FGF2 (fibroblast growth factor 2 (basic))
2010-05-01 Leigh-Ann MacFarlane , Paul Murphy AffiliationDalhousie University, Department of Physiology, Biophysics, Faculty of Medicine, 5850 College Street Sir Charles Tupper Medical Building, Halifax, Nova Scotia B3H 1X5, Canada
Identity
HGNC
LOCATION
4q27
LOCUSID
ALIAS
BFGF,FGF-2,FGFB,HBGF-2
FUSION GENES
DNA/RNA
Note
Human FGF2 is located on chromosome 4 in the region of 4q25-4q27 on the forward DNA strand, opposite to the NUDT6 gene locus. FGF2 and NUDT6 overlap at their 3 ends.
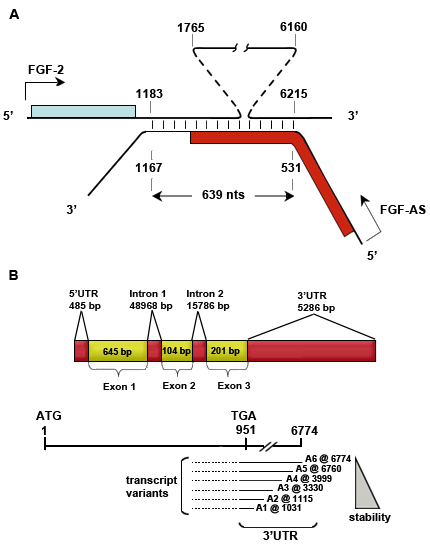
Figure A. The schematic representation of the human FGF2 and NUDT6 (FGF-AS) gene transcript overlap (colored boxes, coding region; connecting vertical lines, complementary regions between transcripts). Adapted from: MacFarlane LA, et al., 2010. Molecular Endocrinology 24.
Figure B. The schematic representation of the human FGF2 gene and its RNA transcript (yellow rectangles, coding regions; UTR, untranslated region; ATG, translation start codon; TGA, translation stop codon; A1, polyadenylation site 1).
Description
Human FGF2 gene is 70990 bp in length, composed of a 5UTR, 3 exons, 2 introns and an extremely long 3UTR. The 5 and 3UTR contain a variety of regulatory elements that regulate FGF2 expression in response to growth factors, cell density, neurotransmitters, hormones and second messenger pathways. The FGF2 core promoter maps from -1800 to +314 (relative to the transcription start site +1, up-stream -), which is 44 kb upstream of the NUDT6 promoter.
The promoter lacks the typical consensus CATT and TATA box motifs. The distal -512/-854 region contains a single negative regulatory domain (-521/-854), a cell density dependent element (-512/-650) with STAT transcription factor binding sites, a growth factor responsive element (-512/-554) with STAT transcription factors binding sites, a protein kinase C (PKC)/cyclic adenosine monophosphate (cAMP) responsive element (-556/-624), and a dyad symmetry element (-597/-643). The proximal -511/+314 region maintains low promoter basal transcription activity and contains specific transcription factor binding sites which include AP-1 at the -243 position, p53 (wild type and mutant) between -20/+50, and Sp1 at positions -166, -139, -83, and -65.
The unusually long AU-rich 3UTR of FGF2 contains multiple regulatory elements that regulate polyadenylation, translation initiation, and RNA stability. A unique translation enhancer located in the 3UTR just upstream of the most distal polyadenylation site (+5404/+6775) is involved in selecting the active polyadenylation site and modulating the use of alternative translation initiation sites. A destabilizing element (referred to as DEST) located between the first and second polyadenylation sites (+1019/+3326) alters mRNA stability. Additionally, two regions of the FGF2 3UTR (+1183/+1765 and +6160/+6215) are fully complementary to the 3end of the NUDT6 transcript, which enables mRNAs to form a sense-antisense pair. The formation of a sense-antisense pair has been implicated in the regulation of FGF2 mRNA stability.
The promoter lacks the typical consensus CATT and TATA box motifs. The distal -512/-854 region contains a single negative regulatory domain (-521/-854), a cell density dependent element (-512/-650) with STAT transcription factor binding sites, a growth factor responsive element (-512/-554) with STAT transcription factors binding sites, a protein kinase C (PKC)/cyclic adenosine monophosphate (cAMP) responsive element (-556/-624), and a dyad symmetry element (-597/-643). The proximal -511/+314 region maintains low promoter basal transcription activity and contains specific transcription factor binding sites which include AP-1 at the -243 position, p53 (wild type and mutant) between -20/+50, and Sp1 at positions -166, -139, -83, and -65.
The unusually long AU-rich 3UTR of FGF2 contains multiple regulatory elements that regulate polyadenylation, translation initiation, and RNA stability. A unique translation enhancer located in the 3UTR just upstream of the most distal polyadenylation site (+5404/+6775) is involved in selecting the active polyadenylation site and modulating the use of alternative translation initiation sites. A destabilizing element (referred to as DEST) located between the first and second polyadenylation sites (+1019/+3326) alters mRNA stability. Additionally, two regions of the FGF2 3UTR (+1183/+1765 and +6160/+6215) are fully complementary to the 3end of the NUDT6 transcript, which enables mRNAs to form a sense-antisense pair. The formation of a sense-antisense pair has been implicated in the regulation of FGF2 mRNA stability.
Transcription
The full length 6774 bp FGF2 transcript contains the 3 exons and the 3untranslated region, which contains at least 6 alternative polyadenylation sites. Alternative use of polyadenylation sites yields a variety of transcripts that have the same coding region but different length 3UTR and contained regulatory elements. Consequently, transcript stability varies with the length of the 3UTR, the shortest transcript is the most stable and the longest is the least stable.
Pseudogene
NA.
Proteins
Note
Human FGF2 encodes 5 biologically active isoforms that differ in molecular weight, subcellular localization and function.
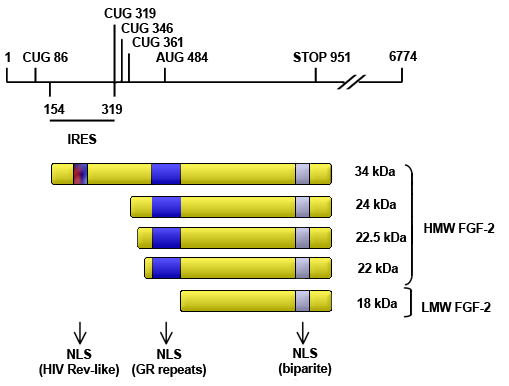
Schematic representing human FGF2 isoform expression by alternative translation initiation (CUG, alternative leucine translation initiation codon; AUG, classical methionine translation initiation codon; IRES, internal ribosome entry sire; kDa, kilo Dalton; LMW, low molecular weight; HMW, high molecular weight; NLS, nuclear localization sequence; GR, Glutamic acid - Arginine; HIV, human immunodeficiency virus).
Description
The 6774-nt human FGF2 mRNA can have translation initiated at one of five in-frame codons indicated to generate five different molecular weight isoforms by cap-dependent or IRES dependent translation. All isoforms contain a carboxyl-terminal bipartite NLS. The HMW isoforms (34, 24, 22.5 and 22 kDa), initiated from CUG codons (86, 319, 346, 361), also contain an amino-terminal Glutamic acid - Arginine repeat domain that acts as an NLS. The 34 kDa isoform contains an additional NLS similar in structure to that of the human immunodeficiency virus (HIV) Rev protein.
Expression
FGF2 is expressed in a developmental and tissue specific manner. Differentiating populations of cells also have shifting levels of FGF2 protein content. Cell phenotype and environment can affect the length of the FGF2 mRNA and isoform expression by post-transcriptional regulation of polyadenylation. Primary cell types almost exclusively use the distal polyadenylation site to generate the full length 6775-nt FGF2 mRNA encompassing the full 3UTR and all regulatory elements, whereas transformed and stressed cells favor the use of the most proximal polyadenylation site to generate transcripts with a much shorter 3UTR lacking critical regulatory elements. In contrast to primary cells that predominantly express the LMW FGF2, the shorter FGF2 mRNA transcripts in stressed and transformed cells translate from the upstream CUG initiation codons to generate HMW FGF2 isoforms.
FGF2 protein expression has been classified into two distinct patterns. The first, characterized by high levels of the AUG-initiated LMW isoform accompanied by low/undetectable levels of CUG-initiated HMW isoforms, is observed in normal cells such as skin fibroblasts, retinal pigment epithelial cells and aortic endothelial cells. In contrast, the second pattern, defined by high levels of HMW isoforms and low/undetectable levels of LMW, is seen in transformed cells including uterus carcinoma (HeLa cells), liver adenocarcinoma (SK-Hep-1 cells), pancreatic carcinoma (MIA PaCa-2 cells), epidermoid carcinoma (A-431 cells), breast adenocarcinoma (MCF-7 cells) and colon adenocarcinoma (HT-29). Fluctuations in FGF2 protein expression and localization occur in response to cell density, cell cycle and differentiation.
Low density cell cultures have significantly higher LMW FGF2 expression which is predominantly nuclear, compared to high density cultures that express low levels of cytosolic FGF2. This variation can be attributed to differences in proliferation among the populations, as FGF2 is expressed in a cell cycle dependent manner. Increased proliferation in low density populations correlates with elevated FGF2 levels during the G0-G1 transition of the cell cycle and nuclear accumulation of FGF2.
FGF2 protein expression has been classified into two distinct patterns. The first, characterized by high levels of the AUG-initiated LMW isoform accompanied by low/undetectable levels of CUG-initiated HMW isoforms, is observed in normal cells such as skin fibroblasts, retinal pigment epithelial cells and aortic endothelial cells. In contrast, the second pattern, defined by high levels of HMW isoforms and low/undetectable levels of LMW, is seen in transformed cells including uterus carcinoma (HeLa cells), liver adenocarcinoma (SK-Hep-1 cells), pancreatic carcinoma (MIA PaCa-2 cells), epidermoid carcinoma (A-431 cells), breast adenocarcinoma (MCF-7 cells) and colon adenocarcinoma (HT-29). Fluctuations in FGF2 protein expression and localization occur in response to cell density, cell cycle and differentiation.
Low density cell cultures have significantly higher LMW FGF2 expression which is predominantly nuclear, compared to high density cultures that express low levels of cytosolic FGF2. This variation can be attributed to differences in proliferation among the populations, as FGF2 is expressed in a cell cycle dependent manner. Increased proliferation in low density populations correlates with elevated FGF2 levels during the G0-G1 transition of the cell cycle and nuclear accumulation of FGF2.
Localisation
Subcellular localization and expression of FGF2 isoforms is determined by cell types, environment, level of differentiation, cell cycle phase and cell density. FGF2 isoform subcellular localization is essential for specific biological functions. Although all FGF2 isoforms can be found in the nucleus, cytoplasm and extracellular space at one time or another, they exhibit preferential localization.
The LMW18 kDa FGF2 is primarily found in the cytoplasm. However, LMW FGF2 can be secreted and subsequently internalized to the cytoplasm and translocated to the nucleus. FGF2 lacks a conventional amino terminal signal sequence and therefore is secreted via a non-classical secretory pathway. The HMW FGF2 isoforms are predominantly located in the nucleus but are able to shuttle back to the cytoplasm. HMW FGF2 isoforms can be released from cells through vesicle shedding at the plasma membrane and as a result of cell injury or death that compromises cell membrane integrity. However, it is unclear to what extent HMW FGF2 isoforms exist extracellularly in vivo.
Changes in isoform distribution can occur in response to cAMP and PKC signaling, cell density, cellular stress and post-translational modification.
The LMW18 kDa FGF2 is primarily found in the cytoplasm. However, LMW FGF2 can be secreted and subsequently internalized to the cytoplasm and translocated to the nucleus. FGF2 lacks a conventional amino terminal signal sequence and therefore is secreted via a non-classical secretory pathway. The HMW FGF2 isoforms are predominantly located in the nucleus but are able to shuttle back to the cytoplasm. HMW FGF2 isoforms can be released from cells through vesicle shedding at the plasma membrane and as a result of cell injury or death that compromises cell membrane integrity. However, it is unclear to what extent HMW FGF2 isoforms exist extracellularly in vivo.
Changes in isoform distribution can occur in response to cAMP and PKC signaling, cell density, cellular stress and post-translational modification.
Function
FGF2 is a pleiotropic signaling molecule involved in many biological processes including angiogenesis, embryonic development (brain, limb, lung, heart, muscle, bone, blood, eye and skin) and wound healing. Despite complex involvement in any aspects of embryogenesis FGF2 knockout mice are viable, functioning and do not display any apparent neurological deficit. FGF2 deficient mice have impaired brain development, blood pressure regulation, wound repair and bone formation.
LMW FGF2 stimulates cell growth, proliferation, migration and differentiation via FGFR signaling and ligand receptor complex internalization. The FGF2 mitogenic response is controlled by direct and indirect regulation of nuclear kinase and transcription factor activity essential for ribosome biogenesis during cell proliferation and growth. Translokin, a cytoplasmic protein of relative molecular mass 55 kDa, interacts specifically with the 18 kDa form of FGF-2 and mediates its translocation to the nucleus. Nuclear LMW FGF2 binds the transcription factor UBF to directly regulate ribosomal RNA (rRNA) transcription. Nuclear LMW FGF2 also binds and modulates the nuclear kinases CK2 and ribosomal S6 kinase 2 (RSK2) responsible for nucleolin and histone phosphorylation, respectively, which are essential for ribosome biogenesis and cell cycle progression. LMW FGF2 indirectly influences rRNA transcription through receptor-mediated ERK dependent phosphorylation of the transcription initiation factor TIF-1A, which is essential for RNA polymerase 1 transcription.
LMW FGF2 stimulates a mitogenic response in most cell types. However the particular signaling pathway activated appears to be dependent on cell type and the specific FGFR. Receptor-mediated ERK activation also stimulates cell migration and differentiation. However, other signaling pathways such as PI3K and MAPK have also been implicated in regulation of these processes. LMW FGF2 stimulated migration, growth and differentiation responses mediate angiogenesis, wound repair, embryonic development and maintenance of vascular tone.
The affects of HMW are dependent on isoform, expression level and cell type. The majority of HMW FGF2 functions require nuclear localization. The HMW nuclear forms of FGF-2 have been reported to interact with a 55 kDa nuclear protein, FIF (FGF-2-interacting-factor), which interacts specifically with FGF-2 but not with FGF-1, FGF-3, or FGF-6. Some of the biological effects of FGF-2 may be mediated by interaction with FIF, which has anti-apoptotic activity.
Nuclear 34 kDa HMW FGF2 acts as a survival factor, sustaining cell growth in low-serum conditions. However, in normal conditions the effects of HMW FGF2 on proliferation vary with cell type and expression level. High levels of HMW FGF2 induce proliferation in a variety of cells including aortic endothelial, fibroblasts, glioma, pancreatic tumor and liver adenocarcinoma cells. Low levels of HMW FGF2 inhibit cell proliferation in glioma and fibroblast cells. These effects have been attributed in part to the ability of HMW FGF2 to control mitosis by inhibiting phosphorylation of the translation initiation factor 4E-BP1, which is critical for translation associated with cell cycle progression. In contrast, low levels of HMW FGF2 favor proliferation in cardiomyocytes and embryonic kidney cells while high levels inhibit proliferation and promote cell death by inducing chromatin compaction and cytosolic release of cytochrome C. Furthermore, HMW FGF2 up-regulates the growth inhibiting nuclear protein 1 (Nupr1) which is related to the High Mobility Group (HMG) of proteins that function in chromatin remodeling and transcription factor recruitment.
HMW FGF2 has also been implicated in apoptosis, cell adhesion, migration and differentiation. HMW FGF2 can suppress apoptosis, which could be in part attributable to its ability to bind to the prosurvival factor API5. However, FGF2 has also been shown to induce apoptosis. This effect has been associated with the observation that FGF2 overexpression reduces the antiapoptotic protein BCL-2 to promote apoptosis. HMW FGF2 up-regulates cell adhesion molecules and stabilizes focal adhesion complexes in a variety of tissue, which may explain the ability of HMW FGF2 to suppress migration. Additionally, migration suppression could be associated with the level of cellular differentiation. HMW FGF2 has been shown to induce high level differentiation.
LMW FGF2 stimulates cell growth, proliferation, migration and differentiation via FGFR signaling and ligand receptor complex internalization. The FGF2 mitogenic response is controlled by direct and indirect regulation of nuclear kinase and transcription factor activity essential for ribosome biogenesis during cell proliferation and growth. Translokin, a cytoplasmic protein of relative molecular mass 55 kDa, interacts specifically with the 18 kDa form of FGF-2 and mediates its translocation to the nucleus. Nuclear LMW FGF2 binds the transcription factor UBF to directly regulate ribosomal RNA (rRNA) transcription. Nuclear LMW FGF2 also binds and modulates the nuclear kinases CK2 and ribosomal S6 kinase 2 (RSK2) responsible for nucleolin and histone phosphorylation, respectively, which are essential for ribosome biogenesis and cell cycle progression. LMW FGF2 indirectly influences rRNA transcription through receptor-mediated ERK dependent phosphorylation of the transcription initiation factor TIF-1A, which is essential for RNA polymerase 1 transcription.
LMW FGF2 stimulates a mitogenic response in most cell types. However the particular signaling pathway activated appears to be dependent on cell type and the specific FGFR. Receptor-mediated ERK activation also stimulates cell migration and differentiation. However, other signaling pathways such as PI3K and MAPK have also been implicated in regulation of these processes. LMW FGF2 stimulated migration, growth and differentiation responses mediate angiogenesis, wound repair, embryonic development and maintenance of vascular tone.
The affects of HMW are dependent on isoform, expression level and cell type. The majority of HMW FGF2 functions require nuclear localization. The HMW nuclear forms of FGF-2 have been reported to interact with a 55 kDa nuclear protein, FIF (FGF-2-interacting-factor), which interacts specifically with FGF-2 but not with FGF-1, FGF-3, or FGF-6. Some of the biological effects of FGF-2 may be mediated by interaction with FIF, which has anti-apoptotic activity.
Nuclear 34 kDa HMW FGF2 acts as a survival factor, sustaining cell growth in low-serum conditions. However, in normal conditions the effects of HMW FGF2 on proliferation vary with cell type and expression level. High levels of HMW FGF2 induce proliferation in a variety of cells including aortic endothelial, fibroblasts, glioma, pancreatic tumor and liver adenocarcinoma cells. Low levels of HMW FGF2 inhibit cell proliferation in glioma and fibroblast cells. These effects have been attributed in part to the ability of HMW FGF2 to control mitosis by inhibiting phosphorylation of the translation initiation factor 4E-BP1, which is critical for translation associated with cell cycle progression. In contrast, low levels of HMW FGF2 favor proliferation in cardiomyocytes and embryonic kidney cells while high levels inhibit proliferation and promote cell death by inducing chromatin compaction and cytosolic release of cytochrome C. Furthermore, HMW FGF2 up-regulates the growth inhibiting nuclear protein 1 (Nupr1) which is related to the High Mobility Group (HMG) of proteins that function in chromatin remodeling and transcription factor recruitment.
HMW FGF2 has also been implicated in apoptosis, cell adhesion, migration and differentiation. HMW FGF2 can suppress apoptosis, which could be in part attributable to its ability to bind to the prosurvival factor API5. However, FGF2 has also been shown to induce apoptosis. This effect has been associated with the observation that FGF2 overexpression reduces the antiapoptotic protein BCL-2 to promote apoptosis. HMW FGF2 up-regulates cell adhesion molecules and stabilizes focal adhesion complexes in a variety of tissue, which may explain the ability of HMW FGF2 to suppress migration. Additionally, migration suppression could be associated with the level of cellular differentiation. HMW FGF2 has been shown to induce high level differentiation.
Homology
FGF2 is a member of a large family of structurally related heparin-binding proteins (the FGFs) involved in the regulation of cell proliferation, growth and differentiation.
Implicated in
Entity name
Glioma
Prognosis
Accumulation of FGF-2 in the nucleus is a negative prognostic indicator for survival of patients with astrocytic tumors (Fukui et al., 2003).
Oncogenesis
FGF-2 plays a critical role in nervous system development and dysregulated expression has been implicated in the pathogenesis of CNS tumors of glial origin. FGF-2 is upregulated during reactive gliosis (Frautschy et al., 1991), and in transformed cells of glial origin (Murphy et al., 1989), and is overexpressed in >90% of malignant gliomas. The level of expression of FGF-2 correlates with tumor grade and extent of anaplasia in gliomas (Zagzag et al., 1990), and with clinical outcome (Takahashi et al., 1992). Transfection of fetal astrocytes with a vector expressing FGF-2 modified to include a secretory signal peptide sequence results in anchorage-independent growth, loss of contact inhibition, and decreased glial fibrillary acidic protein immunoreactivity consistent with cellular transformation (Gately et al., 1995). Furthermore, glioma cell proliferation and colony formation in soft agar can be inhibited by FGF-2 specific antisense oligonucleotides, demonstrating a direct role of this factor in the transformed phenotype (Murphy et al., 1992). These data support the concept that FGF-2 expression could be a key event in glial tumorigenesis and may be necessary for the sustained growth of human gliomas.
Entity name
Esophageal carcinoma
Prognosis
Overexpression of FGF-2 was associated with significantly increased risk for tumor recurrence and reduced overall survival.
Oncogenesis
FGF-2 has been shown to be expressed in all of 13 esophageal squamous cell carcinoma cell lines tested, but in only one of seven gastric carcinoma cell lines (Iida et al., 1994). FGF-2 is also elevated in esophageal adenocarcinoma and in pre-malignant and dysplastic Barrett esophagus tissues (Lord et al., 2003; Barclay et al., 2005) suggesting an autocrine or paracrine role in the development of esophageal tumorigenesis. In a study of esophageal tumor samples and corresponding normal tissue from 41 males and 7 females, FGF-2 protein was not detected in any normal esophageal squamous epithelia but was found to be overexpressed in 83% (40 of 48) of tumors, where immunoreactivity was localized exclusively to the cytoplasm (Barclay et al., 2005). Remarkably, co-expression of the FGF antisense gene (NUDT6) ameliorated the effects of FGF-2 expression, suggesting that FGF-2 expression may be regulated by an endogenous antisense RNA.
Oncogenesis
MEN-1 is an autosomal dominant syndrome characterized by hyperplasia and tumors of parathyroid, pancreatic islet, and anterior pituitary glands. Mitogenic activity in the serum of MEN-1 patients stimulates in vitro growth of mixed cultures of epithelial and mesenchymal cells of parathyroid origin (Brandi et al., 1986), and this activity is inhibited by neutralizing anti-FGF-2 antibodies (Zimering et al., 1990). Patients with MEN-1 and pituitary tumors have significantly elevated levels of immunoreactive FGF-2 in their circulation which is decreased following pituitary surgery or after initiation of bromocryptine therapy, suggesting that pituitary tumors are a possible source of high circulating bFGF immunoreactivity in MEN-1 plasma (Zimering et al., 1993).
Entity name
Lymphoproliferative diseases
Oncogenesis
Intracellular FGF-2 has been detected in several lymphoproliferative diseases and is associated with more advanced disease.
Entity name
Chronic lymphocytic leukemia
Oncogenesis
Chronic lymphocytic leukemia is associated with elevated intracellular levels of FGF-2, which correlated with disease stage and associated with resistance to chemotherapy (Menzel et al., 1996).
Entity name
Oncogenesis
Hairy cell leukemia tumor cells express FGF-2, which has been suggested to mediate the resistance to chemotherapy and survival of the malignant cells (Gruber et al., 1999).
Entity name
Oncogenesis
FGF-2 levels are significantly higher in plasma cell lysates from patients with active multiple myeloma, compared to patients with inactive disease, and this correlates with increased bone marrow angiogenesis (Vacca et al., 1999). Furthermore, multiple myeloma patients who respond to chemotherapy show a significant decrease in serum FGF-2 levels, whereas nonresponders do not (Sezer et al., 2001).
Entity name
Non-Hodgkins lymphoma
Oncogenesis
FGF-2 and its receptor are thought to be involved in the survival of the lymphoma cells and their resistance to therapy. The expression of FGF-2 and its receptor in lymphoma cells has a prognostic significance: patients who express FGF-2 have a significantly worse survival than those who do not, while patients expressing fibroblast growth factor receptor-1 were less likely to achieve complete remission than those lacking the receptor (Pazgal et al., 2002).
Entity name
Renal cell carcinoma
Oncogenesis
Renal cell carcinoma is associated with elevated serum levels of FGF-2 (Fujimoto et al., 1991). Horstmann et al. (2005) reported that circulating FGF-2 levels are significantly higher in patients with renal cell carcinoma compared to healthy volunteers or patients with benign urologic diseases. Although overall there was no significant correlation between serum FGF-2 levels and tumor stage, patients with T3 tumors had higher levels of FGF-2 compared to patients with tumors classified as T2 disease. Significantly higher serum FGF-2 levels were detected in patients with metastatic disease compared to patients with non-metastatic tumors.
Entity name
Pancreatic carcinoma
Oncogenesis
In pancreatic carcinomas, there was a significant correlation between the presence of either FGF-1 or FGF-2 in pancreatic cancer cells and advanced tumor stage, and between the presence of FGF-2 and reduced patient survival (Yamanaka et al., 1993). Yamazaki et al. (1997) reported that FGF-2-positive tumors exhibited the highest proliferative indices for both tumor and endothelial cells, and suggested that FGF-2 overexpression may give pancreatic carcinoma cells a growth advantage through autocrine/paracrine mechanisms, and by stimulation of angiogenesis.
Entity name
Lung carcinomas: non-small cell lung carcinoma and small cell lung carcinoma
Oncogenesis
Non-small cell lung carcinoma (NSCLC): Volm et al. (1997) reported that all tumor specimens examined expressed some level of FGF-2 and its receptor FGFR-1. Patients with high FGFR-1 expression had significantly shorter survival than patients with weak or moderate expression, but no correlation was found between FGF-2 expression and patient survival.
Small cell lung carcinoma (SCLC): FGF-2 has been implicated in promoting the development of chemoresistance, which is the hallmark of these tumors. This occurs in part by FGF-2 regulated up-regulation of Bcl-XL and Bcl-2 and inhibition of apoptosis via a MEK-2 mediated pathway (Pardo et al., 2002).
Small cell lung carcinoma (SCLC): FGF-2 has been implicated in promoting the development of chemoresistance, which is the hallmark of these tumors. This occurs in part by FGF-2 regulated up-regulation of Bcl-XL and Bcl-2 and inhibition of apoptosis via a MEK-2 mediated pathway (Pardo et al., 2002).
Entity name
Breast carcinomas
Oncogenesis
The evidence in the literature is somewhat contradictory regarding the expression and role of FGF-2 in breast carcinoma. Luqmani et al. reported that, in cultured cells FGF-2 expression was found only in normal cells while it is largely undetectable in most malignant cell lines, including MCF-7, T-47D, ZR-75-1, and MDA-MB-231 (Luqmani et al., 1992). In contrast, FGF-2 has been reported to be elevated in the urine (Nguyen et al., 1994) and in nipple fluid (Sartippour et al., 2005) of patients with breast carcinoma. Smith et al. (1999) reported that FGF-2 levels were more than 10-fold higher in tumor cytosols compared to reduction mammoplasty tissue and 3-fold compared to non-neoplastic cytosols from the same breast as the tumor. However, high FGF-2 levels were significantly related to tumors of low grade and of small size. They reported no significant relationship between FGF-2 and angiogenesis or relapse free survival. Others have similarly reported that higher levels of FGF-2 are associated with improved overall and disease-free survival (Yiangou et al., 1997). In vitro, forced expression of FGF-2 promotes differentiation of T47D breast cancer cells (Korah et al., 2000). Similarly, in MDA-MB-134 breast carcinoma cells, which overexpress FGF receptors, FGF ligands are growth inhibitory (McLeskey et al., 1994). Taken together, these data suggest that, contrary to its role in promoting transformation in cells of mesodermal and neuroectodermal origin, in breast epithelial cells FGF-2 appears to promote a more differentiated phenotype.
Entity name
Prostate cancer
Oncogenesis
Giri et al. (1999) found that FGF-2 is significantly increased in prostate cancer relative to the normal prostate tissue. Using the transgenic adenocarcinoma of the mouse prostate (TRAMP) model system Polnaszek et al. (2003) demonstrated that hemi- or homozygous inactivation of the FGF-2 allele was associated with increased survival, decreased metastasis, and inhibition of progression to the poorly differentiated phenotype in primary prostatic tumors. These findings suggest that prostatic FGF2 activity may promote tumor progression and support the hypothesis that FGF2 plays a significant role in prostate cancer progression in vivo.
Entity name
Colorectal cancer
Prognosis
Elevated FGF-2 expression is associated with poor prognosis.
Oncogenesis
The evidence in the literature is somewhat contradictory regarding the expression of FGF-2 in colorectal cancers. However, the consensus is that FGF-2 expression is associated with poor prognosis. Tabara et al. (2001) reported that elevated FGF-2 expression in colorectal tumors correlates to tumor microvessel density and tumor size. In contrast, Landriscina et al. (1998) and Mathonnet et al. (2006) reported that FGF-2 levels are significantly reduced in colorectal tumors compared to adjacent non-tumor tissue. However, they speculated that this may be due to increased FGF-2 secretion leading to reduced tumor levels compared to normal tissues. Consistent with this model, George et al. (2002) reported that patients with colorectal cancer had elevated FGF-2 levels in their serum and plasma compared to cancer-free controls. The highest levels of serum FGF-2 were detected in patients with tumor metastasis and this was associated with reduced patient survival whereas patients with the lowest levels of serum FGF-2 had the greatest disease free survival 1-year post-treatment. These findings demonstrate that FGF-2 expression levels in colorectal cancer are correlated with cancer progression, metastasis, prognosis and patient survival.
Entity name
Ewing sarcoma family of tumors
Oncogenesis
Ewing tumors are related by their neural crest origin and primitive neural characteristics. The group includes Ewing tumor of bone (ETB or Ewing sarcoma of bone), extraosseous Ewing (EOE) tumors, primitive neuroectodermal tumors (PNET or peripheral neuroepithelioma), and Askin tumors (PNET of the chest wall). FGF-2 is a critical signaling molecule in primitive neural crest cells and its expression has been implicated in the pathogenesis of Ewing Tumors. Kim et al. (2004) reported that JK-GMS and SK-N-MC Ewing tumor cells lines underwent neuronal differentiation when treated with FGF-2, which was associated with inhibition of growth and induction of apoptosis. The effects on growth and differentiation are mediated via ERK1/ERK2 pathways while activation of JNK pathways and down-regulation of Blc-2 promote apoptosis. Evidence from other neuronal tumors indicates growth factor induces differentiation and apoptosis increases the efficacy of chemotherapy and radiation therapy.
Entity name
Hepatocellular carcinoma
Prognosis
Elevated FGF-2 serum levels is associated with poor prognosis.
Oncogenesis
Hsu et al. (1997), Poon et al. (2001) and more recently Uematsu et al. (2005) reported that FGF-2 serum levels are significantly elevated in patients with hepatocellular carcinoma (HCC) compared to cancer-free individuals. Elevated FGF-2 serum in these patients was associated with increased tumor size, vascularization, aggressiveness, progression and metastasis, which correlated with poor prognosis. These findings can in part be explained by the observed effects of FGF-2 on HCC cell lines. Ogasawara et al. (1996) reported that exogenous FGF-2 treatment stimulated HCC proliferation and treatment with an FGF-2 neutralizing antibody inhibited HCC proliferation. Furthermore, Maret et al. (1995) reported that decreased FGF-2 expression following transfection with an FGF-2 antisense molecule was associated with loss of anchorage independent growth and tumorigenicity. Collectively, these findings suggest a role for FGF-2 signaling in HCC growth and invasion.
Entity name
Ovarian cancer
Prognosis
Elevated cytoplasmic FGF-2 within ovarian tumors is associated with increased survival rates compared to patients with low levels of FGF-2.
Oncogenesis
Le Page et al. (2006) reported that FGF-2 levels are elevated in the serum of patients with ovarian cancer compared to cancer-free individuals and in tumor tissue compared to non-tumorous tissue. Secord et al. (2007) reported that high levels of cytoplasmic FGF-2 within ovarian tumors are associated with reduced tumor aggressiveness and increased survival rates compared to patients with low levels of FGF-2. However, Lin et al. (2003a; 2003b) and Zhang et al. (2003) reported that FGF-2 stimulates proliferation, migration, angiogenesis and invasion in ovarian cancer cell lines OVCA3 and SKOV3. They also report that treatment with an FGF-2 antibody can inhibit FGF-2 dependent proliferation and angiogenesis. Furthermore, Gan et al. (2006) reported that high FGF-2 tumor levels reduced drug sensitivity, in part due to the direct effects of FGF-2 on proliferation and apoptosis.
Entity name
Bladder carcinoma
Oncogenesis
Nguyen et al. (1994) and others have reported that FGF-2 levels are elevated in the urine of patient with bladder carcinoma compared to cancer-free individuals. The level of FGF-2 in the urine corresponds to disease stage; metastatic cancers have the highest levels followed by localized bladder carcinoma and patient with no evidence of disease following surgical resection have the lowest levels. Gazzaniga et al. (1999) reported that FGF-2 levels were also elevated in serum of patients with bladder carcinoma. Furthermore, high levels of FGF-2 in urine and serum were associated with early disease recurrence, which was suggested to be in part due to increases in Bcl-2 expression associated with elevated FGF-2 levels. Miyake et al. (1997, 1998) reported that transfection of the human FGF-2 gene into the bladder cancer cell lines HT1376 and KoTCC-1 resulted in increased expression of the matrix metalloproteinases MMP-2 and MMP-9, and this was associated with an increase in the in vitro invasiveness of the cells. Additionally, transfected cells exhibited increased resistance to the chemotheraphy drug cisplatin. Chikazawa et al. (2008) implanted clones of the human bladder cancer cell line 253JB-V that expressed either high or low levels of FGF-2 in athymic nude mice and reported that clones with high FGF-2 expression demonstrated increased tumorigenicity and metastasis compared to clones expressing low levels of FGF-2. Collectively, these findings indicate that FGF-2 levels in tumors, urine and serum provide useful information pertaining to disease progression, invasiveness, metastasis, recurrence and prognosis.
Entity name
Gastric carcinoma
Prognosis
High FGF-2 tumor expression is associated with poor prognosis.
Oncogenesis
Bilgic et al. (2009) reported that patients with gastric carcinoma have significantly higher levels of FGF-2 in their serum compared to cancer-free patients. Ueki et al. (1995) and Zhao et al. (2005) reported that gastric carcinomas expressing high levels of FGF-2 were larger in size, more invasive and had a higher rate of metastasis than carcinomas with low FGF-2 expression. Elevated FGF-2 in combination with MMP-9 expression was correlated with increased invasiveness, growth, tumor angiogenesis, metastasis and poor prognosis.
Entity name
Kaposis sarcoma
Oncogenesis
Kaposis sarcoma is a classification of tumors most commonly found in skin caused by the Human Herpesvirus 8 (HHV8). Ascherl et al. (2001) reported that patients with Kaposis sarcoma have elevated serum levels of FGF-2 compared to disease-free patients and higher serum FGF-2 levels were associated with reduced survival rates. Studies with Kaposis sarcoma model cell lines reported that FGF-2 promoted growth, angiogenesis and cell transformation, suggesting contribution to tumor growth, progression, invasion and metastasis.
Entity name
Mental disorders: depression, bipolar disorder and schizophrenia
Disease
Depression describes extreme feelings of sadness, worthlessness, hopelessness that last for an extended period of time. Bipolar disorder refers to the medical condition that consists of cycling between extreme moods of depression and mania (elevated, happy). Schizophrenia is a mental disorder characterized by altered perception of reality. Those inflicted by the disorder suffer from sever delusions, hallucinations, paranoia and disorganized speech and thinking.
Deficiencies in FGF2 expression within the frontal cortex and hippocampus have been associated with depression, bipolar disorder and schizophrenia. These disorders are commonly treated with selective serotonin reuptake inhibitors that increase FGF2 expression independent of increases in extracellular serotonin levels.
Deficiencies in FGF2 expression within the frontal cortex and hippocampus have been associated with depression, bipolar disorder and schizophrenia. These disorders are commonly treated with selective serotonin reuptake inhibitors that increase FGF2 expression independent of increases in extracellular serotonin levels.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 12731865 | 2003 | Analysis of the leukemia inhibitory factor receptor functional domains by chimeric receptors and cytokines. | Aasland D et al |
| 3780670 | 1986 | Human basic fibroblast growth factor: nucleotide sequence and genomic organization. | Abraham JA et al |
| 8387074 | 1993 | Expression of basic fibroblast growth factor and its receptor in an invasive bladder carcinoma cell line. | Allen LE et al |
| 9858574 | 1999 | A new 34-kilodalton isoform of human fibroblast growth factor 2 is cap dependently synthesized by using a non-AUG start codon and behaves as a survival factor. | Arnaud E et al |
| 14753849 | 2003 | Expression of fibroblast growth factor receptor genes in human hepatoma-derived cell lines. | Asada N et al |
| 11485620 | 2001 | Serum concentrations of fibroblast growth factor 2 are increased in HIV type 1-infected patients and inversely related to survival probability. | Ascherl G et al |
| 17306451 | 2007 | The fibroblast growth factor-2 antisense gene inhibits nuclear accumulation of FGF-2 and delays cell cycle progression in C6 glioma cells. | Baguma-Nibasheka M et al |
| 10657989 | 2000 | Uncoupling of cell proliferation and differentiation activities of basic fibroblast growth factor. | Bailly K et al |
| 2328724 | 1990 | Translocation of bFGF to the nucleus is G1 phase cell cycle specific in bovine aortic endothelial cells. | Baldin V et al |
| 16278388 | 2005 | Basic fibroblast growth factor (FGF-2) overexpression is a risk factor for esophageal cancer recurrence and reduced survival, which is ameliorated by coexpression of the FGF-2 antisense gene. | Barclay C et al |
| 2731576 | 1989 | Autocrine regulation of bovine retinal capillary endothelial cell (BREC) proliferation by BREC-derived basic fibroblast growth factor. | Bensaid M et al |
| 7577000 | 1995 | Significance of angiogenesis in tumour progression and metastasis. | Bikfalvi A et al |
| 18447253 | 2008 | Serum bFGF concentrations in gastric cancer patients. | Bilgic I et al |
| 12859421 | 2003 | Impact of extracellular matrix and strain on proliferation of bovine airway smooth muscle. | Bonacci JV et al |
| 15525641 | 2005 | Heterogeneous nuclear ribonucleoprotein A1 is a novel internal ribosome entry site trans-acting factor that modulates alternative initiation of translation of the fibroblast growth factor 2 mRNA. | Bonnal S et al |
| 12857733 | 2003 | A single internal ribosome entry site containing a G quartet RNA structure drives fibroblast growth factor 2 gene expression at four alternative translation initiation codons. | Bonnal S et al |
| 8798749 | 1996 | Fibroblast growth factor-2 binds to the regulatory beta subunit of CK2 and directly stimulates CK2 activity toward nucleolin. | Bonnet H et al |
| 12717444 | 2003 | Translokin is an intracellular mediator of FGF-2 trafficking. | Bossard C et al |
| 7735329 | 1994 | Activation of rDNA transcription by FGF-2: key role of protein kinase CKII. | Bouche G et al |
| 2871488 | 1986 | Parathyroid mitogenic activity in plasma from patients with familial multiple endocrine neoplasia type 1. | Brandi ML et al |
| 19086919 | 2009 | Fibroblast growth factor 2 (FGF-2) is a novel substrate for arginine methylation by PRMT5. | Bruns AF et al |
| 1986249 | 1991 | Alternative initiation of translation determines cytoplasmic or nuclear localization of basic fibroblast growth factor. | Bugler B et al |
| 1713785 | 1991 | Direct evidence for methylation of arginine residues in high molecular weight forms of basic fibroblast growth factor. | Burgess WH et al |
| 8768072 | 1996 | High-mobility-group chromosomal proteins: architectural components that facilitate chromatin function. | Bustin M et al |
| 18392794 | 2008 | R Regulation of tumor angiogenesis and metastasis by FGF and PDGF signaling pathways. | Cao Y et al |
| 11409912 | 2000 | Exogenous fibroblast growth factor-2 induces a transformed phenotype in vascular kaposi's sarcoma-like cells. | Cavallaro U et al |
| 19096229 | 2008 | Expression of angiogenesis-related genes regulates different steps in the process of tumor growth and metastasis in human urothelial cell carcinoma of the urinary bladder. | Chikazawa M et al |
| 18850066 | 2009 | High molecular weight FGF2: the biology of a nuclear growth factor. | Chlebova K et al |
| 1660310 | 1991 | Potential oncogenic effects of basic fibroblast growth factor requires cooperation between CUG and AUG-initiated forms. | Couderc B et al |
| 11990524 | 2002 | Basic fibroblast growth factor infusion increases tumour vascularity, blood flow and chemotherapy uptake. | Davies MM et al |
| 12209881 | 2002 | Overexpression of the 18 kDa and 22/24 kDa FGF-2 isoforms results in differential drug resistance and amplification potential. | Dini G et al |
| 9572496 | 1998 | A GR-motif functions in nuclear accumulation of the large FGF-2 isoforms and interferes with mitogenic signalling. | Dono R et al |
| 9687490 | 1998 | Impaired cerebral cortex development and blood pressure regulation in FGF-2-deficient mice. | Dono R et al |
| 19544431 | 2009 | A complex role for FGF-2 in self-renewal, survival, and adhesion of human embryonic stem cells. | Eiselleova L et al |
| 10699930 | 2000 | FGF-2 isoforms of 18 and 22.5 kDa differentially modulate t-PA and PAI-1 expressions on the pancreatic carcinoma cells AR4-2J: consequences on cell spreading and invasion. | Escaffit F et al |
| 7679948 | 1993 | Morphological and biological modifications induced in a rat pancreatic acinar cancer cell line (AR4-2J) by unscheduled expression of basic fibroblast growth factors. | Estival A et al |
| 15483108 | 2004 | Dysregulation of the fibroblast growth factor system in major depression. | Evans SJ et al |
| 1785797 | 1991 | Basic fibroblast growth factor gene expression. | Florkiewicz RZ et al |
| 1933286 | 1991 | Localization of basic fibroblast growth factor and its mRNA after CNS injury. | Frautschy SA et al |
| 12784342 | 2003 | Nuclear accumulation of basic fibroblast growth factor in human astrocytic tumors. | Fukui S et al |
| 11313915 | 2001 | Tumour suppressor p53 inhibits human fibroblast growth factor 2 expression by a post-transcriptional mechanism. | Galy B et al |
| 9892202 | 1999 | Cell transformation results in the loss of the density-dependent translational regulation of the expression of fibroblast growth factor 2 isoforms. | Galy B et al |
| 16741658 | 2006 | Expression of basic fibroblast growth factor correlates with resistance to paclitaxel in human patient tumors. | Gan Y et al |
| 8559302 | 1995 | The potential role of basic fibroblast growth factor in the transformation of cultured primary human fetal astrocytes and the proliferation of human glioma (U-87) cells. | Gately S et al |
| 16861106 | 2006 | Hippocampal FGF-2 and FGFR1 mRNA expression in major depression, schizophrenia and bipolar disorder. | Gaughran F et al |
| 1898777 | 1991 | Interleukin 1 regulates heparin-binding growth factor 2 gene expression in vascular smooth muscle cells. | Gay CG et al |
| 10339668 | 1999 | Detection of basic fibroblast growth factor mRNA in urinary bladder cancer: correlation with local relapses. | Gazzaniga P et al |
| 12553380 | 2002 | Plasma basic fibroblast growth factor levels in colorectal cancer: a clinically useful assay? | George ML et al |
| 10745274 | 2000 | E1A stimulates FGF-2 release promoting differentiation of primary endothelial cells. | Giampietri C et al |
| 10353739 | 1999 | Alterations in expression of basic fibroblast growth factor (FGF) 2 and its receptor FGFR-1 in human prostate cancer. | Giri D et al |
| 9710742 | 1998 | A possible role of multidrug resistance-associated protein (MRP) in basic fibroblast growth factor secretion by AIDS-associated Kaposi's sarcoma cells: a survival molecule? | Gupta S et al |
| 18040697 | 2008 | Temporal and regional morphological differences as a consequence of FGF-2 deficiency are mirrored in the myenteric proteome. | Hagl CI et al |
| 12841351 | 2003 | High levels of CUG-initiated FGF-2 expression cause chromatin compaction, decreased cardiomyocyte mitosis, and cell death. | Hirst CJ et al |
| 16080018 | 2005 | Correlation of bFGF expression in renal cell cancer with clinical and histopathological features by tissue microarray analysis and measurement of serum levels. | Horstmann M et al |
| 11002286 | 2000 | Fibroblast growth factors (FGF-1, FGF-2) promote migration and neurite growth of mouse cochlear ganglion cells in vitro: immunohistochemistry and antibody perturbation. | Hossain WA et al |
| 9252719 | 1997 | Implications of serum basic fibroblast growth factor levels in chronic liver diseases and hepatocellular carcinoma. | Hsu PI et al |
| 7511892 | 1994 | Expression of fibroblast growth factor gene family and its receptor gene family in the human upper gastrointestinal tract. | Iida S et al |
| 15460907 | 2004 | Correlation of vascular endothelial cell proliferation with microvessel density and expression of vascular endothelial growth factor and basic fibroblast growth factor in hepatocellular carcinoma. | Imura S et al |
| 17076636 | 2006 | Controlled releases of FGF-2 and paclitaxel from chitosan hydrogels and their subsequent effects on wound repair, angiogenesis, and tumor growth. | Ishihara M et al |
| 15023562 | 2004 | Non-angiogenic FGF-2 protects the ischemic heart from injury, in the presence or absence of reperfusion. | Jiang ZS et al |
| 9010219 | 1997 | Nuclear accumulation of FGF-2 is associated with proliferation of human astrocytes and glioma cells. | Joy A et al |
| 8570185 | 1995 | Translational enhancement of FGF-2 by eIF-4 factors, and alternate utilization of CUG and AUG codons for translation initiation. | Kevil C et al |
| 14694527 | 2004 | Fibroblast growth factor 2 induces differentiation and apoptosis of Askin tumour cells. | Kim MS et al |
| 10521360 | 1999 | Autocrine FGF-2 is responsible for the cell density-dependent susceptibility to apoptosis of HUVEC : A role of a calpain inhibitor-sensitive mechanism. | Kinoshita M et al |
| 15380477 | 2004 | Enlarged infarct volume and loss of BDNF mRNA induction following brain ischemia in mice lacking FGF-2. | Kiprianova I et al |
| 10652299 | 2000 | Biochemical analysis of the arginine methylation of high molecular weight fibroblast growth factor-2. | Klein S et al |
| 7999082 | 1994 | Basic fibroblast growth factor sense (FGF) and antisense (gfg) RNA transcripts are expressed in unfertilized human oocytes and in differentiated adult tissues. | Knee RS et al |
| 15538042 | 2004 | Expression of FGF-2 alters focal adhesion dynamics in migration-restricted MDA-MB-231 breast cancer cells. | Korah R et al |
| 11027671 | 2000 | Intracellular FGF-2 promotes differentiation in T-47D breast cancer cells. | Korah RM et al |
| 9743297 | 1998 | Quantitative analysis of basic fibroblast growth factor and vascular endothelial growth factor in human colorectal cancer. | Landriscina M et al |
| 16217764 | 2006 | From gene profiling to diagnostic markers: IL-18 and FGF-2 complement CA125 as serum-based markers in epithelial ovarian cancer. | Le Page C et al |
| 9016566 | 1997 | A common RNA structural motif involved in the internal initiation of translation of cellular mRNAs. | Le SY et al |
| 18930044 | 2008 | Overexpression of high molecular weight FGF-2 forms inhibits glioma growth by acting on cell-cycle progression and protein translation. | Lemiere S et al |
| 3756876 | 1986 | Suppression of glioma growth in vitro and in vivo by glia maturation factor. | Lim R et al |
| 19303400 | 2009 | FAK mediates the inhibition of glioma cell migration by truncated 24 kDa FGF-2. | Lin AH et al |
| 14669225 | 2003 | [The effect of basic fibroblast growth factor in ovarian cancer growth and angiogenesis]. | Lin W et al |
| 12579092 | 2003 | Vascular endothelial growth factor and basic fibroblast growth factor expression in esophageal adenocarcinoma and Barrett esophagus. | Lord RV et al |
| 9088738 | 1997 | Expression of FGF-1 and FGF-2 mRNA during lens morphogenesis, differentiation and growth. | Lovicu FJ et al |
| 1380281 | 1992 | Expression of basic fibroblast growth factor, FGFR1 and FGFR2 in normal and malignant human breast, and comparison with other normal tissues. | Luqmani YA et al |
| 17503459 | 2007 | Chromatin compaction and cell death by high molecular weight FGF-2 depend on its nuclear localization, intracrine ERK activation, and engagement of mitochondria. | Ma X et al |
| 10573108 | 1999 | Overexpression of basic fibroblast growth factor (FGF-2) downregulates Bcl-2 and promotes apoptosis in MCF-7 human breast cancer cells. | Maloof P et al |
| 7585554 | 1995 | Inhibition of fibroblast growth factor 2 expression by antisense RNA induced a loss of the transformed phenotype in a human hepatoma cell line. | Maret A et al |
| 16773701 | 2006 | Quantitative analysis using ELISA of vascular endothelial growth factor and basic fibroblast growth factor in human colorectal cancer, liver metastasis of colorectal cancer and hepatocellular carcinoma. | Mathonnet M et al |
| 11781339 | 2002 | p38 MAP kinase negatively regulates endothelial cell survival, proliferation, and differentiation in FGF-2-stimulated angiogenesis. | Matsumoto T et al |
| 7506125 | 1994 | MDA-MB-134 breast carcinoma cells overexpress fibroblast growth factor (FGF) receptors and are growth-inhibited by FGF ligands. | McLeskey SW et al |
| 9105031 | 1997 | Loss, restoration, and maintenance of plasma membrane integrity. | McNeil PL et al |
| 8562930 | 1996 | Elevated intracellular level of basic fibroblast growth factor correlates with stage of chronic lymphocytic leukemia and is associated with resistance to fludarabine. | Menzel T et al |
| 15145049 | 2004 | Cap-dependent and cap-independent translation in eukaryotic systems. | Merrick WC et al |
| 9489477 | 1998 | Expression of basic fibroblast growth factor is associated with resistance to cisplatin in a human bladder cancer cell line. | Miyake H et al |
| 9146669 | 1997 | Basic fibroblast growth factor regulates matrix metalloproteinases production and in vitro invasiveness in human bladder cancer cell lines. | Miyake H et al |
| 9693381 | 1998 | Transcriptional regulation of fibroblast growth factor-2 expression in human astrocytes: implications for cell plasticity. | Moffett J et al |
| 16397244 | 2006 | Protease-activated receptor-2 is essential for factor VIIa and Xa-induced signaling, migration, and invasion of breast cancer cells. | Morris DR et al |
| 2710130 | 1989 | Elevated expression of basic fibroblast growth factor messenger ribonucleic acid in acoustic neuromas. | Murphy PR et al |
| 1323055 | 1992 | Phosphorothioate antisense oligonucleotides against basic fibroblast growth factor inhibit anchorage-dependent and anchorage-independent growth of a malignant glioblastoma cell line. | Murphy PR et al |
| 7508518 | 1994 | Elevated levels of an angiogenic peptide, basic fibroblast growth factor, in the urine of patients with a wide spectrum of cancers. | Nguyen M et al |
| 1811792 | 1991 | Bibliographic update: fibroblast growth factor. | |
| 8988054 | 1997 | Two mechanisms of basic fibroblast growth factor-induced angiogenesis in bladder cancer. | O'Brien T et al |
| 9751258 | 1998 | Influence of intratumoral basic fibroblast growth factor concentration on survival in ovarian cancer patients. | Obermair A et al |
| 8707262 | 1996 | Expressions of basic fibroblast growth factor and its receptors and their relationship to proliferation of human hepatocellular carcinoma cell lines. | Ogasawara S et al |
| 10597279 | 1999 | Nuclear 24 kD fibroblast growth factor (FGF)-2 confers metastatic properties on rat bladder carcinoma cells. | Okada-Ban M et al |
| 1655776 | 1991 | Endogenous basic fibroblast growth factor-dependent induction of collagenase and interleukin-6 in tumor necrosis factor-treated human microvascular endothelial cells. | Okamura K et al |
| 9576942 | 1998 | Neuronal defects and delayed wound healing in mice lacking fibroblast growth factor 2. | Ortega S et al |
| 11815602 | 2002 | Fibroblast growth factor-2 induces translational regulation of Bcl-XL and Bcl-2 via a MEK-dependent pathway: correlation with resistance to etoposide-induced apoptosis. | Pardo OE et al |
| 16810323 | 2006 | FGF-2 protects small cell lung cancer cells from apoptosis through a complex involving PKCepsilon, B-Raf and S6K2. | Pardo OE et al |
| 8119973 | 1994 | Vascular endothelial growth factor is induced in response to transforming growth factor-beta in fibroblastic and epithelial cells. | Pertovaara L et al |
| 11919185 | 2002 | A ligand-inducible epidermal growth factor receptor/anaplastic lymphoma kinase chimera promotes mitogenesis and transforming properties in 3T3 cells. | Piccinini G et al |
| 15962299 | 2005 | PDGF-BB induces vascular smooth muscle cell expression of high molecular weight FGF-2, which accumulates in the nucleus. | Pintucci G et al |
| 10048578 | 1999 | Dual activities of 22-24 kDA basic fibroblast growth factor: inhibition of migration and stimulation of proliferation. | Piotrowicz RS et al |
| 14522896 | 2003 | Fibroblast growth factor 2 promotes tumor progression in an autochthonous mouse model of prostate cancer. | Polnaszek N et al |
| 11587697 | 2001 | Correlation of serum basic fibroblast growth factor levels with clinicopathologic features and postoperative recurrence in hepatocellular carcinoma. | Poon RT et al |
| 1406661 | 1992 | cis-acting elements involved in the alternative translation initiation process of human basic fibroblast growth factor mRNA. | Prats AC et al |
| 2538817 | 1989 | High molecular mass forms of basic fibroblast growth factor are initiated by alternative CUG codons. | Prats H et al |
| 16023796 | 2005 | Gene profiling of cells expressing different FGF-2 forms. | Quarto N et al |
| 1660309 | 1991 | Selective expression of high molecular weight basic fibroblast growth factor confers a unique phenotype to NIH 3T3 cells. | Quarto N et al |
| 2195042 | 1990 | Nuclear and cytoplasmic localization of different basic fibroblast growth factor species. | Renko M et al |
| 15650239 | 2004 | Angiogenesis in neuroblastoma. | Ribatti D et al |
| 11747364 | 2001 | Intracellular signaling pathway of FGF-2-modulated corneal endothelial cell migration during wound healing in vitro. | Rieck PW et al |
| 7999354 | 1994 | Studies on FGF-2: nuclear localization and function of high molecular weight forms and receptor binding in the absence of heparin. | Rifkin DB et al |
| 16365024 | 2005 | Nipple fluid basic fibroblast growth factor in patients with breast cancer. | Sartippour MR et al |
| 17481705 | 2007 | Co-expression of angiogenic markers and associations with prognosis in advanced epithelial ovarian cancer: a Gynecologic Oncology Group study. | Secord AA et al |
| 20299037 | 2010 | Association of angiogenesis related markers with bladder cancer outcomes and other molecular markers. | Shariat SF et al |
| 16227592 | 2005 | Direct regulation of rRNA transcription by fibroblast growth factor 2. | Sheng Z et al |
| 1764264 | 1991 | Functional characterization of the human basic fibroblast growth factor gene promoter. | Shibata F et al |
| 10442194 | 1999 | Upregulation of basic fibroblast growth factor in breast carcinoma and its relationship to vascular density, oestrogen receptor, epidermal growth factor receptor and survival. | Smith K et al |
| 7893598 | 1994 | Spatial and temporal expression of basic fibroblast growth factor (FGF-2) mRNA and protein in early Xenopus development. | Song J et al |
| 16615083 | 2006 | Functional diversity of FGF-2 isoforms by intracellular sorting. | Sørensen V et al |
| 15879597 | 2005 | Exogenously added fibroblast growth factor 2 (FGF-2) to NIH3T3 cells interacts with nuclear ribosomal S6 kinase 2 (RSK2) in a cell cycle-dependent manner. | Soulet F et al |
| 15731686 | 2005 | FGF-2 acts through an ERK1/2 intracellular pathway to affect osteoblast differentiation. | Spector JA et al |
| 7929563 | 1994 | Regulation of bFGF gene expression and subcellular distribution of bFGF protein in adrenal medullary cells. | Stachowiak MK et al |
| 11085540 | 2000 | Induction of cell death by basic fibroblast growth factor in Ewing's sarcoma. | Sturla LM et al |
| 11669335 | 2001 | Concurrent expression of angiogenic growth factors and neovascularization during tumourigenesis in colorectal carcinoma patients. | Tabara H et al |
| 1564542 | 1992 | Correlation of basic fibroblast growth factor expression levels with the degree of malignancy and vascularity in human gliomas. | Takahashi JA et al |
| 14523006 | 2003 | Shedding of membrane vesicles mediates fibroblast growth factor-2 release from cells. | Taverna S et al |
| 15122340 | 2004 | Nuclear FGF-2 facilitates cell survival in vitro and during establishment of metastases. | Thomas-Mudge RJ et al |
| 10409702 | 1999 | Expression of human fibroblast growth factor 2 mRNA is post-transcriptionally controlled by a unique destabilizing element present in the 3'-untranslated region between alternative polyadenylation sites. | Touriol C et al |
| 10858460 | 2000 | Alternative translation initiation of human fibroblast growth factor 2 mRNA controlled by its 3'-untranslated region involves a Poly(A) switch and a translational enhancer. | Touriol C et al |
| 8090761 | 1994 | Transcriptional regulation of basic fibroblast growth factor gene by p53 in human glioblastoma and hepatocellular carcinoma cells. | Ueba T et al |
| 8568589 | 1995 | Expression of basic fibroblast growth factor and fibroblast growth factor receptor in advanced gastric carcinoma. | Ueki T et al |
| 15836707 | 2005 | Altered expression of vascular endothelial growth factor, fibroblast growth factor-2 and endostatin in patients with hepatocellular carcinoma. | Uematsu S et al |
| 7799942 | 1995 | Alternative translation of human fibroblast growth factor 2 mRNA occurs by internal entry of ribosomes. | Vagner S et al |
| 8947560 | 1996 | Translation of CUG- but not AUG-initiated forms of human fibroblast growth factor 2 is activated in transformed and stressed cells. | Vagner S et al |
| 11075807 | 2000 | FIF [fibroblast growth factor-2 (FGF-2)-interacting-factor], a nuclear putatively antiapoptotic factor, interacts specifically with FGF-2. | Van den Berghe L et al |
| 8397111 | 1993 | Phosphorylation of basic fibroblast growth factor (FGF-2) in the nuclei of SK-Hep-1 cells. | Vilgrain I et al |
| 15645710 | 2004 | Signaling, internalization, and intracellular activity of fibroblast growth factor. | Wiedłocha A et al |
| 9293890 | 1997 | Expression of basic fibroblast growth factor (FGF-2)-associated with tumour proliferation in human pancreatic carcinoma. | Yamazaki K et al |
| 9000594 | 1997 | Fibroblast growth factor 2 in breast cancer: occurrence and prognostic significance. | Yiangou C et al |
| 17968311 | 2008 | Thrombin cleaves the high molecular weight forms of basic fibroblast growth factor (FGF-2): a novel mechanism for the control of FGF-2 and thrombin activity. | Yu PJ et al |
| 2171762 | 1990 | Immunohistochemical localization of basic fibroblast growth factor in astrocytomas. | Zagzag D et al |
| 19533745 | 2009 | Expression of hepatocyte growth factor and basic fibroblast growth factor as prognostic indicators in gastric cancer. | Zhang W et al |
| 14613644 | 2003 | [Expression of aFGF and bFGF in ovarian cancer and their effect on ovarian cancer cell proliferation]. | Zhang Y et al |
| 19616043 | 2009 | Basic fibroblast growth factor upregulates adrenomedullin expression in ovarian epithelial carcinoma cells via JNK-AP-1 pathway. | Zhang Y et al |
| 12620228 | 2003 | ERK-dependent phosphorylation of the transcription initiation factor TIF-IA is required for RNA polymerase I transcription and cell growth. | Zhao J et al |
| 15929172 | 2005 | Correlative studies on bFGF mRNA and MMP-9 mRNA expressions with microvascular density, progression, and prognosis of gastric carcinomas. | Zhao ZS et al |
| 8098714 | 1993 | Increased basic fibroblast growth factor in plasma from multiple endocrine neoplasia type 1: relation to pituitary tumor. | Zimering MB et al |
Other Information
Locus ID:
NCBI: 2247
MIM: 134920
HGNC: 3676
Ensembl: ENSG00000138685
Variants:
dbSNP: 2247
ClinVar: 2247
TCGA: ENSG00000138685
COSMIC: FGF2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA134941638 | FGFR1OP | Gene | Pathway | associated | 20124951 | ||
| PA134972108 | FGFR1OP2 | Gene | Pathway | associated | 20124951 | ||
| PA162363968 | lenalidomide | Chemical | ClinicalAnnotation | associated | PD | 28373444 | |
| PA28127 | FGFR1 | Gene | Pathway | associated | 20124951 | ||
| PA28128 | FGFR2 | Gene | Pathway | associated | 20124951 | ||
| PA28129 | FGFR3 | Gene | Pathway | associated | 20124951 | ||
| PA28130 | FGFR4 | Gene | Pathway | associated | 20124951 | ||
| PA28131 | FGFR3P1 | Gene | Pathway | associated | 20124951 | ||
| PA28132 | FGFRL1 | Gene | Pathway | associated | 20124951 | ||
| PA444985 | Multiple Myeloma | Disease | ClinicalAnnotation | associated | PD | 28373444 | |
| PA447207 | Depressive Disorder | Disease | ClinicalAnnotation | associated | PD | ||
| PA449690 | fluvoxamine | Chemical | ClinicalAnnotation | associated | PD | ||
| PA451644 | thalidomide | Chemical | ClinicalAnnotation | associated | PD | 28373444 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37165492 | 2024 | miR-129-5p/FGF2 Axis is Associated with Homocysteine-induced Human Umbilical Vein Endothelial Cell Injury. | 0 |
| 37848720 | 2024 | Angiogenesis related genes in Takayasu Arteritis (TAK): robust association with Tag SNPs of IL-18 and FGF-2 in a South Asian Cohort. | 0 |
| 37988847 | 2024 | Trimethylamine-N-oxide (TMAO) and basic fibroblast growth factor (bFGF) are possibly involved in corticosteroid resistance in adult patients with immune thrombocytopenia. | 0 |
| 38247274 | 2024 | Assessment of serum levels and placental bed tissue expression of IGF-1, bFGF, and PLGF in patients with placenta previa complicated with placenta accreta spectrum disorders. | 0 |
| 38253356 | 2024 | The optimized priming effect of FGF-1 and FGF-2 enhances preadipocyte lineage commitment in human adipose-derived mesenchymal stem cells. | 0 |
| 38355415 | 2024 | Serum fibroblast growth factor-2 levels complement vital biomarkers for diagnosing heart failure. | 0 |
| 38479160 | 2024 | Cancer-associated fibroblasts promote gastric cancer cell proliferation by paracrine FGF2-driven ribosome biogenesis. | 2 |
| 38518781 | 2024 | Direct programming of human pluripotent stem cells into endothelial progenitors with SOX17 and FGF2. | 0 |
| 38811382 | 2024 | Knockdown the moyamoya disease susceptibility gene, RNF213, upregulates the expression of basic fibroblast growth factor and matrix metalloproteinase-9 in bone marrow derived mesenchymal stem cells. | 0 |
| 37165492 | 2024 | miR-129-5p/FGF2 Axis is Associated with Homocysteine-induced Human Umbilical Vein Endothelial Cell Injury. | 0 |
| 37848720 | 2024 | Angiogenesis related genes in Takayasu Arteritis (TAK): robust association with Tag SNPs of IL-18 and FGF-2 in a South Asian Cohort. | 0 |
| 37988847 | 2024 | Trimethylamine-N-oxide (TMAO) and basic fibroblast growth factor (bFGF) are possibly involved in corticosteroid resistance in adult patients with immune thrombocytopenia. | 0 |
| 38247274 | 2024 | Assessment of serum levels and placental bed tissue expression of IGF-1, bFGF, and PLGF in patients with placenta previa complicated with placenta accreta spectrum disorders. | 0 |
| 38253356 | 2024 | The optimized priming effect of FGF-1 and FGF-2 enhances preadipocyte lineage commitment in human adipose-derived mesenchymal stem cells. | 0 |
| 38355415 | 2024 | Serum fibroblast growth factor-2 levels complement vital biomarkers for diagnosing heart failure. | 0 |
Citation
Leigh-Ann MacFarlane ; Paul Murphy
FGF2 (fibroblast growth factor 2 (basic))
Atlas Genet Cytogenet Oncol Haematol. 2010-05-01
Online version: http://atlasgeneticsoncology.org/gene/511/fgf2-(fibroblast-growth-factor-2-(basic))
