COPS2 (COP9 constitutive photomorphogenic homolog subunit 2 (Arabidopsis))
2009-09-01 Susanne Jennek , Florian Kraft , Aria Baniahmad AffiliationInstitute of Human Genetics, Anthropology, Jena University Hospital, Kollegiengasse 10, 07743 Jena, Germany
Identity
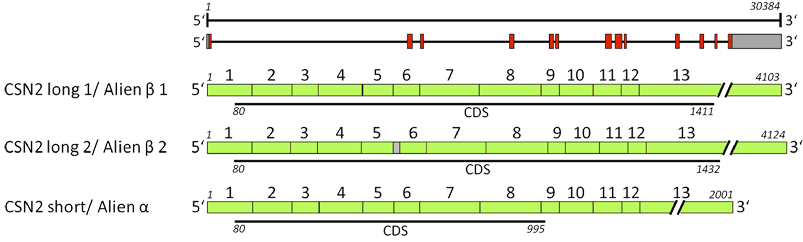
DNA/RNA
Transcription
Furthermore, CSN2/Alien gene expression in vivo is activated by thyroid hormone receptor (TR) and thyroid hormone suggesting a regulatory feedback mechanism between TR and CSN2/Alien expression (Tenbaum et al., 2003).
Proteins

Description
CSN2 activity can be regulated through phosphorylation and dephosphorylation (Kapelari et al., 2000).
Expression
Localisation
Function
Also, corepression function was identified by CSN2 short/Alien a for transcription factors involved in cell cycle regulation and DNA repair such as several members of the E2F transcription factor family (Escher et al., 2007; reviewed in: Papaioannou et al., 2007). Alien is recruited to the E2F1 gene promoter repressing endogenous E2F1 gene expression in vivo. The data also suggest that Alien inhibits transactivation of E2F1, a positive regulator of cell cycle progression. In line with this, Alien represses cell cycle progression. Remarkably, the inhibition of E2F1-mediated transactivation is independent of retinoblastoma protein pRB (Tenbaum et al., 2007). pRB represses E2F1 transcriptional activation. It is not yet known whether Alien is able to substitute pRB function during cell cycle progression (Tenbaum et al., 2007). Furthermore, a direct interaction between Alien and pRB is detected. Interestingly, a pRB-mutant lacking silencing function also lacks interaction with CSN2 short/Alien (Escher et al., 2007).
In addition, Alien interacts with the highly conserved chromatin associated tumor suppressor proteins Inhibitor of growth 1 (p33-ING1b) and 2 (p33-ING2) in vivo and both p33ING1b and p33ING2 are known to induce premature cellular senescence. It is shown that p33ING proteins enhance Alien-mediated gene silencing (Fegers et al., 2007).
The recruitment of HDAC-activity is one mechanism by which Alien realizes its corepression functions (Dressel et al., 1999). However, it is suggested that Alien exhibits both HDAC-dependent and -independent options for gene repression (reviewed in: Papaioannou et al., 2007). Moreover, CSN2 short/Alien a interacts with nucleosome assembly protein 1 (NAP1) in vivo and in vitro regulating its activity through enhancing NAP1-mediated nucleosome assembly on DNA and thereby leading to gene repression (Eckey et al., 2007).
The CSN2 long/Alien b isoform is an essential part of the COP9 signalosome (CSN) complex which is highly conserved in eukaryotes and consists of eight subunits (reviewed in: Wie et al., 2008). The CSN complex plays a central role in the regulation of degradation of multiple proteins through the ability to de-neddylate cullin, which enables the association of cullin with CAND1, a negative regulator of the cullin-based E3 ubiquitin ligases (reviewed in: von Arnim, 2003; reviewed in: Wolf et al., 2003; Chamovitz, 2009; Wu et al., 2009). A role for CSN2 long/Alien b is suggested by the interaction between CSN2 and subunits of the 26S proteasome was already shown (Huang et al., 2005). The promoter region of the Csn2 gene contains NF-kB binding sites like other CSN subunits. Accordingly, these members of the CSN complex are regulated by NF-kB. Snail, a transcription factor, which is a part of the TGF-b pathway and is involved in inflammatory-triggered migration, invasiveness and metastasis of tumor cells, is stabilized by the induction of the CSN complex via NF-kB (Wu et al., 2009). There are also COP9 subcomplexes with yet unknown functions (reviewed in: Wei et al, 2008).
Interaction of COP9 via CSN2 with p53 in tumors can raise the stability of p53, the most important protein involving in a variety of essential tumor suppressive functions and induction of cellular senescence. But in contrast to Snail, the lower turnover does not lead to an increase in transcription activity and therefore neither to an increased p21 expression nor to cell cycle arrest (Leal et al., 2008).
Moreover, CSN2 protein interacts physically with the anaphase-promoting complex (APC/C), a major regulator of the cell cycle and affects specifically its stability (Kob et al., 2008).
Homology
Originally, the name Alien was given to a gene in the Drosophila genome with an unknown function (Goubeaud et al., 1996). It shares high homologies with Thyroid hormone receptor-interacting protein 15 (TRIP15), a mammalian protein (Lee et al.,1995; Dressel et al., 1999).
Mutations
Note
Implicated in
It was shown by quantitative analysis that the CSN2 expression is reduced up to 50% in tumors of prostate, lung and colon (Leal et al., 2008).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12522100 | 2003 | The role of transcriptional corepressor Nif3l1 in early stage of neural differentiation via cooperation with Trip15/CSN2. | Akiyama H et al |
| 10713076 | 2000 | Interaction of the corepressor Alien with DAX-1 is abrogated by mutations of DAX-1 involved in adrenal hypoplasia congenita. | Altincicek B et al |
| 19305390 | 2009 | Revisiting the COP9 signalosome as a transcriptional regulator. | Chamovitz DA et al |
| 10207062 | 1999 | Alien, a highly conserved protein with characteristics of a corepressor for members of the nuclear hormone receptor superfamily. | Dressel U et al |
| 17339334 | 2007 | The nucleosome assembly activity of NAP1 is enhanced by Alien. | Eckey M et al |
| 17330949 | 2007 | Various members of the E2F transcription factor family interact in vivo with the corepressor alien. | Escher N et al |
| 17929852 | 2007 | The tumor suppressors p33ING1 and p33ING2 interact with alien in vivo and enhance alien-mediated gene silencing. | Fegers I et al |
| 11702949 | 2001 | Mouse germline restriction of Oct4 expression by germ cell nuclear factor. | Fuhrmann G et al |
| 8817453 | 1996 | The Drosophila gene alien is expressed in the muscle attachment sites during embryogenesis and encodes a protein highly conserved between plants, Drosophila and vertebrates. | Goubeaud A et al |
| 16045761 | 2005 | Consequences of COP9 signalosome and 26S proteasome interaction. | Huang X et al |
| 10903862 | 2000 | Electron microscopy and subunit-subunit interaction studies reveal a first architecture of COP9 signalosome. | Kapelari B et al |
| 19535905 | 2009 | Regulation of the anaphase-promoting complex by the COP9 signalosome. | Kob R et al |
| 17968325 | 2008 | Cellular senescence bypass screen identifies new putative tumor suppressor genes. | Leal JF et al |
| 7776974 | 1995 | Two classes of proteins dependent on either the presence or absence of thyroid hormone for interaction with the thyroid hormone receptor. | Lee JW et al |
| 18174916 | 2007 | The coregulator Alien. | Papaioannou M et al |
| 10877839 | 2000 | VDR-Alien: a novel, DNA-selective vitamin D(3) receptor-corepressor partnership. | Polly P et al |
| 15571808 | 2004 | The COP9 signalosome (CSN): an evolutionary conserved proteolysis regulator in eukaryotic development. | Schwechheimer C et al |
| 17570542 | 2007 | Alien inhibits E2F1 gene expression and cell proliferation. | Tenbaum SP et al |
| 14570571 | 2003 | The COP9 signalosome. | Wei N et al |
| 19411070 | 2009 | Stabilization of snail by NF-kappaB is required for inflammation-induced cell migration and invasion. | Wu Y et al |
| 14647295 | 2003 | The COP9 signalosome: an assembly and maintenance platform for cullin ubiquitin ligases? | Wolf DA et al |
| 14611949 | 2003 | On again-off again: COP9 signalosome turns the key on protein degradation. | von Arnim AG et al |
Other Information
Locus ID:
NCBI: 9318
MIM: 604508
HGNC: 30747
Ensembl: ENSG00000166200
Variants:
dbSNP: 9318
ClinVar: 9318
TCGA: ENSG00000166200
COSMIC: COPS2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 26942699 | 2016 | DDA1 promotes stage IIB-IIC colon cancer progression by activating NFκB/CSN2/GSK-3β signaling. | 4 |
| 27017521 | 2016 | Apolipoprotein A-1 binding protein promotes macrophage cholesterol efflux by facilitating apolipoprotein A-1 binding to ABCA1 and preventing ABCA1 degradation. | 34 |
| 26942699 | 2016 | DDA1 promotes stage IIB-IIC colon cancer progression by activating NFκB/CSN2/GSK-3β signaling. | 4 |
| 27017521 | 2016 | Apolipoprotein A-1 binding protein promotes macrophage cholesterol efflux by facilitating apolipoprotein A-1 binding to ABCA1 and preventing ABCA1 degradation. | 34 |
| 23441852 | 2013 | The corepressor activity of Alien is controlled by CREB-binding protein/p300. | 0 |
| 23441852 | 2013 | The corepressor activity of Alien is controlled by CREB-binding protein/p300. | 0 |
| 19400951 | 2009 | Analysis of the role of COP9 Signalosome (CSN) subunits in K562; the first link between CSN and autophagy. | 14 |
| 19535905 | 2009 | Regulation of the anaphase-promoting complex by the COP9 signalosome. | 4 |
| 19400951 | 2009 | Analysis of the role of COP9 Signalosome (CSN) subunits in K562; the first link between CSN and autophagy. | 14 |
| 19535905 | 2009 | Regulation of the anaphase-promoting complex by the COP9 signalosome. | 4 |
| 17968325 | 2008 | Cellular senescence bypass screen identifies new putative tumor suppressor genes. | 29 |
| 17968325 | 2008 | Cellular senescence bypass screen identifies new putative tumor suppressor genes. | 29 |
| 17339334 | 2007 | The nucleosome assembly activity of NAP1 is enhanced by Alien. | 16 |
| 17438371 | 2007 | Detection and identification of transcription factors as interaction partners of alien in vivo. | 2 |
| 17570542 | 2007 | Alien inhibits E2F1 gene expression and cell proliferation. | 2 |
Citation
Susanne Jennek ; Florian Kraft ; Aria Baniahmad
COPS2 (COP9 constitutive photomorphogenic homolog subunit 2 (Arabidopsis))
Atlas Genet Cytogenet Oncol Haematol. 2009-09-01
Online version: http://atlasgeneticsoncology.org/gene/47362/cops2
