MAF (v-maf musculoaponeurotic fibrosarcoma oncogene homolog (avian))
2009-11-01 Alain Eychène , Celio Pouponnot AffiliationInstitut Curie, CNRS UMR 146, Bat110 Centre Universitaire, F-91405 Orsay, France
Identity
HGNC
LOCATION
16q23.2
IMAGE

IMAGE

LEGEND
MAF (v-maf musculoaponeurotic fibrosarcoma oncogene homolog (avian)) Hybridization with Vysis LSI IGH/MAF dual color, dual fusion probe (Abbott Molecular, US) showing MAF on 16q23.2 (red signals) - Courtesy Adriana Zamecnikova.
LOCUSID
ALIAS
AYGRP,CCA4,CTRCT21,c-MAF
FUSION GENES
DNA/RNA
Note
The MAF gene is located on chromosome 16q22-q23. Its paralogs MAFA, MAFB and NRL are located on 8q24.3, 20q11.2-q13.1 and 14q11.1-q11.2, respectively. MAF (c-MAF) was first cloned in chicken as the homolog of the founding member of the family, the viral v-MAF gene, encoded by the AS42 retrovirus that induces Musculo Aponeuorotic Fibrosarcoma (M.A.F.) in chicken. The MAF acronym originates from this tumor.

The two different MAF transcripts are represented. Variant#2 corresponds to unspliced transcript. Variant#1 corresponds to alternative splicing, which contains two exons. Note that the sequence of these two exons is present on transcript variant#2.
Description
The MAF gene encodes two isoforms generated through alternative splicing. These two protein isoforms are called the MAF short form and MAF long form. The MAF short form is translated from the longest mRNA (mRNA variant#2) encoded by one exon while the longest is encoded by two exons (mRNA variant#1) (see diagram). These two products differ in their carboxy terminal part, the long form containing 30 extra amino-acids.
Due to putative alternative polyadenylation signals, other transcripts might encode MAF short form.
Due to putative alternative polyadenylation signals, other transcripts might encode MAF short form.
Transcription
MAF is expressed in many tissues, but its expression is lineage-specific within a given tissue. For example in T cells, MAF is expressed only in the TH2 lineage.
Proteins
Note
The Maf oncoproteins are b-ZIP transcription factors that belong to the AP-1 super-family, which notably includes JUN and FOS. The Maf family contains seven members, which can be subdivided into two subfamilies: the large and small Maf proteins. While the small Maf proteins, MAFF, MAFG and MAFK, are essentially composed of a b-Zip domain, the large Maf proteins, MAFA/L-MAF, MAFB, MAF/c-MAF and NRL contain an additional amino-terminal transactivation domain. Consequently, as homodimers large Maf activate transcription while small Maf repress it. Two different isoforms exist for MAF, which differ in their carboxy-terminus. The short form is constituted of 373 amino acids while the longest form contains 30 additional amino-acids. No specific function has been attributed to these two products.

Schematic representation of MAF protein structure. The two different isoforms are depicted. The basic, leucine zipper, and EHR domains are indicated. The basic domain contacts directly DNA while the leucine zipper allows dimerization, which is a prerequisite for DNA binding. The EHR domain (extended homology region) is specific for the Maf proteins and is required for Maf binding to DNA. MAF protein is phosphorylated by GSK3, although the residues involved have not been formally identified. Putative GSK3 phosphorylation sites are indicated by analogy with MAFA. Phosphorylation of MAFA transactivation domain by GSK3 induces its ubiquitination and proteasome-dependent degradation. This is coupled to an increase in MAFA transactivating activity. These phosphorylations are required for MAFA transforming activity. Like MAFA and MAFB, MAF is also subjected to sumoylation, which decreases its transactivating activity. The sumoylated Lysine (K33) is indicated.
Description
MAF, like all large Maf proteins, contains an amino-terminal transactivation domain and a carboxy-terminal b-ZIP DNA binding domain. Large Maf proteins stimulate transcription of their target genes through their binding to two types of palindromic sequences called TRE- or CRE- type MARE (Maf Responsive Element) (TGCTGACTCAGCA and TGCTGACGTCAGCA, respectively). The Extended Homology Region (EHR, also called ancillary domain, which is specific of the Maf proteins, allows them to recognize a longer palindromic sequence than other AP-1 family members. The MARE sequence is composed of a TRE or CRE core (underlined) flanked by a TGC sequence. While the TGC motif is crucial for Maf binding, the TRE/CRE core can be more degenerate. As other large Maf, MAF is post-translationaly modified by phosphorylation, sumoylation and ubiquitination, mostly occurring in its amino-terminal transactivation domain. Due to posttranslational modifications, notably through GSK3-mediated phosphorylation, MAF apparent molecular weight differs from the calculated one. The protein migrates as different forms ranging from 35 to 50 kDa in SDS-PAGE.
Expression
MAF is expressed in many tissues including neural tissue, kidney and lens. However, its expression is lineage-specific within a given tissue. In T cells for example, MAF is expressed only in the TH2 lineage.
Localisation
MAF is a nuclear protein.
Function
MAF is a transcription factor of the b-Zip family that contacts DNA as homo- or hetero- dimers. MAF knockout mice show peri-natal lethality. They are microphthalmic secondary to defective lens formation. Knockout studies have revealed key roles for MAF in lens terminal differentiation where it regulates the expression of the crystallins, in the specification of the TH2 lineage by regulating the expression of the TH2-specific IL4 cytokine, as well as in chondrocyte terminal differentiation. The role of MAF in lens development is further highlighted by the existence of MAF germinal mutations responsible for congenital cataract in human.
Beside its physiological roles, MAF is an oncogene involved in human cancers (see below).
Beside its physiological roles, MAF is an oncogene involved in human cancers (see below).
Homology
MAF belongs to the AP-1 superfamilly that includes the JUN, FOS, ATF, CREB and Maf family. The Maf family is composed of seven members: the large Maf: MAF, MAFB, MAFA and NRL and the small Maf: MAFF, MAFG, MAFK that are highly homologous in their EHR- b-ZIP domains. Large Maf are closest to each others, MAF showing 77%, 67% and 53% identities with MAFB, MAFA and NRL, respectively. An ortholog of the large MAF, called TrafficJam, is found in Drosophila.
Mutations
Germinal
Congenital cataract (see below)
Implicated in
Entity name
Disease
Multiple myeloma (cancer of plasma B cells).
Prognosis
MAF translocations are of poor prognosis.
Cytogenetics
Fusion protein
No fusion protein. These translocations lead to the juxtaposition of a strong immunoglobulin enhancer to the MAF promoter leading to its strong ectopic expression.
Oncogenesis
Large Maf proteins, MAFA, MAFB and MAF/c-MAF, are bona fide oncoproteins as demonstrated in tissue culture, animal models and in human cancer. Their oncogenic activity depends on their ability to act as transcription factors, relies on overexpression and does not require activating mutations (no such mutations have been found in human cancers), although it can be enhanced by point mutation. It is noteworthy that the transforming activity of Maf proteins is context-dependent. It is regulated by post-translational modifications, notably phosphorylation. They can occasionally display tumor suppressor-like activity in specific cellular settings. In embryonic fibroblasts, MAFA displays the strongest transforming activity. Its transforming activity depends on its phosphorylation by GSK3. In human, MAF/c-MAF, MAFB and MAFA genes are translocated to the immunoglobulin heavy chain (IgH) locus in 8-10% of multiple myelomas (MM). These translocations lead to overexpression through a strong enhancer effect without any alteration in their coding sequence. MAF translocations are present in 5% of MM. Beside these translocations, MAF is overexpressed in 50% of MM through an unknown mechanism. MAF overexpression plays a causative role in MM by promoting proliferation through cyclinD2 (CCND2) induction. It also promotes the pathological interactions of bone marrow stroma with cancer cells by enhancing integrin B7 (ITGB7) expression. These pathological cellular interactions lead to increased production of the angiogenic cytokine VEGF. MAF also activates the Ser/Thr AKT activity by inducing the expression of DEPTOR (DEPDC6), an inhibitor of mTORC1 and mTORC2 complexes.
While MM is associated with osteolytic lesions, patients with MAF deregulation show low incidence of bone disease. This observation is correlated with low expression by MAF-expressing myeloma cells of DKK1, a Wnt antagonist associated with bone disease in MM.
Chemical screens have identified glucocorticoids as potential therapeutical agents that target MAF activity through ubiquitin dependent degradation.
While MM is associated with osteolytic lesions, patients with MAF deregulation show low incidence of bone disease. This observation is correlated with low expression by MAF-expressing myeloma cells of DKK1, a Wnt antagonist associated with bone disease in MM.
Chemical screens have identified glucocorticoids as potential therapeutical agents that target MAF activity through ubiquitin dependent degradation.
Entity name
Prognosis
Not determined.
Oncogenesis
MAF has been shown to be overexpressed in 60% of angioimmunoblastic T cell lymphoma. The mechanisms underlying these deregulations are unknown. Although its causative role has not been demonstrated, its deregulation, as in multiple myeloma, is associated to that of CCND2 and ITGB7. MAF has also been found overexpressed in diverse human T cell lymphomas. Accordingly, MAF overexpression in the T cell compartment leads to T cell lymphoma associated with CCND2 and ITGB7 overexpression in transgenic mice.
Entity name
Congenital cataract
Disease
In human, the MAF gene is mutated in congenital cataract that could be associated with microcornea or colomboma. These heterozygote point mutations correspond to residues located in the DNA binding domain and appear to be dominant. They disrupt DNA binding activity by targeting the different conserved basic residues R288P, K297R and R299S. Another mutation in the leucine zipper dimerization domain (K320E) was reported. In support for a role of MAF in congenital cataract, knockout studies in mice have demonstrated an important role of MAF during lens development. Moreover, heterozygote point mutants of murine MAF in its transactivation (D90V) or basic domain (R291Q, corresponding to human residue R294) lead to cataract in mice.
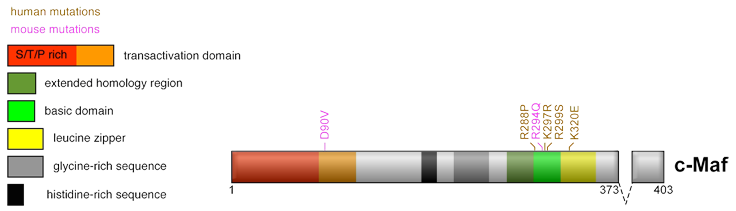
Critical residues mutated in congenital cataract in human and mice are indicated.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 9616139 | 1998 | Frequent dysregulation of the c-maf proto-oncogene at 16q23 by translocation to an Ig locus in multiple myeloma. | Chesi M et al |
| 10747965 | 2000 | Maf transcriptionally activates the mouse p53 promoter and causes a p53-dependent cell death. | Hale TK et al |
| 17982426 | 2007 | Novel MAF mutation in a family with congenital cataract-microcornea syndrome. | Hansen L et al |
| 19182255 | 2009 | Comprehensive mutational screening in a cohort of Danish families with hereditary congenital cataract. | Hansen L et al |
| 9566892 | 1998 | c-Maf interacts with c-Myb to regulate transcription of an early myeloid gene during differentiation. | Hedge SP et al |
| 15090448 | 2004 | Advances in biology of multiple myeloma: clinical applications. | Hideshima T et al |
| 8674125 | 1996 | The proto-oncogene c-maf is responsible for tissue-specific expression of interleukin-4. | Ho IC et al |
| 14998494 | 2004 | Overexpression of c-maf is a frequent oncogenic event in multiple myeloma that promotes proliferation and pathological interactions with bone marrow stroma. | Hurt EM et al |
| 11772997 | 2002 | Domain disruption and mutation of the bZIP transcription factor, MAF, associated with cataract, ocular anterior segment dysgenesis and coloboma. | Jamieson RV et al |
| 8264639 | 1994 | Maf nuclear oncoprotein recognizes sequences related to an AP-1 site and forms heterodimers with both Fos and Jun. | Kataoka K et al |
| 17569705 | 2007 | Multiple mechanisms and functions of maf transcription factors in the regulation of tissue-specific genes. | Kataoka K et al |
| 10383433 | 1999 | Regulation of lens fiber cell differentiation by transcription factor c-Maf. | Kawauchi S et al |
| 10403649 | 1999 | The transcription factor c-Maf controls the production of interleukin-4 but not other Th2 cytokines. | Kim JI et al |
| 10097114 | 1999 | Requirement for the c-Maf transcription factor in crystallin gene regulation and lens development. | Kim JI et al |
| 11990854 | 2002 | Multiple myeloma: evolving genetic events and host interactions. | Kuehl WM et al |
| 19553542 | 2009 | SUMO conjugation contributes to immune deviation in nonobese diabetic mice by suppressing c-Maf transactivation of IL-4. | Leavenworth JW et al |
| 14678826 | 2004 | Comparison of maf gene expression patterns during chick embryo development. | Lecoin L et al |
| 14512017 | 2003 | Absence of transcription factor c-maf causes abnormal terminal differentiation of hypertrophic chondrocytes during endochondral bone development. | MacLean HE et al |
| 17875808 | 2007 | A chemical biology screen identifies glucocorticoids that regulate c-maf expression by increasing its proteasomal degradation through up-regulation of ubiquitin. | Mao X et al |
| 15735737 | 2005 | Gene expression profiling of plasma cell dyscrasias reveals molecular patterns associated with distinct IGH translocations in multiple myeloma. | Mattioli M et al |
| 17550853 | 2007 | TACI expression is associated with a mature bone marrow plasma cell signature and C-MAF overexpression in human myeloma cell lines. | Moreaux J et al |
| 16424013 | 2006 | Overexpression of c-Maf contributes to T-cell lymphoma in both mice and human. | Morito N et al |
| 18059226 | 2007 | c-Maf expression in angioimmunoblastic T-cell lymphoma. | Murakami YI et al |
| 19687312 | 2009 | Characterization of c-Maf transcription factor in normal and neoplastic hematolymphoid tissue and its relevance in plasma cell neoplasia. | Natkunam Y et al |
| 12970735 | 2003 | MafA has strong cell transforming ability but is a weak transactivator. | Nishizawa M et al |
| 19446321 | 2009 | DEPTOR is an mTOR inhibitor frequently overexpressed in multiple myeloma cells and required for their survival. | Peterson TR et al |
| 16247450 | 2006 | Cell context reveals a dual role for Maf in oncogenesis. | Pouponnot C et al |
| 14692531 | 2003 | C-MAF oncogene dysregulation in multiple myeloma: frequency and biological relevance. | Rasmussen T et al |
| 10603348 | 2000 | Regulation of mouse lens fiber cell development and differentiation by the Maf gene. | Ring BZ et al |
| 17044113 | 2007 | Osteopontin dysregulation and lytic bone lesions in multiple myeloma. | Robbiani DF et al |
| 18042454 | 2007 | GSK-3-mediated phosphorylation enhances Maf-transforming activity. | Rocques N et al |
| 16044163 | 2005 | ARK5 is transcriptionally regulated by the Large-MAF family and mediates IGF-1-induced cell invasion in multiple myeloma: ARK5 as a new molecular determinant of malignant multiple myeloma. | Suzuki A et al |
| 19196658 | 2009 | CS1 promotes multiple myeloma cell adhesion, clonogenic growth, and tumorigenicity via c-maf-mediated interactions with bone marrow stromal cells. | Tai YT et al |
| 18216867 | 2008 | Genetic aberrations and survival in plasma cell leukemia. | Tiedemann RE et al |
| 16470690 | 2006 | A novel mutation in the DNA-binding domain of MAF at 16q23.1 associated with autosomal dominant "cerulean cataract" in an Indian family. | Vanita V et al |
| 18159220 | 2007 | Large Maf Transcription Factors: Cousins of AP-1 Proteins and Important Regulators of Cellular Differentiation. | Yang Y et al |
| 17303695 | 2007 | CKS1B, overexpressed in aggressive disease, regulates multiple myeloma growth and survival through SKP2- and p27Kip1-dependent and -independent mechanisms. | Zhan F et al |
| 16728703 | 2006 | The molecular classification of multiple myeloma. | Zhan F et al |
| 19013005 | 2009 | Identification of primary MAFB target genes in multiple myeloma. | van Stralen E et al |
Other Information
Locus ID:
NCBI: 4094
MIM: 177075
HGNC: 6776
Ensembl: ENSG00000178573
Variants:
dbSNP: 4094
ClinVar: 4094
TCGA: ENSG00000178573
COSMIC: MAF
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000178573 | ENST00000326043 | O75444 |
| ENSG00000178573 | ENST00000393350 | O75444 |
| ENSG00000178573 | ENST00000569649 | H3BP11 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA283 | MAPK8 | Gene | Pathway | associated | 23922006 | ||
| PA30621 | MAPK14 | Gene | Pathway | associated | 23922006 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37710417 | 2024 | Transcription factor c-Maf drives macrophages to promote hypertrophic scar formation. | 0 |
| 37710417 | 2024 | Transcription factor c-Maf drives macrophages to promote hypertrophic scar formation. | 0 |
| 36719481 | 2023 | Novel cataract-causing variant c.177dupC in c-MAF regulates the expression of crystallin genes for cell apoptosis via a mitochondria-dependent pathway. | 1 |
| 36719481 | 2023 | Novel cataract-causing variant c.177dupC in c-MAF regulates the expression of crystallin genes for cell apoptosis via a mitochondria-dependent pathway. | 1 |
| 34852950 | 2022 | Association between WWOX/MAF variants and dementia-related neuropathologic endophenotypes. | 5 |
| 35359922 | 2022 | Molecular Mechanisms Driving IL-10- Producing B Cells Functions: STAT3 and c-MAF as Underestimated Central Key Regulators? | 7 |
| 35383094 | 2022 | Transcription Factor c-Maf Promotes Immunoregulation of Programmed Cell Death 1-Expressed CD8(+) T Cells in Multiple Sclerosis. | 7 |
| 35395181 | 2022 | c-Maf: The magic wand that turns on LSEC fate. | 1 |
| 35467036 | 2022 | c-Maf enforces cytokine production and promotes memory-like responses in mouse and human type 2 innate lymphoid cells. | 10 |
| 35613277 | 2022 | Differential metabolic requirement governed by transcription factor c-Maf dictates innate γδT17 effector functionality in mice and humans. | 7 |
| 34852950 | 2022 | Association between WWOX/MAF variants and dementia-related neuropathologic endophenotypes. | 5 |
| 35359922 | 2022 | Molecular Mechanisms Driving IL-10- Producing B Cells Functions: STAT3 and c-MAF as Underestimated Central Key Regulators? | 7 |
| 35383094 | 2022 | Transcription Factor c-Maf Promotes Immunoregulation of Programmed Cell Death 1-Expressed CD8(+) T Cells in Multiple Sclerosis. | 7 |
| 35395181 | 2022 | c-Maf: The magic wand that turns on LSEC fate. | 1 |
| 35467036 | 2022 | c-Maf enforces cytokine production and promotes memory-like responses in mouse and human type 2 innate lymphoid cells. | 10 |
Citation
Alain Eychène ; Celio Pouponnot
MAF (v-maf musculoaponeurotic fibrosarcoma oncogene homolog (avian))
Atlas Genet Cytogenet Oncol Haematol. 2009-11-01
Online version: http://atlasgeneticsoncology.org/gene/41234/maf-(v-maf-musculoaponeurotic-fibrosarcoma-oncogene-homolog-(avian))
