HDAC1 (histone deacetylase 1)
2017-08-01 Emanuely Silva Chrun , Filipe Modolo , Filipe Ivan Daniel AffiliationFederal University of Santa Catarina, Florianopolis, Santa Catarina, [email protected] (ESC), Pathology Department, Federal University of Santa Catarina, Florianopolis, SC, Brazil. [email protected]; [email protected] (FM, FID)
Abstract
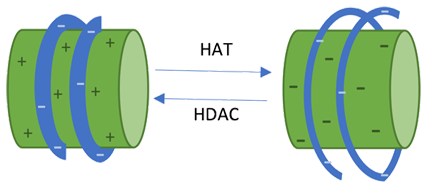
HDAC1 is a Class I histone deacetylase (HDACs) encoded by this gene. HDACs remove acetyl group from amino-terminal tail of histone lysine recovering the positive charge of histone tail, permitting interactions between negatively charged DNA and histone protein, resulting in chromatin structure condensation which is associated with gene repression. Overexpression of this protein has been related to several malignancies, controlling tumour suppressor genes expression, feasible prognostics factor, and medications target. This paper propound data about HDAC1 gene, its protein encoded and function.
DNA/RNA
Description
Proteins
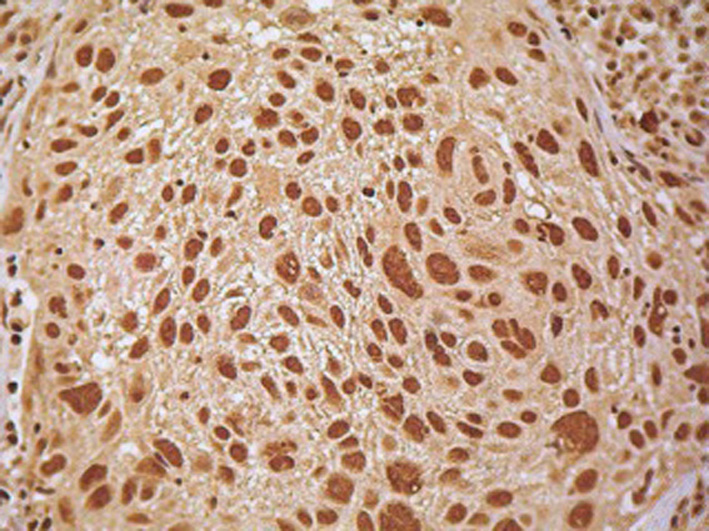
Expression
Localisation
Function
Homology
Table 1. Comparative identity of human HDAC1 and its homologs in other species).
Gene | Identity | ||
| H. sapiens vs. | Symbol | Protein | DNA |
| P. troglogytes | HDAC1 | 99.8 | 99.4 |
| P. troglogytes | HDAC1 | 99.6 | 99.4 |
| M. mulata | LOC708441 | 99.8 | 97.8 |
| C. lupus | HDAC1 | 99.2 | 94.1 |
| B. taurus | HDAC1 | 99.2 | 93.9 |
| M. musculus | Hdac1 | 99.4 | 90.5 |
| R. norvegicus | Hdac1 | 99.2 | 90.9 |
| G. gallus | Hdac1 | 93.3 | 79.2 |
| D. melanogaster | Rpd3 | 82.1 | 72.2 |
| A. gambiae | AgaP_AGAP006511 | 83.1 | 72.8 |
| C. elegans | hda-3 | 69.5 | 63.5 |
| S. cerevisiae | RPD3 | 64.8 | 60.3 |
| K. lactis | KLLA0E01981g | 65.4 | 61.2 |
| E. gossypii | AGOS_AGR395W | 65.0 | 62.2 |
| S. pombe | clr6 | 64.5 | 63.2 |
| M. oryzae | MGG_05857 | 66.7 | 63.9 |
| N. crassa | NCU00824 | 66.1 | 62.8 |
| A. thaliana | HD1 | 67.6 | 63.4 |
| O. sativa | Os02g02124900 | 68.3 | 64.0 |
| O. sativa | Os06g0583400 | 69.2 | 65.4 |
Mutations
Note
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 20415600 | 2010 | Class I histone deacetylases 1, 2 and 3 are highly expressed in classical Hodgkin's lymphoma. | Adams H et al |
| 22147512 | 2012 | Recruitment of histone deacetylases HDAC1 and HDAC2 by the transcriptional repressor ZEB1 downregulates E-cadherin expression in pancreatic cancer. | Aghdassi A et al |
| 27127168 | 2016 | Immunohistochemical Characterization of Histone Deacetylase as a Potential Prognostic Marker and Therapeutic Target in Endometrial Stromal Sarcoma. | Baek MH et al |
| 25307864 | 2015 | Nuclear expression of histone deacetylases and their histone modifications predicts clinical outcome in colorectal cancer. | Benard A et al |
| 11749695 | 2001 | Expression profile of histone deacetylase 1 in gastric cancer tissues. | Choi JH et al |
| 28107582 | 2017 | Immunoexpression of HDAC1, HDAC2, and HAT1 in actinic cheilitis and lip squamous cell carcinoma. | Chrun ES et al |
| 20404188 | 2010 | Histone deacetylase 1 (HDAC1), but not HDAC2, controls embryonic stem cell differentiation. | Dovey OM et al |
| 20178884 | 2010 | Immunohistochemical detection of histone deacetylases in endometrial carcinoma: involvement of histone deacetylase 2 in the proliferation of endometrial carcinoma cells. | Fakhry H et al |
| 23873102 | 2014 | Clinical significance of histone deacetylase (HDAC)-1, HDAC-2, HDAC-4, and HDAC-6 expression in human malignant and benign thyroid lesions. | Giaginis C et al |
| 23948281 | 2013 | The expression of histone deacetylase 4 is associated with prednisone poor-response in childhood acute lymphoblastic leukemia. | Gruhn B et al |
| 23420051 | 2013 | Investigation of the expression patterns and correlation of DNA methyltransferases and class I histone deacetylases in ovarian cancer tissues. | Gu Y et al |
| 15042618 | 2004 | Upregulation and nuclear recruitment of HDAC1 in hormone refractory prostate cancer. | Halkidou K et al |
| 20049841 | 2010 | Type-specific roles of histone deacetylase (HDAC) overexpression in ovarian carcinoma: HDAC1 enhances cell proliferation and HDAC3 stimulates cell migration with downregulation of E-cadherin. | Hayashi A et al |
| 17720775 | 2007 | Expression of HDAC1 and CBP/p300 in human colorectal carcinomas. | Ishihama K et al |
| 10777477 | 2000 | Histone deacetylases specifically down-regulate p53-dependent gene activation. | Juan LJ et al |
| 23697933 | 2013 | The physiological roles of histone deacetylase (HDAC) 1 and 2: complex co-stars with multiple leading parts. | Kelly RD et al |
| 15770522 | 2005 | Histone deacetylase-1 and -3 protein expression in human breast cancer: a tissue microarray analysis. | Krusche CA et al |
| 20924032 | 2010 | Expression of class I histone deacetylases (HDAC1 and HDAC2) in oesophageal adenocarcinomas: an immunohistochemical study. | Langer R et al |
| 25076845 | 2014 | Expression of histone deacetylases in diffuse large B-cell lymphoma and its clinical significance. | Lee SH et al |
| 23627572 | 2013 | Differential expression of histone deacetylases HDAC1, 2 and 3 in human breast cancer--overexpression of HDAC2 and HDAC3 is associated with clinicopathological indicators of disease progression. | Müller BM et al |
| 23109994 | 2012 | Expression of HAT1 and HDAC1, 2, 3 in Diffuse Large B-Cell Lymphomas, Peripheral T-Cell Lymphomas, and NK/T-Cell Lymphomas. | Min SK et al |
| 20636436 | 2010 | Differential expression of HDAC3, HDAC7 and HDAC9 is associated with prognosis and survival in childhood acute lymphoblastic leukaemia. | Moreno DA et al |
| 17786334 | 2007 | Expression profile of class I histone deacetylases in human cancer tissues. | Nakagawa M et al |
| 11563853 | 2001 | Histone deacetylase and DNA methyltransferase in human prostate cancer. | Patra SK et al |
| 15474665 | 2004 | Histone deacetylase 1 mRNA expression in lung cancer. | Sasaki H et al |
| 25548579 | 2014 | Expression of Histone Deacetylases HDAC1, HDAC2, HDAC3, and HDAC6 in Invasive Ductal Carcinomas of the Breast. | Seo J et al |
| 21725604 | 2011 | Histone deacetylase 1 expression in gastric cancer. | Sudo T et al |
| 19383825 | 2009 | Protein acetylation and histone deacetylase expression associated with malignant breast cancer progression. | Suzuki J et al |
| 21457345 | 2011 | Histone deacetylase-1 and -2 expression in mobile tongue squamous cell carcinoma: associations with clinicopathological parameters and patients survival. | Theocharis S et al |
| 12579268 | 2003 | Histone H4 acetylation and histone deacetylase 1 expression in esophageal squamous cell carcinoma. | Toh Y et al |
| 22347520 | 2012 | An atlas of histone deacetylase expression in breast cancer: fluorescence methodology for comparative semi-quantitative analysis. | Ververis K et al |
| 19103471 | 2009 | HDAC expression and clinical prognosis in human malignancies. | Weichert W et al |
| 18212746 | 2008 | Histone deacetylases 1, 2 and 3 are highly expressed in prostate cancer and HDAC2 expression is associated with shorter PSA relapse time after radical prostatectomy. | Weichert W et al |
| 25944469 | 2015 | Analysis of class I and II histone deacetylase gene expression in human leukemia. | Yang H et al |
| 16940178 | 2006 | Negative and positive regulation of gene expression by mouse histone deacetylase 1. | Zupkovitz G et al |
| 19362090 | 2009 | E-cadherin regulates metastasis of pancreatic cancer in vivo and is suppressed by a SNAIL/HDAC1/HDAC2 repressor complex. | von Burstin J et al |
Other Information
Locus ID:
NCBI: 3065
MIM: 601241
HGNC: 4852
Ensembl: ENSG00000116478
Variants:
dbSNP: 3065
ClinVar: 3065
TCGA: ENSG00000116478
COSMIC: HDAC1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000116478 | ENST00000373548 | Q13547 |
| ENSG00000116478 | ENST00000373548 | Q6IT96 |
| ENSG00000116478 | ENST00000428704 | Q5TEE2 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA10832 | corticosteroids | Chemical | ClinicalAnnotation | associated | PD | 24307847 | |
| PA443450 | Asthma | Disease | ClinicalAnnotation | associated | PD | 24307847 | |
| PA451846 | valproic acid | Chemical | Pathway | associated | 23407051 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38254102 | 2024 | HDAC1/2 control mesothelium/ovarian cancer adhesive interactions impacting on Talin-1-α5β1-integrin-mediated actin cytoskeleton and extracellular matrix protein remodeling. | 0 |
| 38329647 | 2024 | ZNF787 and HDAC1 Mediate Blood-Brain Barrier Permeability in an In Vitro Model of Alzheimer's Disease Microenvironment. | 0 |
| 38505872 | 2024 | HDAC1-Mediated Downregulation of NEU1 Exacerbates the Aggressiveness of Cervical Cancer. | 0 |
| 38812060 | 2024 | Unveiling the role of HP1α-HDAC1-STAT1 axis as a therapeutic target for HP1α-positive intrahepatic cholangiocarcinoma. | 1 |
| 38254102 | 2024 | HDAC1/2 control mesothelium/ovarian cancer adhesive interactions impacting on Talin-1-α5β1-integrin-mediated actin cytoskeleton and extracellular matrix protein remodeling. | 0 |
| 38329647 | 2024 | ZNF787 and HDAC1 Mediate Blood-Brain Barrier Permeability in an In Vitro Model of Alzheimer's Disease Microenvironment. | 0 |
| 38505872 | 2024 | HDAC1-Mediated Downregulation of NEU1 Exacerbates the Aggressiveness of Cervical Cancer. | 0 |
| 38812060 | 2024 | Unveiling the role of HP1α-HDAC1-STAT1 axis as a therapeutic target for HP1α-positive intrahepatic cholangiocarcinoma. | 1 |
| 36287107 | 2023 | HDAC1 regulates the chromatin landscape to control transcriptional dependencies in chronic lymphocytic leukemia. | 3 |
| 36346011 | 2023 | Regulation of osteogenesis in bone marrow-derived mesenchymal stem cells via histone deacetylase 1 and 2 co-cultured with human gingival fibroblasts and periodontal ligament cells. | 4 |
| 36587730 | 2023 | Proteomics-based trapping with single or multiple inactive mutants reproducibly profiles histone deacetylase 1 substrates. | 0 |
| 37121053 | 2023 | HDAC1 mediates epithelial-mesenchymal transition and promotes cancer cell invasion in glioblastoma. | 3 |
| 37661113 | 2023 | Evaluation of epigenetic-related gene expression (DNMT, HDAC1) in Iranian patients with systemic lupus erythematosus. | 0 |
| 37737560 | 2023 | BCL11B and the NuRD complex cooperatively guard T-cell fate and inhibit OPA1-mediated mitochondrial fusion in T cells. | 1 |
| 37878419 | 2023 | GSE1 links the HDAC1/CoREST co-repressor complex to DNA damage. | 0 |
Citation
Emanuely Silva Chrun ; Filipe Modolo ; Filipe Ivan Daniel
HDAC1 (histone deacetylase 1)
Atlas Genet Cytogenet Oncol Haematol. 2017-08-01
Online version: http://atlasgeneticsoncology.org/gene/40802/hdac1-(histone-deacetylase-1)
