DAPK1 (death-associated protein kinase 1)
2009-02-01 Regine Schneider-Stock , Albert Roessner , Khouloud Bajbouj AffiliationDepartment of Pathology, Otto-von-Guericke University, Leipziger Strasse 44, 39120 Magdeburg, Germany
DNA/RNA
Description
DAPKβ has an additional unique carboxyl-terminal 12- amino acid extension.
Transcription
Proteins
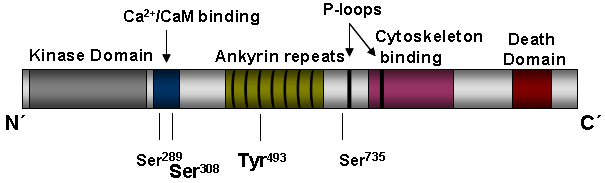
Description
Function
DAPK-mediated cell death is characterized by two main characteristics: formation of autophagic vesicles and membrane blebbing (Shohat et al., 2002).
Migration
DAPK is involved in stabilization of stress fibers through phosphorylation of MLC which occurs prior the onset of apoptosis (Kuo et al., 2003).
Signal transduction
DAP kinase is up-regulated by hyperproliferative signals, and operates upstream of p19-ARF and p53 to induce apoptosis. Whereas the inactivation or loss of DAP kinase significantly reduces the p53 responses to c-Myc or E2F-1, it does not completely eliminate them, indicating that DAP kinase is not an exclusive player upstream of p19ARF/p53 (Raveh et al., 2001). Recent studies showed several mechanisms influencing DAPK activity. These include RSK dependent inactivation of DAPK1 (Anjum et al., 2005) and ERK-dependent activation of the proapoptotic function of DAPK (Chen et al., 2005). Death-promoting effects of DAPK are counteracted by Bcl2 (Cohen et al., 1997).
It was shown that the apoptosis regulatory activities mediated by DAPK are controlled both by phosphorylation status and protein stability.
Phosphorylation sites
There are three well characterized phosphorylation sites on DAPK protein;
1) the phosphorylation by RSK at Ser289, which triggers a suppression of DAPK pro-apoptotic function (Anjum et al., 2005),
2) the autophosphorylation site, which was mapped to Ser308 within the CaM-regulatory domain (Shohat et al., 2002), and
3) ERK-phosphorylation of DAPK at Ser735, which stimulates DAPK-mediated apoptosis by switching off the ERK-C/EBP-beta pathway (Chen et al., 2005). Src phosphorylates DAPK at Y491/492, which induces DAPK intra-/intermolecular interaction and inactivation (Wang et al., 2007).
Interaction partners
Cathepsin B can directly interact with DAPK, forming a stable immune complex (Lin et al., 2007). It has been found that inhibition of HSP90 results in degradation of active dephosphorylated DAPK via the ubiquitin proteasome pathway. DAPK can also form heterocomplexes composed of HSP90 and CHIP or DIP1/Mib1, indicating that the heightened surveillance and modulation of DAPK activities is critical to accurate regulation of apoptosis and cellular homeostasis (Zhang et al., 2007). An interaction between UNC5C (UNC5H3) and DAPK1 was demonstrated (Llambi et al., 2005), whereby this interaction was shown to be dependent on both UNC5H2 lipid raft localization and palmitoylation (Maisse et al., 2008). An interaction between DAPK1 promoter and transcription factors ATF2 and c-jun was demonstrated in cisplatin-treated human breast cancer cells (Hayakawa et al., 2004). Amino acid starvation of cells induced a stable immune complex between microtubule-associated protein MAP1B and DAPK-1 (Harrison et al., 2008) highlighting a new mechanism for authophagy and membrane blebbing. There was found an interaction between DAPK and TSC2 proteins in response to growth factor stimulation that links the DAPK and mTORC1 signaling pathways affecting cell survival, autophagy, and apoptosis (Stevens et al., 2009).
Known substrates
DAPK phosphorylates the myosin II regulatory light chain (Jin et al., 2001) and tropomyosin in response to ERK activation by hydrogen peroxide leading to stress fiber formation (Houle et al., 2007). Furthermore, Syntaxin is a DAP kinase substrate and provides a novel signal transduction pathway by which syntaxin function could be regulated in response to intracellular [Ca2+] and synaptic activity (Tian et al., 2003). Also one of the common DAPK substrate is p19ARF (Raveh et al., 2001). Shani el al. (2004) and Mukhopadhyay et al. (2008) showed that ZIPK serves as a substrate for DAPK. It has been reported that the mammalian 40S ribosomal protein S6 is a DAPK substrate (Schumacher et al., 2006).
Homology
Mutations
Germinal
Implicated in
DAPK is inactive in normal brain tissues, where it is found in its phosphorylated state and becomes rapidly and persistently dephosphorylated and activated in response to ischemia in vivo (Shamloo et al., 2005).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16213824 | 2005 | The tumor suppressor DAP kinase is a target of RSK-mediated survival signaling. | Anjum R et al |
| 15616583 | 2005 | Bidirectional signals transduced by DAPK-ERK interaction promote the apoptotic effect of DAPK. | Chen CH et al |
| 9118961 | 1997 | DAP-kinase is a Ca2+/calmodulin-dependent, cytoskeletal-associated protein kinase, with cell death-inducing functions that depend on its catalytic activity. | Cohen O et al |
| 18195017 | 2008 | DAPK-1 binding to a linear peptide motif in MAP1B stimulates autophagy and membrane blebbing. | Harrison B et al |
| 15546613 | 2004 | Identification of promoters bound by c-Jun/ATF2 during rapid large-scale gene activation following genotoxic stress. | Hayakawa J et al |
| 17895359 | 2007 | DAP kinase mediates the phosphorylation of tropomyosin-1 downstream of the ERK pathway, which regulates the formation of stress fibers in response to oxidative stress. | Houle F et al |
| 11485996 | 2001 | Identification of a new form of death-associated protein kinase that promotes cell survival. | Jin Y et al |
| 16331610 | 2006 | Comparison of DNA hypermethylation patterns in different types of uterine cancer: cervical squamous cell carcinoma, cervical adenocarcinoma and endometrial adenocarcinoma. | Kang S et al |
| 17943730 | 2008 | Methylated TMS1 and DAPK genes predict prognosis and response to chemotherapy in gastric cancer. | Kato K et al |
| 15492995 | 2004 | Death-associated protein kinase (DAP kinase) alteration in soft tissue leiomyosarcoma: Promoter methylation or homozygous deletion is associated with a loss of DAP kinase expression. | Kawaguchi K et al |
| 17401463 | 2007 | Early involvement of death-associated protein kinase promoter hypermethylation in the carcinogenesis of Barrett's esophageal adenocarcinoma and its association with clinical progression. | Kuester D et al |
| 14600263 | 2003 | Uncoordinated regulation of stress fibers and focal adhesions by DAP kinase. | Kuo JC et al |
| 16847012 | 2006 | DAPK1 variants are associated with Alzheimer's disease and allele-specific expression. | Li Y et al |
| 18451218 | 2008 | Promoter hypermethylation of hallmark cancer genes in atypical adenomatous hyperplasia of the lung. | Licchesi JD et al |
| 17324927 | 2007 | Identification of a dominant negative functional domain on DAPK-1 that degrades DAPK-1 protein and stimulates TNFR-1-mediated apoptosis. | Lin Y et al |
| 15729359 | 2005 | The dependence receptor UNC5H2 mediates apoptosis through DAP-kinase. | Llambi F et al |
| 18582460 | 2008 | Lipid raft localization and palmitoylation: identification of two requirements for cell death induction by the tumor suppressors UNC5H. | Maisse C et al |
| 16246486 | 2006 | DAPK promotor methylation is an early event in colorectal carcinogenesis. | Mittag F et al |
| 18995835 | 2008 | DAPK-ZIPK-L13a axis constitutes a negative-feedback module regulating inflammatory gene expression. | Mukhopadhyay R et al |
| 11146619 | 2001 | DAP kinase activates a p19ARF/p53-mediated apoptotic checkpoint to suppress oncogenic transformation. | Raveh T et al |
| 17087515 | 2006 | Death-associated protein kinase phosphorylates mammalian ribosomal protein S6 and reduces protein synthesis. | Schumacher AM et al |
| 16204252 | 2005 | Death-associated protein kinase is activated by dephosphorylation in response to cerebral ischemia. | Shamloo M et al |
| 15367680 | 2004 | Death-associated protein kinase phosphorylates ZIP kinase, forming a unique kinase hierarchy to activate its cell death functions. | Shani G et al |
| 18974095 | 2009 | Peptide combinatorial libraries identify TSC2 as a death-associated protein kinase (DAPK) death domain-binding protein and reveal a stimulatory role for DAPK in mTORC1 signaling. | Stevens C et al |
| 12730201 | 2003 | Ca2+-dependent phosphorylation of syntaxin-1A by the death-associated protein (DAP) kinase regulates its interaction with Munc18. | Tian JH et al |
| 17803936 | 2007 | The tumor suppressor DAPK is reciprocally regulated by tyrosine kinase Src and phosphatase LAR. | Wang WJ et al |
| 18286531 | 2008 | Multiple gene methylation of nonsmall cell lung cancers evaluated with 3-dimensional microarray. | Wang Y et al |
Other Information
Locus ID:
NCBI: 1612
MIM: 600831
HGNC: 2674
Ensembl: ENSG00000196730
Variants:
dbSNP: 1612
ClinVar: 1612
TCGA: ENSG00000196730
COSMIC: DAPK1
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA445062 | Neoplasms | Disease | ClinicalAnnotation | associated | PD | 22293537 | |
| PA449748 | gemcitabine | Chemical | ClinicalAnnotation | associated | PD | 22293537 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 36412004 | 2024 | Knockdown of DAPK1 inhibits IL-1β-induced inflammation and cartilage degradation in human chondrocytes by modulating the PEDF-mediated NF-κB and NLRP3 inflammasome pathway. | 1 |
| 38142898 | 2024 | BNIP3 and DAPK1 methylation in peripheral blood leucocytes are noninvasive biomarkers for gastric cancer. | 0 |
| 38282318 | 2024 | The targeting of DAPK1 with miR-190a-3p promotes autophagy in trophoblast cells. | 0 |
| 38438055 | 2024 | Circ1811 suppresses gastric cancer progression by regulating the miR-632/DAPK1 axis. | 0 |
| 38925973 | 2024 | Frequent CDKN2B/P15 and DAPK1 methylation in duodenal follicular lymphoma is related to duodenal reactive lymphoid hyperplasia. | 0 |
| 36412004 | 2024 | Knockdown of DAPK1 inhibits IL-1β-induced inflammation and cartilage degradation in human chondrocytes by modulating the PEDF-mediated NF-κB and NLRP3 inflammasome pathway. | 1 |
| 38142898 | 2024 | BNIP3 and DAPK1 methylation in peripheral blood leucocytes are noninvasive biomarkers for gastric cancer. | 0 |
| 38282318 | 2024 | The targeting of DAPK1 with miR-190a-3p promotes autophagy in trophoblast cells. | 0 |
| 38438055 | 2024 | Circ1811 suppresses gastric cancer progression by regulating the miR-632/DAPK1 axis. | 0 |
| 38925973 | 2024 | Frequent CDKN2B/P15 and DAPK1 methylation in duodenal follicular lymphoma is related to duodenal reactive lymphoid hyperplasia. | 0 |
| 36502323 | 2023 | NMDA Receptor GluN2B Subunit Is Involved in Excitotoxicity Mediated by Death-Associated Protein Kinase 1 in Alzheimer's Disease. | 3 |
| 37626345 | 2023 | Diagnostic value of the combination of DAPK methylation in urinary sediment and B ultrasound for recurrent urinary bladder cancer. | 0 |
| 37804415 | 2023 | Death-associated protein kinase 1 phosphorylates MDM2 and inhibits its protein stability and function. | 0 |
| 36502323 | 2023 | NMDA Receptor GluN2B Subunit Is Involved in Excitotoxicity Mediated by Death-Associated Protein Kinase 1 in Alzheimer's Disease. | 3 |
| 37626345 | 2023 | Diagnostic value of the combination of DAPK methylation in urinary sediment and B ultrasound for recurrent urinary bladder cancer. | 0 |
Citation
Regine Schneider-Stock ; Albert Roessner ; Khouloud Bajbouj
DAPK1 (death-associated protein kinase 1)
Atlas Genet Cytogenet Oncol Haematol. 2009-02-01
Online version: http://atlasgeneticsoncology.org/gene/417/dapk1
