AVEN (apoptosis, caspase activation inhibitor)
2013-10-01 Inga Maria Melzer , Martin Zörnig AffiliationInstitute for Biomedical Research Georg-Speyer-Haus, Paul-Ehrlich-Strasse 42-44, 60596 Frankfurt, Germany
DNA/RNA
Description
Transcription
Pseudogene
Proteins
Description
Expression
Localisation
Function
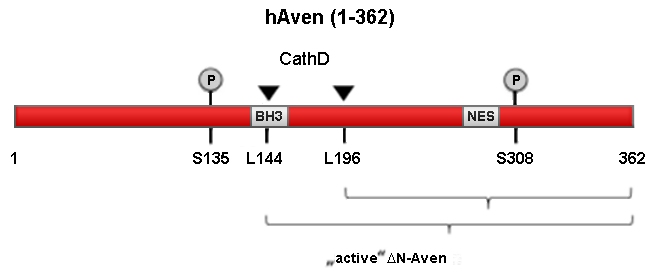
AVEN was first discovered as an interactor of the antiapoptotic BCl-xL protein by Chau et al. (2000). It was also shown to bind to the proapoptotic APAF-1 protein and postulated to prevent the oligomerization of APAF-1 (apoptosome formation) in the intrinsic apoptosis pathway and to stabilize the Bcl-xL protein by binding to it (Kutuk et al., 2010). Putative binding sites in Bcl-xL are predicted to be located in the Bcl-xL BH1 and BH4 domains (Hawley et al., 2012). Recently, it was shown that AVEN can be processed by the lysosomal protease Cathepsin D at aa 144 and 196, and that this processing is neccessary to activate AVENs antiapoptotic function (Melzer et al., 2012). It is still unclear whether it is the stabilization of Bcl-xL, the interference with apoptosome assembly or another feature of AVEN that is responsible for the antiapoptotic capacity of this protein.
DNA damage repair:
It was shown by Guo et al. (2008) that AVEN, in addition to binding to the apoptotic machinery, is also able to bind one of the key players in DNA damage repair, the ataxia telangiectasia mutated (ATM) kinase. Overexpression of AVEN in Xenopus laevis egg extracts induced a cell cycle arrest at G2/M which is in large part ATM dependent, whereas the absence of AVEN impaired ATM-mediated checkpoint function. An intrinsic loop of activation exists between AVEN and ATM: AVEN binds to the kinase domain of ATM (appr. aa 2500-3000) and, in turn, is phosphorylated by ATM at S135 and S308. This phosphorylation seems to enhance AVENs activating influence on ATM. Esmaili et al. (2010) were able to demonstrate that AVEN possesses a nuclear export signal (NES) which is located between aa 282 and 293. Under normal physiological conditions, AVEN is shuttled outside of the nucleus by Exportin-1/CRM1 whereas inhibition of CRM1 by leptomycin or mutation of the AVEN NES leads to nuclear accumulation of the protein. The NES/nuclear-cytosolic shuttling of AVEN might be important for its cell cycle regulatory functions and its role in DNA damage repair.
Depending on the degree of DNA damage, AVEN is possibly a multifunctional protein, finetuning the cellular decisions of cell cycle arrest and apoptosis in the DNA damage response.
Homology
Homologs of AVEN can be found in several species, like mouse (NCBI acc. Nr. NP_083120), Drosophila (NP_572817), rat (NP_001101227), chicken (NP_001005791; Vezyri et al., 2011) and Xenopus (NP_001090621; Guo et al., 2008). Of note, two isoforms are postulated to exist in mouse, the second one (NP_001159407) possessing a distinctly shorter N-terminus than the full length protein. However, nothing is known about the function or biological relevance of this predicted second isoform. Functional similarity to the human protein in its cell cycle regulatory properties has been published for the Drosophila (Zou et al., 2011) and the Xenopus homologs (Guo et al., 2008).
Implicated in
First reports that AVEN is overexpressed on mRNA level in acute leukemias were published by Paydas et al. in 2003. The authors investigated a study group consisting of 37 acute myeloblastic leukemias (AML) and 28 acute lymphoblastic leukemia (ALL) patients. Details regarding the number of ALL patients who were either of the frequent B-cell type or had developed T-cell acute lymphoblastic leukemia/lymphoma (T-ALL) were not given. In this study, elevated Aven mRNA expression levels were noted in acute leukemias, and the authors suggest that AVEN could be a new prognostic marker in this cancer entity. Choi et al. (2006) describe a positive correlation between Aven mRNA overexpression and poor prognosis in childhood ALL.
A recent study by Eissmann et al. (2013) shows proof that overexpression of AVEN contributes to increased malignancy in hematopoietic neoplasms. Here, the authors confirm overexpression of AVEN in T-ALL patient samples compared to healthy T cells on protein level. Furthermore, using a transgenic mouse model with T-cell specific overexpression of AVEN, an oncogenic cooperation of AVEN with heterozygous loss of p53 is shown. Additionally, in subcutaneous mouse xenograft models, the authors show that downregulation of AVEN expression via shRNA leads to significantly decreased, if not halted, tumor growth indicating AVEN as a putative novel therapy target for T-ALL and AML.
Kutuk et al. describe decreased nuclear expression of AVEN in breast cancer tissue microarrays, in particular in infiltrative ductal carcinoma and papillary carcinoma compared to non-neoplastic breast tissue and infiltrating lobular breast cancer. They suggest that AVEN might be an important mediator in DNA damage-induced apoptotic signalling and its nuclear downregulation in breast cancer can lead to genomic instability. A recent study by Ouzounova et al. shows that AVEN is an inversely regulated downstream target of the miR-30 family which is important for regulation of breast cancer cells under non-attachment conditions. Overexpression of miR-30 family members reduces breast tumor progression and tumorsphere formation (and AVEN expression), an effect which could be partially rescued by AVEN re-/overexpression, suggesting, in contrast to the other study, that rather overexpression (than downregulation or nuclear depletion) of AVEN is important for breast tumor growth.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 10949025 | 2000 | Aven, a novel inhibitor of caspase activation, binds Bcl-xL and Apaf-1. | Chau BN et al |
| 16388850 | 2006 | Aven overexpression: association with poor prognosis in childhood acute lymphoblastic leukemia. | Choi J et al |
| 22751129 | 2013 | Overexpression of the anti-apoptotic protein AVEN contributes to increased malignancy in hematopoietic neoplasms. | Eißmann M et al |
| 20935510 | 2010 | Regulation of the ATM-activator protein Aven by CRM1-dependent nuclear export. | Esmaili AM et al |
| 18571408 | 2008 | Aven-dependent activation of ATM following DNA damage. | Guo JY et al |
| 22754595 | 2012 | An Integrated Bioinformatics and Computational Biology Approach Identifies New BH3-Only Protein Candidates. | Hawley RG et al |
| 20619636 | 2010 | Aven blocks DNA damage-induced apoptosis by stabilising Bcl-xL. | Kutuk O et al |
| 22388353 | 2012 | The Apaf-1-binding protein Aven is cleaved by Cathepsin D to unleash its anti-apoptotic potential. | Melzer IM et al |
| 23445407 | 2013 | MicroRNA miR-30 family regulates non-attachment growth of breast cancer cells. | Ouzounova M et al |
| 12853345 | 2003 | Survivin and aven: two distinct antiapoptotic signals in acute leukemias. | Paydas S et al |
| 21234663 | 2011 | Molecular cloning and expression of Aven gene in chicken. | Vezyri E et al |
| 21368576 | 2011 | Identification of dAven, a Drosophila melanogaster ortholog of the cell cycle regulator Aven. | Zou S et al |
Other Information
Locus ID:
NCBI: 57099
MIM: 605265
HGNC: 13509
Ensembl: ENSG00000169857
Variants:
dbSNP: 57099
ClinVar: 57099
TCGA: ENSG00000169857
COSMIC: AVEN
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000169857 | ENST00000306730 | Q9NQS1 |
| ENSG00000169857 | ENST00000560649 | H0YKR0 |
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37816050 | 2023 | Downregulation of apoptotic repressor AVEN exacerbates cardiac injury after myocardial infarction. | 0 |
| 37816050 | 2023 | Downregulation of apoptotic repressor AVEN exacerbates cardiac injury after myocardial infarction. | 0 |
| 35322749 | 2022 | MicroRNA-30a contributes to pre-eclampsia through regulating the proliferation, apoptosis, and angiogenesis modulation potential of mesenchymal stem cells by targeting AVEN. | 1 |
| 35322749 | 2022 | MicroRNA-30a contributes to pre-eclampsia through regulating the proliferation, apoptosis, and angiogenesis modulation potential of mesenchymal stem cells by targeting AVEN. | 1 |
| 31768184 | 2019 | Down-regulation of miR-30b-5p protects cardiomyocytes against hypoxia-induced injury by targeting Aven. | 9 |
| 31768184 | 2019 | Down-regulation of miR-30b-5p protects cardiomyocytes against hypoxia-induced injury by targeting Aven. | 9 |
| 29693772 | 2018 | Expression of granulosa cell microRNAs, AVEN and ATRX are associated with human blastocyst development. | 4 |
| 29693772 | 2018 | Expression of granulosa cell microRNAs, AVEN and ATRX are associated with human blastocyst development. | 4 |
| 27388765 | 2016 | miR-30e Blocks Autophagy and Acts Synergistically with Proanthocyanidin for Inhibition of AVEN and BIRC6 to Increase Apoptosis in Glioblastoma Stem Cells and Glioblastoma SNB19 Cells. | 13 |
| 27388765 | 2016 | miR-30e Blocks Autophagy and Acts Synergistically with Proanthocyanidin for Inhibition of AVEN and BIRC6 to Increase Apoptosis in Glioblastoma Stem Cells and Glioblastoma SNB19 Cells. | 13 |
| 25573060 | 2015 | Akt regulation of Aven contributes to the sensitivity of cancer cells to chemotherapeutic agents. | 4 |
| 25757065 | 2015 | Aven-mediated checkpoint kinase control regulates proliferation and resistance to chemotherapy in conventional osteosarcoma. | 0 |
| 26267306 | 2015 | Aven recognition of RNA G-quadruplexes regulates translation of the mixed lineage leukemia protooncogenes. | 49 |
| 25573060 | 2015 | Akt regulation of Aven contributes to the sensitivity of cancer cells to chemotherapeutic agents. | 4 |
| 25757065 | 2015 | Aven-mediated checkpoint kinase control regulates proliferation and resistance to chemotherapy in conventional osteosarcoma. | 0 |
Citation
Inga Maria Melzer ; Martin Zörnig
AVEN (apoptosis, caspase activation inhibitor)
Atlas Genet Cytogenet Oncol Haematol. 2013-10-01
Online version: http://atlasgeneticsoncology.org/gene/43158/aven-(apoptosis-caspase-activation-inhibitor)
