DLX4 (distal-less homeobox 4)
2011-01-01 Patricia E Berg , Saurabh Kirolikar AffiliationThe George Washington University Medical Center, Washington DC 20037, USA (PEB); The George Washington University, Department of Biochemistry, Molecular Biology, Washington DC 20037, USA (SK)
DNA/RNA
Note
BP1, DLX4 and DLX7 are not interchangeable names for the same gene, as sometimes claimed. We cloned a cDNA encoding BP1 from a library made from K562 erythroleukemia cells, often used as a model for hemoglobin switching. After it was sequenced it was apparent that part of the BP1 sequence was identical to that of two other published "genes", DLX4 and DLX7; upstream of nucleotide 565 of BP1, all three had entirely different sequences, while downstream of that site the sequences were identical (Fu et al., 2001; Chase et al., 2002). This suggested that the three might be isoforms of one gene, differing only in the first exon; this can occur by alternative splicing or by use of alternative promoters. DLX7 had been mapped to 17q21-22 (Nakamura et al., 1996). We then mapped BP1, which also mapped to the same chromosomal region (Fu et al., 2001). In fact, our BAC included sequences from both BP1 and DLX7. When we published the paper showing that BP1 is a repressor of the beta-globin gene and identifying BP1, DLX4 and DLX7 as isoforms, to prevent confusion in the literature we gave the gene a single name, DLX4, based on the fact that the DLX4 DNA sequence was published first (Chase et al., 2002). Thus, the DLX4 gene encodes at least three different proteins with presumably different functions, DLX4, BP1 and DLX7. The NCBI Database is somewhat confusing in this regard - the gene is called DLX4, but BP1 is named DLX4 variant 1 and DLX7 is called DLX4 variant 2.
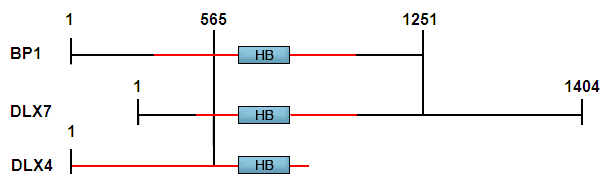
The red lines indicate the predicted ORFs. Number 565 indicates the nucleotide of BP1 where divergence occurs among BP1, DLX4, and DLX7. HB is the homeobox region. The regions between the two vertical lines indicate the regions of DNA identity. The complete ORF is not available for DLX4.
Description
The DLX4 gene is located at 17q21.33 and is about 5761 bp in length (chr17:48,046,562-48,052,322).
Transcription
Three different mRNAs are expressed by DLX4, BP1 and DLX7. BP1 mRNA is about 2012 bp.
Proteins

Amino acid sequence alignment between BP1 (Q92988-1), DLX7 (Q92988-2) and an unidentified isoform of DLX4 (Q92988-3). The sequences were obtained from the UniProt database which has incorrectly identified the BP1 sequence as DLX4.
Description
The BP1 protein is an alternatively spliced isoform derived from the DLX4 gene. The protein is about 240 aa in length with the calculated molecular weight of 26 kDa. The observed weight of the protein is about 36 kDa on Western blots using lysates from breast cancer and prostate cancer cell lines. This difference may be due to post-translational modifications of BP1 protein. BP1 protein has a 116 aa N-terminal region, 60 aa homeobox domain and a 64 aa C-terminal domain, while DLX7 is 168 aa.
Expression
In normal tissue: BP1 protein is expressed in adult kidney and placenta and in low levels in normal breast and fetal liver (Chase et al., 2002).
Localisation
Both nuclear and cytoplasmic immunostaining are seen in BP1 positive breast tumors and prostate tumors (Man et al., 2005; Schwartz et al., 2009).
Function
Functionally, we demonstrated that BP1 is a repressor of the beta-globin gene, while DLX7 binds to the same DNA sequence upstream of the beta-globin gene but lacks the ability to repress it (Fu et al., 2001; Chase et al., 2002). Thus, the functions of BP1 and DLX7 are clearly different in this context. BP1 acts to repress embryonic and fetal globin genes during early development but is itself repressed during normal adult erythropoiesis.
BP1 overexpression induces increased Bcl-2 expression and decreased apoptosis. pBP1 binds to the regulatory region of the bcl-2 gene, an anti-apoptotic gene, resulting in elevated expression of Bcl-2 protein and resistance to TNF-alpha in MCF-7 breast cancer cells (Stevenson et al., 2007). Increased BP1 is associated with decreased cleavage of caspase-7, caspase-8 and caspase-9, and increased expression of PARP. Thus, high BP1 expression can lead to decreased cell death and, as shown below, increased proliferation.
BP1 appears to be a repressor of BRCA1. Three breast cancer cell lines engineered to overexpress BP1 show decreased BRCA1 RNA and protein, while cells in which BP1 is knocked down by siRNA treatment show increased BRCA1 expression, suggesting that BP1 activity may contribute to reduced BRCA1 in some breast cancers (Kluk et al., 2010).
BP1 overexpression induces increased Bcl-2 expression and decreased apoptosis. pBP1 binds to the regulatory region of the bcl-2 gene, an anti-apoptotic gene, resulting in elevated expression of Bcl-2 protein and resistance to TNF-alpha in MCF-7 breast cancer cells (Stevenson et al., 2007). Increased BP1 is associated with decreased cleavage of caspase-7, caspase-8 and caspase-9, and increased expression of PARP. Thus, high BP1 expression can lead to decreased cell death and, as shown below, increased proliferation.
BP1 appears to be a repressor of BRCA1. Three breast cancer cell lines engineered to overexpress BP1 show decreased BRCA1 RNA and protein, while cells in which BP1 is knocked down by siRNA treatment show increased BRCA1 expression, suggesting that BP1 activity may contribute to reduced BRCA1 in some breast cancers (Kluk et al., 2010).
Implicated in
Entity name
Breast cancer
Note
BP1 is activated in about 80% of invasive ductal breast (IDC) tumors. Aberrant expression of BP1 was shown by semi-quantitative RT-PCR, where 80% of tumors were BP1 positive, and by immunostaining, where 81% of tumors were BP1 positive, a remarkable agreement between mRNA and protein expression (Fu et al., 2003; Man et al., 2005). Surprisingly, 89% of the tumors of African American women (AAW) were BP1 positive, compared with 57% of the tumors of Caucasian women (p=0.04). In addition, 100% of ER negative tumors were BP1 positive, compared with 73% of ER positive (p=0.03). Both tumors of AAW and ER negative tumors are associated with aggressiveness. A group in China quantitated BP1 mRNA in the tumors of 142 Chinese women, discovering that 65% of their tumors were BP1 positive, and confirming an association between high BP1 mRNA expression and ER negative tumors (Yu et al., 2008b). Inflammatory breast cancer (IBC) is an extremely aggressive breast cancer, with approximately half the survival seen in IDC; 100% of the forty-six cases of IBC we examined were highly BP1 immunoreactive, suggesting an association between aggressiveness, frequency of BP1 positivity, and BP1 protein (pBP1) staining intensity (Man et al., 2009).
pBP1 expression correlates with breast cancer progression. The frequency of pBP1 positivity, distribution and intensity of BP1 expression all increased with the progression of tumor development from 0% (normal) to 21% in hyperplasia, 46% in ductal carcinoma in situ, and 81% in IDC (p
pBP1 expression correlates with breast cancer progression. The frequency of pBP1 positivity, distribution and intensity of BP1 expression all increased with the progression of tumor development from 0% (normal) to 21% in hyperplasia, 46% in ductal carcinoma in situ, and 81% in IDC (p
Entity name
Leukemia
Note
We examined BP1 in the bone marrow of leukemia patients by semi-quantitative RT-PCR, finding that BP1 was activated in 63% of acute myeloid leukemias (AML), including 81% of pediatric and 47% of adult patients with AML, in 32% of T-cell acute lymphocytic leukemias (ALL) but not in the pre-B ALL cases (Haga et al., 2000). Expression of BP1 occurred in primitive leukemia cells and in CD34 positive progenitors. In the same study we examined expression of DLX4 and DLX7 by designing primers specific for each isoform. Interestingly, the three isoforms were frequently co-expressed in the same cases. Next we compared the growth-inhibitory and cyto-differentiating activities of all-trans retinoic acid (ATRA) in two acute promyelocytic leukemia (APL) cells lines, NB4 (ATRA-responsive) and R4 (ATRA-resistant) cells relative to BP1 levels (Awwad et al., 2008). NB4 cells and R4 cells both expressed BP1; BP1 was repressed after ATRA treatment of NB4 cells but not R4 cells. In NB4 cells engineered to overexpress BP1, proliferation was no longer inhibited and differentiation was reduced two- to three-fold. In patients, BP1 levels were increased in all pre-treatment APL patients tested, while BP1 expression was decreased in 91% of patients after combined ATRA and chemotherapy treatment. Two patients underwent disease relapse during follow up; one patient exhibited a 42-fold increase in BP1 expression, while the other showed no change. This suggests BP1 may be part of a pathway involved in resistance to therapy.
Entity name
Prostate cancer
Note
Prostate cancer, another hormone dependent solid tumor, was examined for activation of BP1 (Schwartz et al., 2008). Significant BP1 immunoreactivity was identified in 70% of prostatic tumors, whether the analysis was performed on tissue sections (50 cases) or tissue microarray platforms (123 cases). We also observed low BP1 immunostaining in 42% of hyperplastic cells, similar to the 46% BP1 positivity in hyperplastic breast cells. Compared to normal and hyperplastic tissues, the malignant tissues consistently showed the highest number of BP1 positive cells and the highest intensity of BP1 immunostaining, similar to our observations in breast. In tissue sections, twelve cases with paired carcinoma and prostatic intraepithelial neoplasia (PIN) showed agreement, both components exhibiting strong immunoreactivity. Tumor proliferation, assayed with Ki67 immunostaining, was higher in cancer cells that were BP1 positive relative to those that were BP1 negative, in agreement with the data in breast cancer cells. These findings suggest that BP1 is an important upstream factor in the carcinogenic pathway of prostate cancer and that the expression of BP1 may reflect or directly contribute to tumor progression and/or invasion.
Entity name
Non-small cell lung cancer (NSCLC)
Note
An interesting study by Yu et al. (2008a) demonstrated that high BP1 mRNA levels occur in NSCLC tumors, compared with adjacent normal cells or normal lung samples. High mRNA levels are associated with stage III tumors, lower disease free survival (DFS) and lower overall survival. In fact, high BP1 mRNA is an independent predictor of DFS.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 18026954 | 2008 | Overexpression of BP1, a homeobox gene, is associated with resistance to all-trans retinoic acid in acute promyelocytic leukemia cells. | Awwad RT et al |
| 18992636 | 2008 | Amplification of the BP1 homeobox gene in breast cancer. | Cavalli LR et al |
| 11909945 | 2002 | BP1, a homeodomain-containing isoform of DLX4, represses the beta-globin gene. | Chase MB et al |
| 11707330 | 2001 | Distinct functions of two isoforms of a homeobox gene, BP1 and DLX7, in the regulation of the beta-globin gene. | Fu S et al |
| 12817998 | 2003 | Correlation of expression of BP1, a homeobox gene, with estrogen receptor status in breast cancer. | Fu SW et al |
| 11069021 | 2000 | BP1, a new homeobox gene, is frequently expressed in acute leukemias. | Haga SB et al |
| 20877436 | 2010 | BP1, an isoform of DLX4 homeoprotein, negatively regulates BRCA1 in sporadic breast cancer. | Kluk BJ et al |
| 15830137 | 2005 | Expression of BP1, a novel homeobox gene, correlates with breast cancer progression and invasion. | Man YG et al |
| 19242057 | 2009 | BP1, a putative signature marker for inflammatory breast cancer and tumor aggressiveness. | Man YG et al |
| 8975708 | 1996 | Genomic analysis of a new mammalian distal-less gene: Dlx7. | Nakamura S et al |
| 18931648 | 2009 | BP1, a homeoprotein, is significantly expressed in prostate adenocarcinoma and is concordant with prostatic intraepithelial neoplasia. | Schwartz AM et al |
| 17854498 | 2007 | BP1 transcriptionally activates bcl-2 and inhibits TNFalpha-induced cell death in MCF7 breast cancer cells. | Stevenson HS et al |
| 18420035 | 2008 | Prognostic significance of BP1 mRNA expression level in patients with non-small cell lung cancer. | Yu M et al |
| 17999690 | 2008 | Expression level of beta protein 1 mRNA in Chinese breast cancer patients: a potential molecular marker for poor prognosis. | Yu M et al |
Other Information
Locus ID:
NCBI: 1748
MIM: 601911
HGNC: 2917
Ensembl: ENSG00000108813
Variants:
dbSNP: 1748
ClinVar: 1748
TCGA: ENSG00000108813
COSMIC: DLX4
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000108813 | ENST00000240306 | Q92988 |
| ENSG00000108813 | ENST00000411890 | Q92988 |
| ENSG00000108813 | ENST00000611342 | Q92988 |
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 35883028 | 2022 | DNA methylation-mediated differential expression of DLX4 isoforms has opposing roles in leukemogenesis. | 3 |
| 35883028 | 2022 | DNA methylation-mediated differential expression of DLX4 isoforms has opposing roles in leukemogenesis. | 3 |
| 32281175 | 2020 | Homeodomain protein DLX4 facilitates nasopharyngeal carcinoma progression via up-regulation of YB-1. | 5 |
| 32281175 | 2020 | Homeodomain protein DLX4 facilitates nasopharyngeal carcinoma progression via up-regulation of YB-1. | 5 |
| 29738288 | 2019 | Association Between DLX4 Polymorphisms and Nonsyndromic Orofacial Clefts in a Chinese Han Population. | 5 |
| 31100338 | 2019 | Overexpression of BP1, an isoform of Homeobox Gene DLX4, promotes cell proliferation, migration and predicts poor prognosis in endometrial cancer. | 12 |
| 29738288 | 2019 | Association Between DLX4 Polymorphisms and Nonsyndromic Orofacial Clefts in a Chinese Han Population. | 5 |
| 31100338 | 2019 | Overexpression of BP1, an isoform of Homeobox Gene DLX4, promotes cell proliferation, migration and predicts poor prognosis in endometrial cancer. | 12 |
| 29575937 | 2018 | BP1, a potential biomarker for breast cancer prognosis. | 5 |
| 29575937 | 2018 | BP1, a potential biomarker for breast cancer prognosis. | 5 |
| 26325005 | 2016 | BP1 overexpression is associated with adverse prognosis in de novo acute myeloid leukemia. | 4 |
| 26485746 | 2016 | Hypermethylation of DLX4 predicts poor clinical outcome in patients with myelodysplastic syndrome. | 6 |
| 27449292 | 2016 | Beta protein 1 homeoprotein induces cell growth and estrogen-independent tumorigenesis by binding to the estrogen receptor in breast cancer. | 0 |
| 26325005 | 2016 | BP1 overexpression is associated with adverse prognosis in de novo acute myeloid leukemia. | 4 |
| 26485746 | 2016 | Hypermethylation of DLX4 predicts poor clinical outcome in patients with myelodysplastic syndrome. | 6 |
Citation
Patricia E Berg ; Saurabh Kirolikar
DLX4 (distal-less homeobox 4)
Atlas Genet Cytogenet Oncol Haematol. 2011-01-01
Online version: http://atlasgeneticsoncology.org/gene/49827/dlx4
