that are not understood), this is not the case in ccRCC. VHL is inactivated in a majority of ccRCC cases, leading to stabilization of HIF1/HIF2 and increased REDD1 levels. However, REDD1 does not appear to inhibit mTORC1, and mTORC1 is frequently activated in ccRCC (Kucejova et al., 2011).
Hypoxia-induced mTORC1 inhibition is dependent on the tuberous sclerosis complex 1 and 2 proteins, which form a complex (TSC1/TSC2). This has been shown in a variety of systems. Tsc2-/- MEFs fail to inhibit mTORC1 when exposed to hypoxia or to ectopic Redd1, but they regain their ability to inhibit the pathway after Tsc2 reconstitution (Brugarolas et al., 2004; Corradetti et al., 2005; Sofer et al., 2005). Depletion of TSC2 with siRNA in U2OS, HeLa, HEK293 and MCF10A cells also compromises mTORC1 inhibition by hypoxia or REDD1 overexpression (Brugarolas et al., 2004; Connolly et al., 2006; Vega-Rubin-de-Celis et al., 2010).
However, hypoxia-induced mTORC1 inhibition can also occur independently of mTORC1. In hepatocytes, mTORC1 inhibition by hypoxia is REDD1-independent. In this cell type, hypoxia signals are transduced in a manner that is also independent of Tsc1/Tsc2 and Hif (Wolff et al., 2011).
It was suggested that REDD1 acts by sequestering 14-3-3 proteins from TSC2 thereby activating TSC2 and inhibiting mTORC1 (DeYoung et al., 2008). However, how REDD1 would selectively sequester TSC2-bound 14-3-3 versus the whole 14-3-3 pool in the cell, which is quite large, is not clear. Furthermore, the putative 14-3-3 binding site in REDD1 does not conform to any of 14-3-3 binding sites known (Vega-Rubin-de-Celis et al., 2010), and endogenous REDD1 does not seemingly bind to 14-3-3 proteins. Moreover, the postulated 14-3-3 binding motif in REDD1 (133RLAYSEP139) is not conserved, and mutations of R133 and S137 that might have expected to be critical for 14-3-3 binding do not abrogate REDD1 function (Vega-Rubin-de-Celis et al., 2010).
REDD1 is developmentally regulated in both flies and in mice, but Redd1 is dispensable for development in mice (Brafman et al., 2004; Yoshida et al., 2010; Molitoris et al., 2011; Wolff et al., 2011). In addition, loss of both scylla and charybdis does not cause lethality in flies. However, their loss severely compromised larval survival in hypoxia conditions (Reiling and Hafen, 2004). Interestingly, disruption of Redd1 in mice does not result in a compensatory induction by the paralogue Redd2, which has been shown to also inhibit mTORC1 in overexpression experiments in HeLa cells (Brugarolas et al., 2004).
Mutations
Note
Somatic
A mutation in prostate cancer (c.253A>C; p.S85R) has been described (Sanger Institute, COSMIC database). It is unknown whether the mutation was somatically acquired and according to PolyPhen-2 it is probably damaging. An additional somatically acquired mutation was found in prostate carcinoma (c.253A>C); however, it is a synonymous substitution (p.L96L) (Sanger Institute, COSMIC database).
Two mutations have been detected in lung squamous cell carcinoma (G75R and A37V) (Hammerman et al., 2012, cBIO Cancer Genomics). They are both somatically acquired (Sanger Institute, COSMIC database), and according to PolyPhen-2 they are predicted to be benign.
Two mutations have been identified in uterine corpus endometrioid carcinoma (H91R and R143W) (cBIO Cancer Genomics). They are somatically acquired, (Sanger Institute, COSMIC database) and according to PolyPhen-2 they are predicted to be benign (H91R) or probably damaging (R143W).
A somatically acquired mutation was identified in two samples of lung carcinoma (c.4C>T; p.P2S) (Sanger Institute, COSMIC database). Pro2 residue is in a dispensable region in REDD1; however, according to Polyphen2 is probably damaging.
A synonymous somatically acquired substitution mutation was also described in lung carcinoma (c.561C>G; p.P187P) (Sanger Institute, COSMIC database).
A somatically acquired mutation was identified in cervix carcinoma (c.79G>C; p.D27H) (Sanger Institute, COSMIC database). It is in a dispensable region in REDD1 and predicted to be benign based on Polyphen2.
In urinary tract carcinoma samples a somatically acquired mutation was found (c.671C>T; p.S224L) (Sanger Institute, COSMIC database). It is predicted to be probably damaging by Polyphen2.
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 17113916 | 2006 | Low glucocorticoid receptor (GR), high Dig2 and low Bcl-2 expression in double positive thymocytes of BALB/c mice indicates their endogenous glucocorticoid hormone exposure. | Boldizsár F et al |
| 15452091 | 2004 | Inhibition of oxygen-induced retinopathy in RTP801-deficient mice. | Brafman A et al |
| 15545625 | 2004 | Regulation of mTOR function in response to hypoxia by REDD1 and the TSC1/TSC2 tumor suppressor complex. | Brugarolas J et al |
| 21095582 | 2010 | mTORC1 signaling under hypoxic conditions is controlled by ATM-dependent phosphorylation of HIF-1α. | Cam H et al |
| 10823831 | 2000 | Hypoxia inducible factor-alpha binding and ubiquitylation by the von Hippel-Lindau tumor suppressor protein. | Cockman ME et al |
| 16648488 | 2006 | Hypoxia inhibits protein synthesis through a 4E-BP1 and elongation factor 2 kinase pathway controlled by mTOR and uncoupled in breast cancer cells. | Connolly E et al |
| 15632201 | 2005 | The stress-inducted proteins RTP801 and RTP801L are negative regulators of the mammalian target of rapamycin pathway. | Corradetti MN et al |
| 21139048 | 2011 | Mass spectrometric analysis of lysine ubiquitylation reveals promiscuity at site level. | Danielsen JM et al |
| 18198340 | 2008 | Hypoxia regulates TSC1/2-mTOR signaling and tumor suppression through REDD1-mediated 14-3-3 shuttling. | DeYoung MP et al |
| 12453409 | 2002 | REDD1, a developmentally regulated transcriptional target of p63 and p53, links p63 to regulation of reactive oxygen species. | Ellisen LW et al |
| 17379067 | 2007 | RTP801 is a novel retinoic acid-responsive gene associated with myeloid differentiation. | Gery S et al |
| 22960745 | 2012 | Comprehensive genomic characterization of squamous cell lung cancers. | |
| 20176937 | 2010 | Negative feedback control of HIF-1 through REDD1-regulated ROS suppresses tumorigenesis. | Horak P et al |
| 11292862 | 2001 | HIFalpha targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. | Ivan M et al |
| 11292861 | 2001 | Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. | Jaakkola P et al |
| 17307335 | 2007 | Hypoxic condition- and high cell density-induced expression of Redd1 is regulated by activation of hypoxia-inducible factor-1alpha and Sp1 through the phosphatidylinositol 3-kinase/Akt signaling pathway. | Jin HO et al |
| 21460850 | 2011 | TXNIP potentiates Redd1-induced mTOR suppression through stabilization of Redd1. | Jin HO et al |
| 10973499 | 2000 | Activation of HIF1alpha ubiquitination by a reconstituted von Hippel-Lindau (VHL) tumor suppressor complex. | Kamura T et al |
| 19557001 | 2009 | REDD1, an inhibitor of mTOR signalling, is regulated by the CUL4A-DDB1 ubiquitin ligase. | Katiyar S et al |
| 21906983 | 2011 | Systematic and quantitative assessment of the ubiquitin-modified proteome. | Kim W et al |
| 18070882 | 2008 | Rapid turnover of the mTOR complex 1 (mTORC1) repressor REDD1 and activation of mTORC1 signaling following inhibition of protein synthesis. | Kimball SR et al |
| 21798997 | 2011 | Interplay between pVHL and mTORC1 pathways in clear-cell renal cell carcinoma. | Kucejova B et al |
| 21987572 | 2011 | Ubiquitin ligase substrate identification through quantitative proteomics at both the protein and peptide levels. | Lee KA et al |
| 15751966 | 2005 | Induction of a cell stress response gene RTP801 by DNA damaging agent methyl methanesulfonate through CCAAT/enhancer binding protein. | Lin L et al |
| 16008523 | 2005 | Arsenite induces a cell stress-response gene, RTP801, through reactive oxygen species and transcription factors Elk-1 and CCAAT/enhancer-binding protein. | Lin L et al |
| 19118169 | 2008 | RTP801 is induced in Parkinson's disease and mediates neuron death by inhibiting Akt phosphorylation/activation. | Malagelada C et al |
| 11566883 | 2001 | Independent function of two destruction domains in hypoxia-inducible factor-alpha chains activated by prolyl hydroxylation. | Masson N et al |
| 21909097 | 2011 | Chemical inhibition of RNA viruses reveals REDD1 as a host defense factor. | Mata MA et al |
| 18781797 | 2008 | Quantitative analysis of global ubiquitination in HeLa cells by mass spectrometry. | Meierhofer D et al |
| 21733849 | 2011 | Glucocorticoid elevation of dexamethasone-induced gene 2 (Dig2/RTP801/REDD1) protein mediates autophagy in lymphocytes. | Molitoris JK et al |
| 10878807 | 2000 | Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel-Lindau protein. | Ohh M et al |
| 15545626 | 2004 | The hypoxia-induced paralogs Scylla and Charybdis inhibit growth by down-regulating S6K activity upstream of TSC in Drosophila. | Reiling JH et al |
| 16740642 | 2006 | Regulation of physiological responses to continuous and intermittent hypoxia by hypoxia-inducible factor 1. | Semenza GL et al |
| 20972266 | 2011 | A data set of human endogenous protein ubiquitination sites. | Shi Y et al |
| 11884613 | 2002 | Identification of a novel hypoxia-inducible factor 1-responsive gene, RTP801, involved in apoptosis. | Shoshani T et al |
| 15988001 | 2005 | Regulation of mTOR and cell growth in response to energy stress by REDD1. | Sofer A et al |
| 10944113 | 2000 | Mechanism of regulation of the hypoxia-inducible factor-1 alpha by the von Hippel-Lindau tumor suppressor protein. | Tanimoto K et al |
| 20166753 | 2010 | Structural analysis and functional implications of the negative mTORC1 regulator REDD1. | Vega-Rubin-de-Celis S et al |
| 21890473 | 2011 | A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles. | Wagner SA et al |
| 12736248 | 2003 | Dexamethasone-induced gene 2 (dig2) is a novel pro-survival stress gene induced rapidly by diverse apoptotic signals. | Wang Z et al |
| 19114033 | 2009 | ATF4 is necessary and sufficient for ER stress-induced upregulation of REDD1 expression. | Whitney ML et al |
| 21383064 | 2011 | Cell-type-dependent regulation of mTORC1 by REDD1 and the tumor suppressors TSC1/TSC2 and LKB1 in response to hypoxia. | Wolff NC et al |
| 20639865 | 2010 | Global analysis of lysine ubiquitination by ubiquitin remnant immunoaffinity profiling. | Xu G et al |
| 20473305 | 2010 | Rtp801, a suppressor of mTOR signaling, is an essential mediator of cigarette smoke-induced pulmonary injury and emphysema. | Yoshida T et al |
| 11504942 | 2001 | HIF-1alpha binding to VHL is regulated by stimulus-sensitive proline hydroxylation. | Yu F et al |
| 22696685 | 2012 | Nuclear factor of activated T-cell c3 inhibition of mammalian target of rapamycin signaling through induction of regulated in development and DNA damage response 1 in human intestinal cells. | Zhou Y et al |
Other Information
Locus ID:
NCBI: 54541
MIM: 607729
HGNC: 24944
Ensembl: ENSG00000168209
Variants:
dbSNP: 54541
ClinVar: 54541
TCGA: ENSG00000168209
COSMIC: DDIT4
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000168209 | ENST00000307365 | Q9NX09 |
| ENSG00000168209 | ENST00000307365 | A0A024QZQ6 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38507057 | 2024 | Pan-cancer analysis of DDIT4 identifying its prognostic value and function in acute myeloid leukemia. | 0 |
| 38546584 | 2024 | NLRP3 Inflammasome Priming in the Retina of Diabetic Mice Requires REDD1-Dependent Activation of GSK3β. | 2 |
| 38507057 | 2024 | Pan-cancer analysis of DDIT4 identifying its prognostic value and function in acute myeloid leukemia. | 0 |
| 38546584 | 2024 | NLRP3 Inflammasome Priming in the Retina of Diabetic Mice Requires REDD1-Dependent Activation of GSK3β. | 2 |
| 36383638 | 2023 | PERK/ATF4-dependent expression of the stress response protein REDD1 promotes proinflammatory cytokine expression in the heart of obese mice. | 6 |
| 36453700 | 2023 | DDIT4 promotes malignancy of head and neck squamous cell carcinoma. | 5 |
| 37016173 | 2023 | DDIT4 Facilitates Lymph Node Metastasis via the Activation of NF-κB Pathway and Epithelial-Mesenchymal Transition. | 1 |
| 37392853 | 2023 | REDD1-dependent GSK3β dephosphorylation promotes NF-κB activation and macrophage infiltration in the retina of diabetic mice. | 2 |
| 37483165 | 2023 | REDD1 (regulated in development and DNA damage 1) modulates the glucocorticoid receptor function in keratinocytes. | 0 |
| 37938616 | 2023 | Nuclear overexpression of DNA damage-inducible transcript 4 (DDIT4) is associated with aggressive tumor behavior in patients with pancreatic tumors. | 0 |
| 36383638 | 2023 | PERK/ATF4-dependent expression of the stress response protein REDD1 promotes proinflammatory cytokine expression in the heart of obese mice. | 6 |
| 36453700 | 2023 | DDIT4 promotes malignancy of head and neck squamous cell carcinoma. | 5 |
| 37016173 | 2023 | DDIT4 Facilitates Lymph Node Metastasis via the Activation of NF-κB Pathway and Epithelial-Mesenchymal Transition. | 1 |
| 37392853 | 2023 | REDD1-dependent GSK3β dephosphorylation promotes NF-κB activation and macrophage infiltration in the retina of diabetic mice. | 2 |
| 37483165 | 2023 | REDD1 (regulated in development and DNA damage 1) modulates the glucocorticoid receptor function in keratinocytes. | 0 |
Citation
Silvia Vega-Rubin-de-Celis ; James Brugarolas
DDIT4 (DNA-damage-inducible transcript 4)
Atlas Genet Cytogenet Oncol Haematol. 2012-12-01
Online version: http://atlasgeneticsoncology.org/gene/45802/ddit4
