GBP1 (guanylate binding protein 1, interferon-inducible, 67kDa)
2009-10-01 Nathalie Britzen-Laurent , Michael Stürzl AffiliationDivision of Molecular, Experimental Surgery, Department of Surgery, Friedrich-Alexander University, Schwabachanlage 10, 91054 Erlangen, Germany
DNA/RNA
Description
The GBP1 gene consists of 11 exons (ranging from 102 to 1164 bp) and 10 introns. The coding sequence starts in the second exon.
Transcription
Only one variant mRNA has been described for GBP-1 (Accession Number: NM_002053). This mRNA is 3050 bp long with a coding sequence of 1779 bp. Four polymorphisms have been described for GBP-1: c.232A>G (codon 78 Ile>Val), c.498G>C (codon 166 Glu>Asp), c.1046C>G (codon 349 Thr>Ser), c.1226C>G (codon 409 Ala>Gly).
Pseudogene
A pseudogene has been identified for GBP1 (LOC400759 alias FLJ17004) on chromosome 1 (1p22.2, chro 1: 89645826-89663081, according to hg18-Mar_2006). Two pseudogenes have also been described in the mouse (pseudomGbp1 and pseudomGbp2).
Proteins
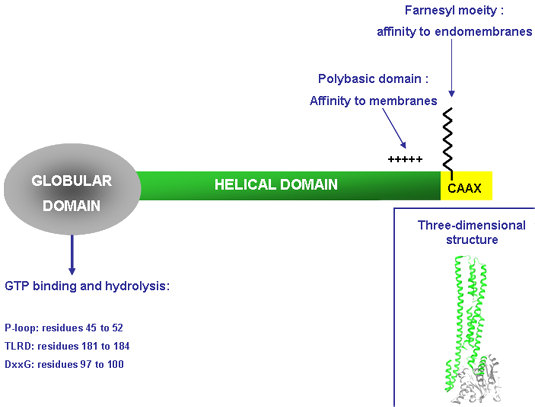
A schematic representation of the domain structure and the three-dimensional structure of GBP-1. GBP-1 consists of a globular domain (residues 1 to 278), which contains the GTP binding and hydrolysis domains, and of a helical domain (residues 279 to 593) terminated by a polybasic sequence and an isoprenylation motif (CAAX). C= Cysteine, A= aliphatic acid, X= any amino-acid (here a serine, specifically recognized by a farnesyl-transferase).
Description
GBP-1 belongs to the class of large GTPases that contains, in addition to the GBPs, three further groups of proteins, which share structural and biochemical properties: the dynamins, the Mx proteins and the atlastins. GBP-1 has a molecular weight of 67 kDa and its crystal structure has revealed the presence of two domains: (1) a N-terminal globular alpha/beta domain harbouring the GTPase activity and (2) a long C-terminal part organized in an index finger-like domain composed exclusively of seven alpha-helices (alpha7 - alpha13). The domains are connected by a short intermediate region consisting of one alpha-helix and a short two-stranded beta-sheet. In addition, GBP-1 harbours a C-terminal CAAX isoprenylation motif. GTPases typically harbour three classical GTP-binding domains: the phosphate-binding P-loop GXXXXGK(S/T), the phosphate- and Mg2+-binding DXXG motif (G, glycine; K, lysine; S, serine; D, aspartic acid; T, threonine; and X, any amino acid) and the guanine nucleotide-specificity providing (N/T)KXD motif (N, asparagine). In GBP-1 the classical (N/T)KXD motif is substituted by a conserved arginine-aspartic acid (RD)-motif (TLRD, with L, leucine and R, arginine).
Expression
GBP-1 was initially shown to be among the most highly induced proteins in human fibroblasts exposed to interferon (IFN)-gamma. Subsequently, it was reported that in vitro hGBP-1 expression can be induced by IFN-gamma in many different cell types including endothelial cells, fibroblasts, keratinocytes, B-cells, T-cells or peripheral blood mononuclear cells. In vivo expression of hGBP-1 has been predominantly detected in inflammatory tissues and has been found to be associated almost exclusively with endothelial cells and monocytes. It has been shown subsequently that hGBP-1 expression in endothelial cells is also induced by other pro-inflammatory cytokines such as IFNalpha, TNFalpha and IL1alpha/IL1beta. Many other cytokines (IL-4, IL-6, IL-10, IL-18), chemokines (MCP-1, PF4) or growth factors (angiopoietin-2, PDGF B/B) tested did not affect GBP-1 expression in these cells. Interestingly, the two major angiogenic growth factors (AGF: bFGF, VEGF) are able to inhibit the expression of GBP-1 induced by inflammatory cytokines.
Localisation
The localization of GBP-1 is primarily cytosolic and its distribution is granular. Plasma membrane association at the level of tight junctions has also been described. In addition, GBP-1 has been shown to be secreted from IFN-gamma-stimulated endothelial cells through a non-classical secretion pathway.
Function
GTPase activity
GBP-1 can bind the three nucleotides GTP, GDP and GMP with a relative low affinity (Kd mant-GMP=0,53 μM, Kd mant-GDP=2,4 μM, Kdmant-GppNHp=1,1 μM). GBP-1 has however the ability to hydrolyse GTP to both GDP and GMP with a high hydrolysis rate (max 95 min-1). At physiological temperature GMP is the major product (90%) of hGBP1. On the contrary to small GTPases like Ras, GBP-1 does not require the presence of GEF (GTP exchange factor) or GAP (GTPase activating protein) proteins for its GTPase activity. In the case of GBP-1, the GTP hydrolysis is self-stimulated by oligomerization of the protein.
GBP-1 is involved in IFN-gamma response, either in infection or in inflammation.
GBP-1 can bind the three nucleotides GTP, GDP and GMP with a relative low affinity (Kd mant-GMP=0,53 μM, Kd mant-GDP=2,4 μM, Kdmant-GppNHp=1,1 μM). GBP-1 has however the ability to hydrolyse GTP to both GDP and GMP with a high hydrolysis rate (max 95 min-1). At physiological temperature GMP is the major product (90%) of hGBP1. On the contrary to small GTPases like Ras, GBP-1 does not require the presence of GEF (GTP exchange factor) or GAP (GTPase activating protein) proteins for its GTPase activity. In the case of GBP-1, the GTP hydrolysis is self-stimulated by oligomerization of the protein.
GBP-1 is involved in IFN-gamma response, either in infection or in inflammation.
Homology
The human GBP family comprises 7 highly homologous members, all located on the chromosome 1. GBP-1 is to 77% similar to GBP-2, 88% to GBP-3, 56% to GBP-4, 68% to GBP-5, 54% to GBP-6 and 56% to GBP-7. Homologues have been found in various species like zebrafish, chimpanzee (99% homology), rat, dog or mouse. GBP-1 shares 59% of homology with murine GBP-1 and murine GBP-2.
Mutations
Note
No somatic or germline mutations have yet been reported for human GBP-1.
Implicated in
Entity name
Infection
Note
GBP-1 exhibits antiviral activity against vesicular stomatitis virus and encephalomyocarditis virus. The expression of GBP-1 is also elevated in the blood of patients with a chronic active Epstein-Barr Virus infection. Furthermore GBP-1 and GBP-2 can potentiate the inhibitory effects of IFN-gamma on Chlamydia trachomatis growth. Finally, elevated concentrations of GBP-1 have been detected in the cerebrospinal fluid of patients with bacterial meningitis.
Entity name
Inflammation
Note
As GBP-1 is mainly expressed in endothelial cells in vivo in a context of inflammation, its effects have been extensively studied in these cells. It has been showed in endothelial cells that GBP-1 mediates the effects of inflammatory cytokines and inhibits proliferation, spreading, migration or invasion. GBP-1 is also involved in the regulation of apoptosis and senescence in endothelial cells stimulated with IFN-alpha. Evidence for an implication in cancer has been found for GBP-1 (see below).
Entity name
Colorectal carcinomas
Note
GBP-1 protein is strongly expressed in the stroma of about one third of colorectal carcinomas in association with an IFN-gamma dominated Th-1-like angiostatic immune response.
Prognosis
This expression correlates with an increased cancer-related 5-year survival.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 10087221 | 1999 | Interferon-induced guanylate binding protein-1 (GBP-1) mediates an antiviral effect against vesicular stomatitis virus and encephalomyocarditis virus. | Anderson SL et al |
| 3934165 | 1985 | Affinity purification of an interferon-induced human guanylate-binding protein and its characterization. | Cheng YS et al |
| 6305951 | 1983 | Interferon induction of fibroblast proteins with guanylate binding activity. | Cheng YS et al |
| 18025219 | 2007 | Extensive characterization of IFN-induced GTPases mGBP1 to mGBP10 involved in host defense. | Degrandi D et al |
| 16511497 | 2006 | How guanylate-binding proteins achieve assembly-stimulated processive cleavage of GTP to GMP. | Ghosh A et al |
| 12881412 | 2003 | The guanylate binding protein-1 GTPase controls the invasive and angiogenic capability of endothelial cells through inhibition of MMP-1 expression. | Guenzi E et al |
| 18260761 | 2008 | Oligonucleotide microarray analysis of gene expression profiles followed by real-time reverse-transcriptase polymerase chain reaction assay in chronic active Epstein-Barr virus infection. | Ito Y et al |
| 12414522 | 2002 | Guanylate-binding protein-1 expression is selectively induced by inflammatory cytokines and is an activation marker of endothelial cells during inflammatory diseases. | Lubeseder-Martellato C et al |
| 15937107 | 2005 | Golgi targeting of human guanylate-binding protein-1 requires nucleotide binding, isoprenylation, and an IFN-gamma-inducible cofactor. | Modiano N et al |
| 8830800 | 1996 | Prenylation of an interferon-gamma-induced GTP-binding protein: the human guanylate binding protein, huGBP1. | Nantais DE et al |
| 18697200 | 2008 | Angiostatic immune reaction in colorectal carcinoma: Impact on survival and perspectives for antiangiogenic therapy. | Naschberger E et al |
| 16689661 | 2006 | In silico genomic analysis of the human and murine guanylate-binding protein (GBP) gene clusters. | Olszewski MA et al |
| 16894355 | 2006 | Interferon-alpha prevents apoptosis of endothelial cells after short-term exposure but induces replicative senescence after continuous stimulation. | Pammer J et al |
| 15504415 | 2004 | Identification of residues in the human guanylate-binding protein 1 critical for nucleotide binding and cooperative GTP hydrolysis. | Praefcke GJ et al |
| 10676968 | 2000 | Structure of human guanylate-binding protein 1 representing a unique class of GTP-binding proteins. | Prakash B et al |
| 10970849 | 2000 | Triphosphate structure of guanylate-binding protein 1 and implications for nucleotide binding and GTPase mechanism. | Prakash B et al |
| 19079332 | 2009 | Guanylate-binding protein-1 is expressed at tight junctions of intestinal epithelial cells in response to interferon-gamma and regulates barrier function through effects on apoptosis. | Schnoor M et al |
| 7512561 | 1994 | The interferon-induced 67-kDa guanylate-binding protein (hGBP1) is a GTPase that converts GTP to GMP. | Schwemmle M et al |
| 19652711 | 2009 | Human guanylate binding proteins potentiate the anti-chlamydia effects of interferon-gamma. | Tietzel I et al |
| 17266443 | 2007 | Unique features of different members of the human guanylate-binding protein family. | Tripal P et al |
| 16108726 | 2005 | The guanylate-binding proteins (GBPs): proinflammatory cytokine-induced members of the dynamin superfamily with unique GTPase activity. | Vestal DJ et al |
| 18697840 | 2008 | Guanylate binding protein-1 inhibits spreading and migration of endothelial cells through induction of integrin alpha4 expression. | Weinländer K et al |
Other Information
Locus ID:
NCBI: 2633
MIM: 600411
HGNC: 4182
Ensembl: ENSG00000117228
Variants:
dbSNP: 2633
ClinVar: 2633
TCGA: ENSG00000117228
COSMIC: GBP1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000117228 | ENST00000370473 | P32455 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38167153 | 2024 | Function and mechanism of GBP1 in the development and progression of cervical cancer. | 0 |
| 38315728 | 2024 | Guanylate-binding protein 1 acts as a pro-viral factor for the life cycle of hepatitis C virus. | 0 |
| 38622605 | 2024 | Interferon-γ-induced GBP1 is an inhibitor of human papillomavirus 18. | 0 |
| 38840385 | 2024 | GBP1 promotes cutaneous squamous cell carcinoma proliferation and invasion through activation of STAT3 by SP1. | 0 |
| 38860570 | 2024 | Guanylate-binding protein 1 inhibits Hantaan virus infection by restricting virus entry. | 0 |
| 38167153 | 2024 | Function and mechanism of GBP1 in the development and progression of cervical cancer. | 0 |
| 38315728 | 2024 | Guanylate-binding protein 1 acts as a pro-viral factor for the life cycle of hepatitis C virus. | 0 |
| 38622605 | 2024 | Interferon-γ-induced GBP1 is an inhibitor of human papillomavirus 18. | 0 |
| 38840385 | 2024 | GBP1 promotes cutaneous squamous cell carcinoma proliferation and invasion through activation of STAT3 by SP1. | 0 |
| 38860570 | 2024 | Guanylate-binding protein 1 inhibits Hantaan virus infection by restricting virus entry. | 0 |
| 36460216 | 2023 | Differential expression of interferon inducible protein: Guanylate binding protein (GBP1 & GBP2) in severe dengue. | 2 |
| 37298652 | 2023 | Human GBP1 Is Involved in the Repair of Damaged Phagosomes/Endolysosomes. | 2 |
| 37737612 | 2023 | Human GBP1 facilitates the rupture of the Legionella-containing vacuole and inflammasome activation. | 0 |
| 36460216 | 2023 | Differential expression of interferon inducible protein: Guanylate binding protein (GBP1 & GBP2) in severe dengue. | 2 |
| 37298652 | 2023 | Human GBP1 Is Involved in the Repair of Damaged Phagosomes/Endolysosomes. | 2 |
Citation
Nathalie Britzen-Laurent ; Michael Stürzl
GBP1 (guanylate binding protein 1, interferon-inducible, 67kDa)
Atlas Genet Cytogenet Oncol Haematol. 2009-10-01
Online version: http://atlasgeneticsoncology.org/gene/50147/gbp1
