PRLR (prolactin receptor)
2011-12-01 Chon-Hwa Tsai-Morris , Maria L Dufau AffiliationSection on Molecular Endocrinology, Program Developmental Endocrinology, Genetics, NICHD, National Institutes of Health, Bethesda, MD 20892-4510, USA
Identity
HGNC
LOCATION
5p13.2
LOCUSID
ALIAS
HPRL,MFAB,RI-PRLR,hPRLrI
FUSION GENES
DNA/RNA
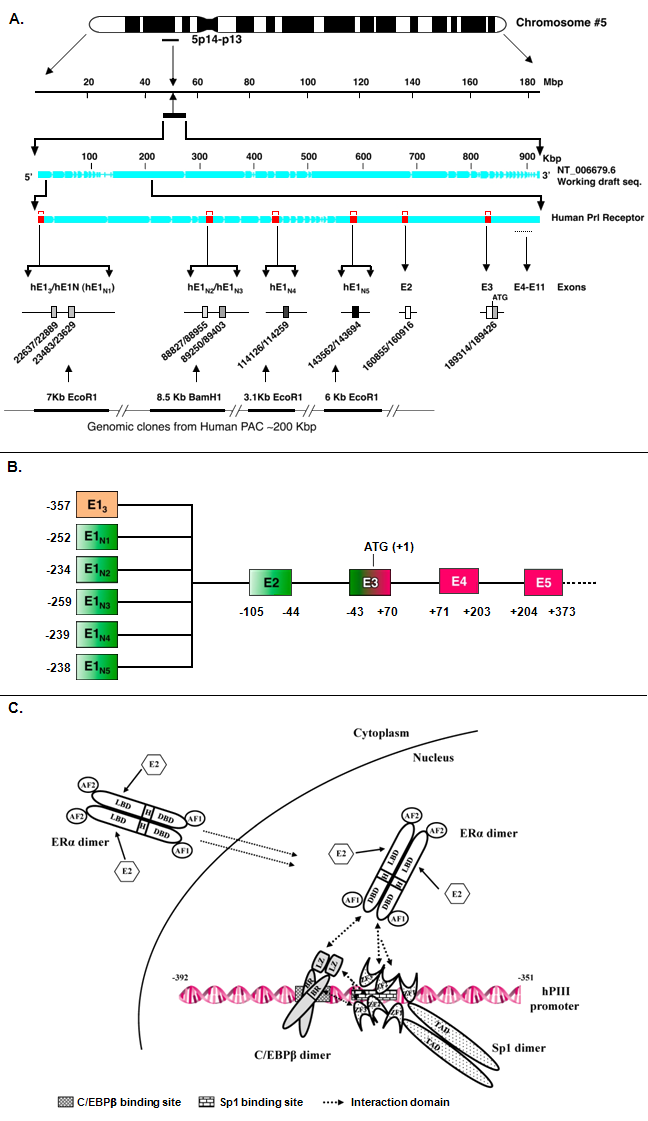
Figure 1. A. Localization of multiple first exons and exons 2-11 of the human prolactin receptor gene in chromosome 5p 14-13. Alternative exons 1: hE13 (generic) and hE1N1-5 (human specific); exon 2: non-coding exon; exon 3: non-coding/coding ATG translation initiation codon; exons 4-11: coding exons. B. Schematic representation of multiple exons 1 and alternative splicing to common exon 2. C. Model for E2-ERα-Sp1-C/EBPβ formation and binding to the hPIII promoter of the human prolactin receptor gene. Schematic presentation of the functional domain of ERα, Sp1 and C/EBPβ and the cognate DNA binding site of Sp1 and C/EBPβ in the hPIII promoter. Each protein is constitutively present as a homodimer dimer. ERα:AF: transactivation domain, DBD: DNA binding domain, H: hinge region, LBD: ligand binding domain; C/EBPβ:BR: basic region, LZ: leucine zipper domain; Sp1:TAD: transactivation domain, ZF: zinc finger motif.
Description
The genomic size of human PRLR gene exceeds 200 kb and contains 11 exons, including six non-coding exons 1 alternative spliced to a common non-coding exon 2 and exon 3-10 that encode the full length activating long form of the receptor. Intermediate and various short forms resulting from alternative splicing. Sequences from exon 11 are present only in the short forms of the receptor S1a and S1b and their respective variants.
Transcription
Transcription of human prolactin receptor gene is regulated by a multiple and tissue-specific promoter (hPIII for exons 1 species hE13 and hPN1-5 for exons 1 species hE1N1-5). The prolactin receptor promoters belong to the TATA-less/non-initiator class. The hPIII promoter contains Sp1 and C/EBP elements that bind Sp1/Sp3 and C/EBPβ required for basal and regulated transcriptional activity, while hPN1 activity is conferred by domains containing an Ets element and an NR half-site. hPN2-5 have not been characterized. Estrogen regulates PRLR transcription through the preferentially utilized PIII promoter via a non-classical ERE independent mechanism in target cells. The protein association induced by estradiol of estrogen receptor α (ERα) with DNA-bound Sp1 (constitutive) and C/EBPβ (recruited by the ERα-SP1 complex) is essential for human prolaction receptor gene transcription (figure 1C). Additional interaction between zinc fingers of Sp1 and leucine zipper of C/EBPβ stabilizes the ERα-Sp1-C/EBPβ complex. The enhanced complex formation of ERα dimer (DNA binding domain) with Sp1 (zinc finger motifs) and C/EBPβ (basic region and leucine zipper) by E2 plays an essential role in the transcriptional activation of the hPRLR gene.
Pseudogene
No known pseudogenes.
Proteins
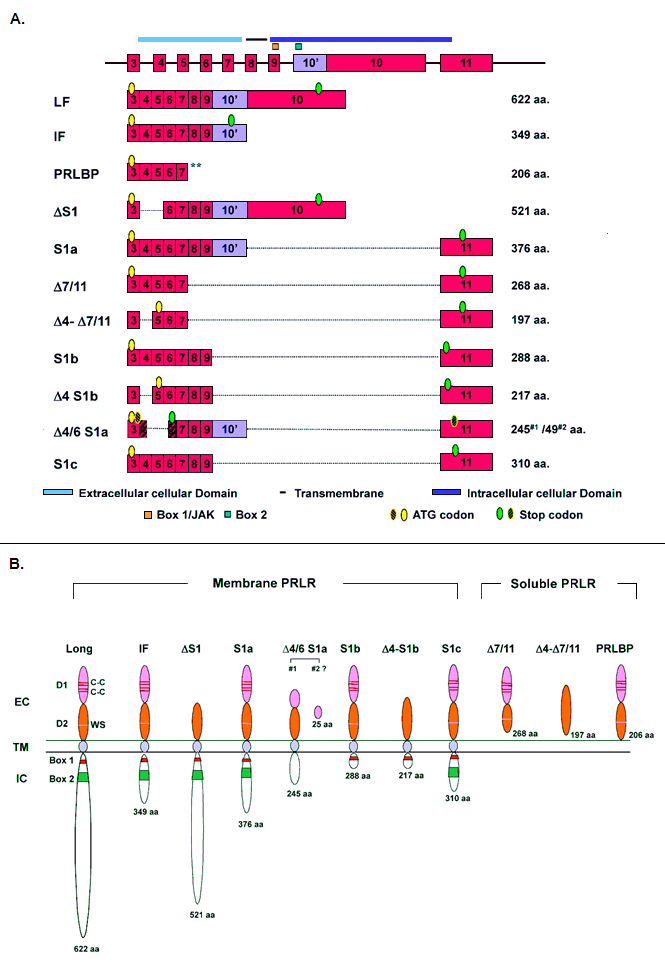
Figure 2. A. Schematic representation of human PRLR variants. Forms generated by alternative splicing. B. Structure of human prolactin receptor variants. Receptor structure of the various forms. LF: long form; IF: intermediate form; S: short forms; 10: partial exon 10; Δ#: deleted exon; #/#: exon/exon splice variant; D1, D2: N-terminal subdomain; WS: WSXWS motif; C: cysteine; Y: tyrosine; EC: extracellular domain; TM: transmembrane domain; IC: intracellular domain. Blue boxes (dark and light) in IC represent two unique sequences of short forms derived from exon 11. Amino acid number includes the signal peptide.
Description
Several forms of the human prolactin receptor have been identified including the full length activating receptor (LF) and at least eight other variants (figures 2A and 2B). These variants differ by the length and composition of their extracellular and/or cytoplasmic domain. In addition to the membrane anchored prolactin receptors variants there is a soluble isoform (prolactin receptor binding protein -PRLRBP) that is generated by proteolytic cleavage of membrane bound prolactin receptor. The human prolactin receptor is composed of a single transmembrane domain, a ligand binding extracellular domain and a cytoplasmic domain which is required for signal transduction. Two disulfide-linked cysteines in the D1 subdomain are involved in ligand binding while WSXWS motif in the D2 subdomain is probably required for correct folding and cellular trafficking. Box 1, a proline rich domain highly conserved in the cytokine receptor family, is the JAK2 docking site. The activated JAK2 induced by prolactin (autophosphorylation), phosphorylates the dimerized receptor preferentially Y587 (at a consensus tyrosine phosphorylation site) which is only present in LF and ΔS1 (figure 2A). This is followed by phosphorylation, dimerization and nuclear translocation of STAT5 which causes transcriptional activation of prolactin responsive genes (i.e. β-casein, β-lactoglobulin, whey acidic protein, interferon-regulatory factor 1 and others). There are other nine tyrosines in the cytoplasmic domain (non-consensus phosphorylation sites) some of which may undergo phosphorylation and may participate in signal transduction. Box2 of unknown function is less conserved in the cytokine receptor family. Prolactin can also activate other tyrosine kinases, including Src family kinases, focal adhesion proteins, Tec kinase, and ErbB kinase. Prolactin induces the GRB2/SOS/Ras/Raf/MAPK signaling cascade. Prolactin through the long form of the receptor stimulates cell proliferation. The Δ-S1 form lacking exon 4 and 5, has reduced affinity for the hormone (due to abbreviated extracellular domain) but displays effective signal transduction. Stimulation of the intermediate form of the receptor (major deletion of exon 10, cytoplasmic domain) only with high concentrations of the ligand exhibits minor cell proliferation. The short forms of the receptor S1a and S1b derived from alternative splicing of exons 10 and 11 are inhibitory of the activation induced by prolactin through the long form of the receptor (see above). Intramolecular disulfide bonds of the prolactin receptor short form are required from its inhibitory action on the function of the long form of the receptor. Δ7/11 and Δ4-Δ7/11 isoforms do not contain transmembrane domain. Δ7/11, Δ4-Δ7/11 and PRLBP are soluble prolactin receptor. Δ4/6 S1a (Acc#. AF512796) variant encodes two open reading frames either 245 or 49 aa truncated prolactin receptor. Spliced variant S1c missing exon 10 was identified in spermatozoa (Acc#. GU133399).
Expression
Prolactin receptors have been identified in number of cells and tissues including the mammary gland, organs of the reproductive system, central nervous system, pituitary, adrenal cortex, skin, bone, lung, heart, liver, pancreas, GI tract, kidney, lymphoid tissue and spermatozoa. These are also present in breast cancer tissues and cells and in other tumoral tissues/cells.
Localisation
Localized in the cell membrane, but also present intracellularly at various compartments.
Function
The prolactin receptor mediates prolactin signaling and triggers intracellular responses that participate in diverse biological functions including, mammary gland development (proliferation and differentiation), initiation and maintenance of lactation, regulation of water and salt balance, reproduction, gonadal steroidogenesis, preservation of sperm integrity, embryonic implantation, brain and behavior, and immune-regulation (see description).
Implicated in
Entity name
Various diseases
Note
Changes in the expression of prolactin receptor variants were found in breast cancer tissues and cells lines when compared to adjacent normal tissues/cells. Polymorphism of prolactin receptor may be related to breast carcinoma, multiple sclerosis and systemic lupus erythematosus. Two missense variants found in patients with breast tumor, Valine for Isoleucine 76 (I76V) and Leucine for Isoleucine 146 (I146L) with gain of function were proposed to participate in breast tumorigenesis.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18779591 | 2008 | Identification of a gain-of-function mutation of the prolactin receptor in women with benign breast tumors. | Bogorad RL et al |
| 2558309 | 1989 | Identification of a cDNA encoding a long form of prolactin receptor in human hepatoma and breast cancer cells. | Boutin JM et al |
| 15119991 | 2004 | Could prolactin receptor gene polymorphism play a role in pathogenesis of breast carcinoma? | Canbay E et al |
| 6145592 | 1984 | Presence and characterization of prolactin receptors in human benign breast tumours. | Di Carlo R et al |
| 16651265 | 2006 | A novel estradiol/estrogen receptor alpha-dependent transcriptional mechanism controls expression of the human prolactin receptor. | Dong J et al |
| 6089861 | 1984 | Absence of prolactin receptors in normal and malignant uterine cervix. | Dowsett M et al |
| 11729217 | 2001 | Expression of prolactin receptors in normal, benign, and malignant breast tissue: an immunohistological study. | Gill S et al |
| 21036240 | 2010 | Identification of gain-of-function variants of the human prolactin receptor. | Goffin V et al |
| 11518703 | 2001 | Isolation and characterization of two novel forms of the human prolactin receptor generated by alternative splicing of a newly identified exon 11. | Hu ZZ et al |
| 12021177 | 2002 | Complex 5' genomic structure of the human prolactin receptor: multiple alternative exons 1 and promoter utilization. | Hu ZZ et al |
| 21670145 | 2011 | Complex formation and interactions between transcription factors essential for human prolactin receptor gene transcription. | Kang JH et al |
| 10585417 | 1999 | Functional characterization of the intermediate isoform of the human prolactin receptor. | Kline JB et al |
| 12351696 | 2002 | Characterization of a novel and functional human prolactin receptor isoform (deltaS1PRLr) containing only one extracellular fibronectin-like domain. | Kline JB et al |
| 10709093 | 2000 | Characterization and modulation of a prolactin receptor mRNA isoform in normal and tumoral human breast tissues. | Laud K et al |
| 12559630 | 2003 | Prolactin and prolactin receptor gene polymorphisms in multiple sclerosis and systemic lupus erythematosus. | Mellai M et al |
| 15313907 | 2004 | Human prolactin receptor variants in breast cancer: low ratio of short forms to the long-form human prolactin receptor associated with mammary carcinoma. | Meng J et al |
| 20032052 | 2010 | Prolactin exerts a prosurvival effect on human spermatozoa via mechanisms that involve the stimulation of Akt phosphorylation and suppression of caspase activation and capacitation. | Pujianto DA et al |
| 16556730 | 2006 | Ligand-independent homo- and heterodimerization of human prolactin receptor variants: inhibitory action of the short forms by heterodimerization. | Qazi AM et al |
| 19906835 | 2010 | Short form 1b human prolactin receptor down-regulates expression of the long form. | Tan D et al |
| 12580759 | 2003 | Alternative splicing to exon 11 of human prolactin receptor gene results in multiple isoforms including a secreted prolactin-binding protein. | Trott JF et al |
| 19273600 | 2009 | Intramolecular disulfide bonds of the prolactin receptor short form are required for its inhibitory action on the function of the long form of the receptor. | Xie YL et al |
Other Information
Locus ID:
NCBI: 5618
MIM: 176761
HGNC: 9446
Ensembl: ENSG00000113494
Variants:
dbSNP: 5618
ClinVar: 5618
TCGA: ENSG00000113494
COSMIC: PRLR
RNA/Proteins
Expression (GTEx)
Pathways
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38713636 | 2024 | The Human Intermediate Prolactin Receptor I-tail Contributes Breast Oncogenesis by Targeting Ras/MAPK Pathway. | 0 |
| 38713636 | 2024 | The Human Intermediate Prolactin Receptor I-tail Contributes Breast Oncogenesis by Targeting Ras/MAPK Pathway. | 0 |
| 36638053 | 2023 | PRL-R Variants Are Not Only Associated With Prolactinomas But Also With Dopamine Agonist Resistance. | 1 |
| 37232489 | 2023 | The prolactin receptor scaffolds Janus kinase 2 via co-structure formation with phosphoinositide-4,5-bisphosphate. | 3 |
| 37993904 | 2023 | The prolactin receptor gene (PRLR) is linked and associated with the risk of polycystic ovarian syndrome. | 0 |
| 36638053 | 2023 | PRL-R Variants Are Not Only Associated With Prolactinomas But Also With Dopamine Agonist Resistance. | 1 |
| 37232489 | 2023 | The prolactin receptor scaffolds Janus kinase 2 via co-structure formation with phosphoinositide-4,5-bisphosphate. | 3 |
| 37993904 | 2023 | The prolactin receptor gene (PRLR) is linked and associated with the risk of polycystic ovarian syndrome. | 0 |
| 35922139 | 2022 | Breast Cancer and Prolactin - New Mechanisms and Models. | 5 |
| 36187116 | 2022 | Prolactin receptor gene transcriptional control, regulatory modalities relevant to breast cancer resistance and invasiveness. | 7 |
| 36744975 | 2022 | [Prolactin and its receptor: From animal models to pituitary pathophysiology]. | 1 |
| 35922139 | 2022 | Breast Cancer and Prolactin - New Mechanisms and Models. | 5 |
| 36187116 | 2022 | Prolactin receptor gene transcriptional control, regulatory modalities relevant to breast cancer resistance and invasiveness. | 7 |
| 36744975 | 2022 | [Prolactin and its receptor: From animal models to pituitary pathophysiology]. | 1 |
| 33259941 | 2021 | Immunohistochemical localization of prolactin receptor (PRLR) to Hodgkin's and Reed-Sternberg cells of Hodgkin's lymphoma. | 4 |
Citation
Chon-Hwa Tsai-Morris ; Maria L Dufau
PRLR (prolactin receptor)
Atlas Genet Cytogenet Oncol Haematol. 2011-12-01
Online version: http://atlasgeneticsoncology.org/gene/42891/prlr
Historical Card
2004-10-01 PRLR (prolactin receptor) by Chon-Hwa Tsai-Morris,Maria L Dufau Affiliation
Section on Molecular Endocrinology, Program Developmental Endocrinology, Genetics, NICHD, National Institutes of Health, Bethesda, MD 20892-4510, USA
