FOXO1 (Forkhead box O1)
2008-05-01 Roddy OConnor , Frederic G Barr AffiliationDepartment of Pathology, Laboratory Medicine, University of Pennsylvania School of Medicine, Philadelphia, PA 19104, USA
Identity
HGNC
LOCATION
13q14.11
LOCUSID
ALIAS
FKH1,FKHR,FOXO1A
FUSION GENES
DNA/RNA
Description
The FOXO1 gene extends approximately 110 kb and consists of 3 exons.
Transcription
Only a single transcript has been reported to be expressed from the FOXO1 gene, measuring 5.7 kb in length and containing an open reading frame of 1965 bp. At the RNA level, the gene is widely expressed.
Proteins
Description
A single protein of 655 amino acids is expressed. This protein is a transcription factor with a forkhead box-containing DNA binding domain in the N-terminal region and an acidic, serine/threonine-rich transcriptional activation domain in the C-terminal region.
Expression
The protein is widely expressed. Covalent attachment of ubiquitin moieties (polyubiquitination) targets FOXO1 protein for degradation, and thus FOXO1 expression can be regulated by the ubiquitin-dependent proteasome. AKT is implicated in the regulation of FOXO1 expression by its enhancement of FOXO1 ubiquitination and proteolysis.
Localisation
The FOXO1 protein shuttles between the nucleus and cytoplasm. The subcellular localization and hence the transcriptional activity of FOXO1 is regulated by intracellular kinases. FOXO1 contains three AKT-phosphorylation motifs [RxRxx(S/T)]. Phosphorylation of these sites by AKT promotes nuclear exclusion, association with 14-3-3 adaptor proteins and cytosolic retention. The serum and glucocorticoid-inducible kinase ( SGK ), dual-specificity tyrosine-phosphorylated regulated kinase DYRK1A and cyclin-dependent kinase 2 ( CDK2 ) may also similarly phosphorylate FOXO1 and contribute to its subceullar localization, thereby acting combinatorially to suppress FOXO1 transcriptional activity. Although the role of phosphatases in FOXO1 activation is unclear, PTEN may have a role in countering the effects of these kinases.
Function
FOXO1 plays an important role in many cellular processes. As a transcription factor, FOXO1 induces expression of target genes involved in apoptosis, glucose metabolism, cell cycle progression, and differentiation. There is increasing evidence of a role for FOXO1 as a tumor suppressor. FOXO1 transcriptional responses are also implicated in cellular protection following DNA damage and oxidative stress, which may be related to a role in longevity.
Homology
The first FOX transcription factor fork head was identified in Drosophila. The subsequent cloning of mammalian FOX transcription factors revealed a common DNA-binding domain (forkhead box) that is highly conserved across species including Drosophila melanogaster, C. elegans and Homo sapiens. Within the larger FOX transcription factor family, there is a subfamily of which FOXO1 is the prototype. Additional members of this FOXO subfamily are: FOXO3 (FKHRL1, FOXO2), FOXO4 (AFX), FOXO6.
A gene homologous to FOXO subfamily members, daf-16, has also been identified in C. elegans and has facilitated analysis of the functional regulation of mammalian FOXO subfamily members. FOXO members share consensus phosphorylaton motifs for multiple kinases including AKT, CDK2, DYRK1A and SGK. Phosphorylation of these motifs regulates subcellular localization, DNA affinity, and protein-protein interactions. Of note, three of the genes in the FOXO1 subfamily are involved in cancer associated-chromosomal translocations. In addition to the rearrangement of FOXO1 in alveolar rhabdomyosarcoma (discussed below), FOXO3 and FOXO4 are joined with the MLL gene by translocations in acute myeloid leukemias.
A gene homologous to FOXO subfamily members, daf-16, has also been identified in C. elegans and has facilitated analysis of the functional regulation of mammalian FOXO subfamily members. FOXO members share consensus phosphorylaton motifs for multiple kinases including AKT, CDK2, DYRK1A and SGK. Phosphorylation of these motifs regulates subcellular localization, DNA affinity, and protein-protein interactions. Of note, three of the genes in the FOXO1 subfamily are involved in cancer associated-chromosomal translocations. In addition to the rearrangement of FOXO1 in alveolar rhabdomyosarcoma (discussed below), FOXO3 and FOXO4 are joined with the MLL gene by translocations in acute myeloid leukemias.
Mutations
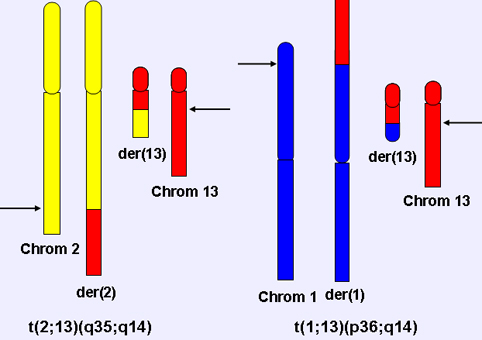
Figure 1. Diagram of t(2;13)(q35;q14) and t(1;13)(p36;q14) chromosomal translocations.
Germinal
Inherited mutations of FOXO1 have not been identified.
Somatic
The FOXO1 gene is rearranged by the recurrent acquired chromosomal translocations - t(2;13)(q35;q14) and t(1;13)(p36;q14) - in the myogenic soft tissue cancer alveolar rhabdomyosarcoma. As a result of the 2;13 or 1;13 translocation, portions of the PAX3 or PAX7 gene (on chromosomes 2 or 1, respectively) are juxtaposed with portions of the FOXO1 gene. In particular, the 5 region of PAX3 or PAX7, including the first seven exons of either gene, is joined to the 3 region of FOXO1, including its last two exons. Though the reciprocal chimeric gene is also generated, the PAX3-FOXO1 and PAX7-FOXO1 chimeric genes are more consistent and highly expressed, and result in expression of fusion proteins consisting of the intact PAX3 or PAX7 N-terminal DNA binding domain fused in-framed to the intact FOXO1 C-terminal transcriptional activation domain.
FOXO1 was identified as only gene within a minimal common region of deletion in chromosomal region 13q14 in prostate carcinoma. FOXO1 deletion was detected in about 30% of prostate cancer samples, and additional cases were identified with reduced expression without evidence of deletion. Based on functional testing in prostate cancer cell lines that indicated that FOXO1 affected cell proliferation, survival, and androgen receptor signaling, the combined data indicates that FOXO1 is a tumor suppressor gene in prostate cancer.
FOXO1 was identified as only gene within a minimal common region of deletion in chromosomal region 13q14 in prostate carcinoma. FOXO1 deletion was detected in about 30% of prostate cancer samples, and additional cases were identified with reduced expression without evidence of deletion. Based on functional testing in prostate cancer cell lines that indicated that FOXO1 affected cell proliferation, survival, and androgen receptor signaling, the combined data indicates that FOXO1 is a tumor suppressor gene in prostate cancer.
Implicated in
Entity name
Alveolar Rhabdomyosarcoma (ARMS)
Disease
ARMS is one subtype of a family of pediatric soft tissue tumors that is related to the skeletal muscle lineage. In contrast to embryonal rhabdomyosarcoma (ERMS), the other major subtype in this family, ARMS often occurs in adolescents and young adults, with primary tumors located in the vicinity of skeletal muscle, such as in the extremities and trunk.
Prognosis
The overall prognosis for patients with ARMS is less favorable compared to those with ERMS. The three year survival rate for patients without metastatic diseae was 66% (IRS-IV clinical trial), and evidence of metastasis decreased the survival rate to only 16%. The pathogenesis of metastatic ARMS is associated with an early and wide dissemination, often involving bone marrow, and to poor response to chemotherapy. One study also indicates that for metastatic patients, tumors with a PAX3-FKHR fusion have a poorer outcome than tumors with a PAX7-FKHR fusion. In contrast, there is no reported difference in outcome between these subtypes in patients with non-metastatic tumors.
Cytogenetics
Translocations involving the q14 band on chromosome 13 and the q35 band on chromosome 2 - t(2;13)(q35;q14) - distinguish ARMS from other soft tissue sarcomas. An additional 1;13 translocation - t(1;13)(p36;q14) - has been identified in a smaller number of ARMS cases.
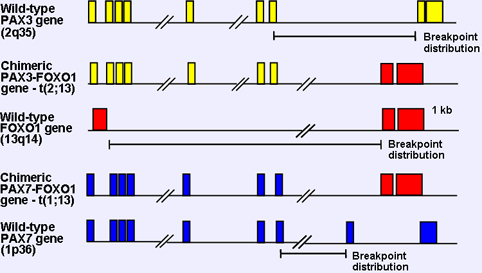
Figure 2. Generation of chimeric genes by the 2;13 and 1;13 translocations in ARMS. The exons of the wild-type and fusion genes are shown as boxes above each map and the translocation breakpoint distributions are shown as line segments below the map of the wild-type genes
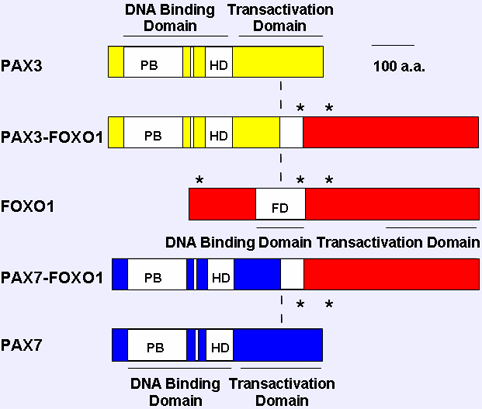
Figure 3. Comparison of wild-type and fusion products associated with the 2;13 and 1;13 translocations. The paired box, octapeptide, homeobox and fork head domain are indicated as open boxes, and transcriptional domains (DNA binding domain, DBD; transcriptional activiation domain) are shown as solid bars. The sites phosphorylated by Akt are indicated by stars. The vertical dash line indicates the translocation fusion point
Entity name
Prostate carcinoma
Disease
Adenocarcinoma of the prostate is a malignant tumor arising from the glandular epithelium of the prostate gland.
Prognosis
In several studies in prostate cancer correlating outcome and chromosomal changes detected by allelic loss or comparative genomic hybridization, deletions or losses involving chromosomal region 13q14 were not found to be correlated with significant differences in outcome.
Cytogenetics
One of the most frequent deletions in prostate cancer involves the q arm of chromosome 13. There are two common regions of deletion in the q arm of chromosome 13: 13q14 and 13q21.
Breakpoints
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 15342912 | 2004 | Proteasomal degradation of the FoxO1 transcriptional regulator in cells transformed by the P3k and Akt oncoproteins. | Aoki M et al |
| 8098985 | 1993 | Rearrangement of the PAX3 paired box gene in the paediatric solid tumour alveolar rhabdomyosarcoma. | Barr FG et al |
| 8643596 | 1996 | Mechanism for transcriptional gain of function resulting from chromosomal translocation in alveolar rhabdomyosarcoma. | Bennicelli JL et al |
| 10377430 | 1999 | Protein kinase B/Akt-mediated phosphorylation promotes nuclear exclusion of the winged helix transcription factor FKHR1. | Biggs WH 3rd et al |
| 12606579 | 2003 | FKHR (FOXO1a) is required for myotube fusion of primary mouse myoblasts. | Bois PR et al |
| 10102273 | 1999 | Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. | Brunet A et al |
| 11689711 | 2001 | Regulation of the Forkhead transcription factor AFX by Ral-dependent phosphorylation of threonines 447 and 451. | De Ruiter ND et al |
| 15905404 | 2005 | Functional interaction between beta-catenin and FOXO in oxidative stress signaling. | Essers MA et al |
| 15184386 | 2004 | Abnormal angiogenesis in Foxo1 (Fkhr)-deficient mice. | Furuyama T et al |
| 12913110 | 2003 | FOXO transcription factors directly activate bim gene expression and promote apoptosis in sympathetic neurons. | Gilley J et al |
| 17038621 | 2006 | CDK2-dependent phosphorylation of FOXO1 as an apoptotic response to DNA damage. | Huang H et al |
| 12891709 | 2003 | Forkhead transcription factor FoxO1 transduces insulin-like growth factor's signal to p27Kip1 in primary skeletal muscle satellite cells. | Machida S et al |
| 14980222 | 2004 | Mammalian SIRT1 represses forkhead transcription factors. | Motta MC et al |
| 12530968 | 2003 | The forkhead transcription factor Foxo1 regulates adipocyte differentiation. | Nakae J et al |
| 11073996 | 2000 | Forkhead transcription factors are critical effectors of cell death and cell cycle arrest downstream of PTEN. | Nakamura N et al |
| 15890677 | 2005 | The coactivator p300 directly acetylates the forkhead transcription factor Foxo1 and stimulates Foxo1-induced transcription. | Perrot V et al |
| 12517744 | 2003 | Akt activation promotes degradation of tuberin and FOXO3a via the proteasome. | Plas DR et al |
| 16100571 | 2005 | Involvement of Foxo transcription factors in angiogenesis and postnatal neovascularization. | Potente M et al |
| 12754525 | 2003 | Insulin-regulated hepatic gluconeogenesis through FOXO1-PGC-1alpha interaction. | Puigserver P et al |
| 15084259 | 2004 | Integration of Smad and forkhead pathways in the control of neuroepithelial and glioblastoma cell proliferation. | Seoane J et al |
| 12039929 | 2002 | PAX3-FKHR and PAX7-FKHR gene fusions are prognostic indicators in alveolar rhabdomyosarcoma: a report from the children's oncology group. | Sorensen PH et al |
| 11994454 | 2002 | The forkhead transcription factor FoxO regulates transcription of p27Kip1 and Bim in response to IL-2. | Stahl M et al |
| 15005655 | 2004 | The ins and outs of FoxO shuttling: mechanisms of FoxO translocation and transcriptional regulation. | Van Der Heide LP et al |
| 2566386 | 1989 | The homeotic gene fork head encodes a nuclear protein and is expressed in the terminal regions of the Drosophila embryo. | Weigel D et al |
| 16113643 | 2005 | The ubiquitin signal: assembly, recognition and termination. Symposium on ubiquitin and signaling. | Wilkinson KD et al |
| 11311120 | 2001 | The kinase DYRK1A phosphorylates the transcription factor FKHR at Ser329 in vitro, a novel in vivo phosphorylation site. | Woods YL et al |
| 15286710 | 2004 | Analysis of the transforming and growth suppressive activities of the PAX3-FKHR oncoprotein. | Xia SJ et al |
| 10602488 | 1999 | Regulation of the forkhead transcription factor FKHR, but not the PAX3-FKHR fusion protein, by the serine/threonine kinase Akt. | del Peso L et al |
| 16964248 | 2006 | FOXO4 transcriptional activity is regulated by monoubiquitination and USP7/HAUSP. | van der Horst A et al |
Other Information
Locus ID:
NCBI: 2308
MIM: 136533
HGNC: 3819
Ensembl: ENSG00000150907
Variants:
dbSNP: 2308
ClinVar: 2308
TCGA: ENSG00000150907
COSMIC: FOXO1
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000150907 | ENST00000379561 | Q12778 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA128406956 | fluorouracil | Chemical | ClinicalAnnotation, VariantAnnotation | associated | PD | ||
| PA443560 | Breast Neoplasms | Disease | ClinicalAnnotation, VariantAnnotation | associated | PD | ||
| PA445113 | Neutropenia | Disease | ClinicalAnnotation, VariantAnnotation | associated | PD | ||
| PA445204 | Ovarian Neoplasms | Disease | MultilinkAnnotation | associated | 26554863 | ||
| PA449165 | cyclophosphamide | Chemical | ClinicalAnnotation, VariantAnnotation | associated | PD | ||
| PA449476 | epirubicin | Chemical | ClinicalAnnotation, VariantAnnotation | associated | PD |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37678798 | 2024 | Hepatocyte FoxO1 Deficiency Protects From Liver Fibrosis via Reducing Inflammation and TGF-β1-mediated HSC Activation. | 4 |
| 37952722 | 2024 | Hyperglycemia-induced miR182-5p drives glycolytic and angiogenic response in Proliferative Diabetic Retinopathy and RPE cells via depleting FoxO1. | 0 |
| 38029456 | 2024 | PTEN-mediated FOXO signaling affects autophagy, migration and invasion of rheumatoid arthritis fibroblast-like synoviocytes. | 0 |
| 38073044 | 2024 | Tumor-derived exosomal miR-1247-3p promotes angiogenesis in bladder cancer by targeting FOXO1. | 2 |
| 38104484 | 2024 | M(6)A demethylase ALKBH5 regulates FOXO1 mRNA stability and chemoresistance in triple-negative breast cancer. | 1 |
| 38224261 | 2024 | The FOXO1/G6PC axis promotes gastric cancer progression and mediates 5-fluorouracil resistance by targeting the PI3K/AKT/mTOR signaling pathway. | 0 |
| 38229320 | 2024 | METTL14-mediated lncRNA XIST silencing alleviates GDM progression by facilitating trophoblast cell proliferation and migration via the miR-497-5p/FOXO1 axis. | 0 |
| 38252663 | 2024 | Brg1/PRMT5 nuclear complex epigenetically regulates FOXO1 in IPF mesenchymal progenitor cells. | 0 |
| 38267912 | 2024 | Genetic variants in the FOXO1 and ZNF469 genes are associated with keratoconus in Sweden: a case-control study. | 0 |
| 38338691 | 2024 | The Roles and Regulatory Mechanisms of Tight Junction Protein Cingulin and Transcription Factor Forkhead Box Protein O1 in Human Lung Adenocarcinoma A549 Cells and Normal Lung Epithelial Cells. | 0 |
| 38343056 | 2024 | Silencing FUT4 Inhibits the Progression of Osteosarcoma through Activation of FOXO1. | 0 |
| 38403332 | 2024 | FoxO1 promotes ovarian cancer by increasing transcription and METTL14-mediated m(6)A modification of SMC4. | 2 |
| 38474048 | 2024 | Human Amniotic MSC Response in LPS-Stimulated Ascites from Patients with Cirrhosis: FOXO1 Gene and Th17 Activation in Enhanced Antibacterial Activation. | 0 |
| 38500269 | 2024 | FOXO1 regulates wound-healing responses in human gingival fibroblasts. | 0 |
| 38519029 | 2024 | FOXO1 promotes cancer cell growth through MDM2-mediated p53 degradation. | 3 |
Citation
Roddy OConnor ; Frederic G Barr
FOXO1 (Forkhead box O1)
Atlas Genet Cytogenet Oncol Haematol. 2008-05-01
Online version: http://atlasgeneticsoncology.org/gene/83/foxo1-(forkhead-box-o1)
