IRS2 (insulin receptor substrate 2)
2016-05-01 Joao Agostinho Machado-Neto, PhD , Paula de Melo Campos , Fabiola Traina AffiliationAbstract
Insulin receptor substrate 2 (IRS2) belongs to the insulin receptor substrate protein family and was initially discovered as an alternative route for signaling mediated by the insulin receptor. Currently, IRS2 has been well-established to mediate mitogenic and antiapoptotic signaling from several important cellular receptors. In the last years, many studies have indicated that IRS2 participates in the regulation of important biological processes involved in cancer phenotype, including cell proliferation, clonogenicity, metabolism and survival. The present review contains data on IRS2 DNA\/RNA, protein encoded and function.
DNA/RNA
Description
Proteins
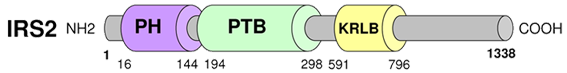
Description
Expression
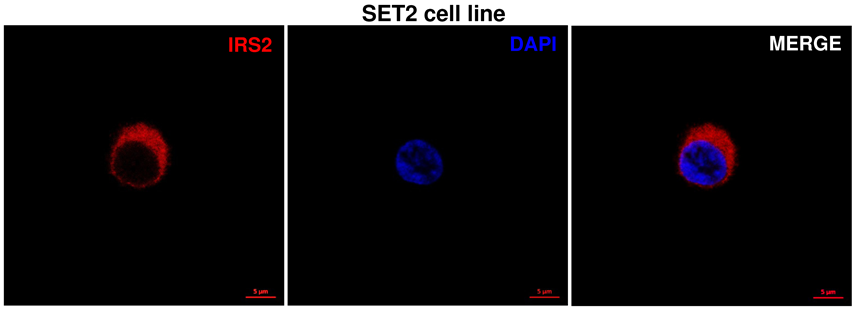
Localisation
Function
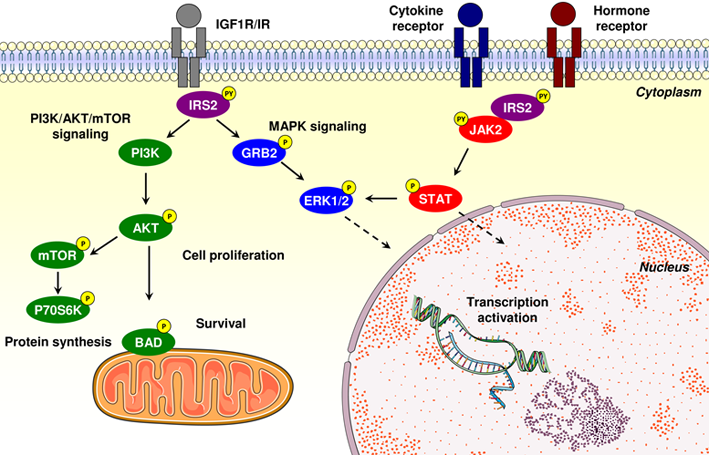
Homology
Table 1. Comparative identity of human IRS2 with other species
| % Identity for: Homo sapiens IRS2 | Symbol | Protein | DNA |
vs. P. troglodytes | IRS2 | 96.9 | 97.7 |
vs. M. mulatta | IRS2 | 97.4 | 95.9 |
vs. C. lupus | IRS2 | 88.8 | 87.4 |
vs. B. taurus | IRS2 | 85.0 | 84.8 |
vs. M. musculus | Irs2 | 84.7 | 80.8 |
vs. R. norvegicus | Irs2 | 85.7 | 81.5 |
vs. G. gallus | IRS2 | 73.7 | 74.4 |
vs. X. tropicalis | LOC100498409 | 59.4 | 57.1 |
vs. D. rerio | Irs2 | 60.7 | 61.7 |
vs. D. rerio | zgc:56306 | 58.9 | 56.5 |
(Source: http://www.ncbi.nlm.nih.gov/homologene)
Mutations
Note
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 24504452 | 2014 | Investigation of insulin resistance gene polymorphisms in patients with differentiated thyroid cancer. | Akker M et al |
| 17443497 | 2007 | DOK4/IRS-5 expression is altered in clear cell renal cell carcinoma. | Al-Sarraf N et al |
| 8910607 | 1996 | Growth hormone, interferon-gamma, and leukemia inhibitory factor utilize insulin receptor substrate-2 in intracellular signaling. | Argetsinger LS et al |
| 16127164 | 2005 | Overexpression of insulin receptor substrate-2 in human and murine hepatocellular carcinoma. | Boissan M et al |
| 11014193 | 2000 | IRS-2 pathways integrate female reproduction and energy homeostasis. | Burks DJ et al |
| 17043687 | 2006 | Insulin receptor substrates mediate distinct biological responses to insulin-like growth factor receptor activation in breast cancer cells. | Byron SA et al |
| 16871543 | 2006 | Aspartyl-asparagyl beta hydroxylase over-expression in human hepatoma is linked to activation of insulin-like growth factor and notch signaling mechanisms. | Cantarini MC et al |
| 12674509 | 2003 | Interaction between leptin and insulin signaling pathways differentially affects JAK-STAT and PI 3-kinase-mediated signaling in rat liver. | Carvalheira JB et al |
| 20184486 | 2010 | Insulin receptor substrate-2 gene polymorphism: is it associated with endometrial cancer? | Cayan F et al |
| 21258861 | 2011 | Membrane localization of insulin receptor substrate-2 (IRS-2) is associated with decreased overall survival in breast cancer. | Clark JL et al |
| 16707456 | 2006 | Epidermal growth factor induces insulin receptor substrate-2 in breast cancer cells via c-Jun NH(2)-terminal kinase/activator protein-1 signaling to regulate cell migration. | Cui X et al |
| 14534541 | 2003 | Progesterone crosstalks with insulin-like growth factor signaling in breast cancer cells via induction of insulin receptor substrate-2. | Cui X et al |
| 23594372 | 2013 | IRS2 is a candidate driver oncogene on 13q34 in colorectal cancer. | Day E et al |
| 17030631 | 2006 | Mammary tumorigenesis and metastasis caused by overexpression of insulin receptor substrate 1 (IRS-1) or IRS-2. | Dearth RK et al |
| 18611262 | 2008 | Genetic variation in candidate obesity genes ADRB2, ADRB3, GHRL, HSD11B1, IRS1, IRS2, and SHC1 and risk for breast cancer in the Cancer Prevention Study II. | Feigelson HS et al |
| 26119932 | 2016 | Targeting melanoma with NT157 by blocking Stat3 and IGF1R signaling. | Flashner-Abramson E et al |
| 9410892 | 1997 | Angiotensin II inhibits insulin signaling in aortic smooth muscle cells at multiple levels. A potential role for serine phosphorylation in insulin/angiotensin II crosstalk. | Folli F et al |
| 24810113 | 2014 | IGF-1R, a target of let-7b, mediates crosstalk between IRS-2/Akt and MAPK pathways to promote proliferation of oral squamous cell carcinoma. | Gao L et al |
| 26029165 | 2015 | Preclinical Effectiveness of Selective Inhibitor of IRS-1/2 NT157 in Osteosarcoma Cell Lines. | Garofalo C et al |
| 17361103 | 2007 | Divergent roles for IRS-1 and IRS-2 in breast cancer metastasis. | Gibson SL et al |
| 17416760 | 2007 | Insulin resistance-related genes and advanced left-sided colorectal adenoma. | Gunter MJ et al |
| 15492273 | 2004 | Selective activation of insulin receptor substrate-1 and -2 in pleural mesothelioma cells: association with distinct malignant phenotypes. | Hoang CD et al |
| 25527633 | 2015 | IRS2 copy number gain, KRAS and BRAF mutation status as predictive biomarkers for response to the IGF-1R/IR inhibitor BMS-754807 in colorectal cancer cell lines. | Huang F et al |
| 22844442 | 2012 | Genetic variants in CASP3, BMP5, and IRS2 genes may influence survival in prostate cancer patients receiving androgen-deprivation therapy. | Huang SP et al |
| 18819936 | 2008 | Progesterone receptor-B regulation of insulin-like growth factor-stimulated cell migration in breast cancer cells via insulin receptor substrate-2. | Ibrahim YH et al |
| 25267499 | 2014 | The tyrphostin NT157 suppresses insulin receptor substrates and augments therapeutic response of prostate cancer. | Ibuki N et al |
| 11704861 | 2001 | Regulation of breast cancer cell motility by insulin receptor substrate-2 (IRS-2) in metastatic variants of human breast cancer cell lines. | Jackson JG et al |
| 7499365 | 1995 | Interleukins 2, 4, 7, and 15 stimulate tyrosine phosphorylation of insulin receptor substrates 1 and 2 in T cells. Potential role of JAK kinases. | Johnston JA et al |
| 2013 | 1976 | Rapid infusion of sodium bicarbonate and albumin into high-risk premature infants soon after birth: a controlled, prospective trial. | Bland RD et al |
Other Information
Locus ID:
NCBI: 8660
MIM: 600797
HGNC: 6126
Ensembl: ENSG00000185950
Variants:
dbSNP: 8660
ClinVar: 8660
TCGA: ENSG00000185950
COSMIC: IRS2
RNA/Proteins
| Gene ID | Transcript ID | Uniprot |
|---|---|---|
| ENSG00000185950 | ENST00000375856 | Q9Y4H2 |
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37481466 | 2024 | Insulin receptor substrate 2 gene Gly1057Asp polymorphism is a risk factor for nonalcoholic fatty liver disease. | 0 |
| 37481466 | 2024 | Insulin receptor substrate 2 gene Gly1057Asp polymorphism is a risk factor for nonalcoholic fatty liver disease. | 0 |
| 35727487 | 2023 | Structural Consequences of IRS-2 nsSNPs and Implication for Insulin Receptor Substrate-2 Protein Stability. | 0 |
| 36631465 | 2023 | Identification of the ageing-related prognostic gene signature, and the associated regulation axis in skin cutaneous melanoma. | 0 |
| 36879410 | 2023 | Enhancer RNA IRS2e is essential for IRS2 expression and the oncogenic properties in oral squamous cell carcinoma. | 0 |
| 37173295 | 2023 | Association of IRS-1 and IRS-2 polymorphisms with predisposition to type-2 diabetes (T2D): a meta-analysis and trial sequential analysis. | 0 |
| 37716291 | 2023 | Influence of polymorphisms in IRS1, IRS2, MC3R, and MC4R on metabolic and inflammatory status and food intake in Brazilian adults: An exploratory pilot study. | 0 |
| 37894751 | 2023 | The IGF-Independent Role of IRS-2 in the Secretion of MMP-9 Enhances the Growth of Prostate Carcinoma Cell Line PC3. | 0 |
| 35727487 | 2023 | Structural Consequences of IRS-2 nsSNPs and Implication for Insulin Receptor Substrate-2 Protein Stability. | 0 |
| 36631465 | 2023 | Identification of the ageing-related prognostic gene signature, and the associated regulation axis in skin cutaneous melanoma. | 0 |
| 36879410 | 2023 | Enhancer RNA IRS2e is essential for IRS2 expression and the oncogenic properties in oral squamous cell carcinoma. | 0 |
| 37173295 | 2023 | Association of IRS-1 and IRS-2 polymorphisms with predisposition to type-2 diabetes (T2D): a meta-analysis and trial sequential analysis. | 0 |
| 37716291 | 2023 | Influence of polymorphisms in IRS1, IRS2, MC3R, and MC4R on metabolic and inflammatory status and food intake in Brazilian adults: An exploratory pilot study. | 0 |
| 37894751 | 2023 | The IGF-Independent Role of IRS-2 in the Secretion of MMP-9 Enhances the Growth of Prostate Carcinoma Cell Line PC3. | 0 |
| 35124585 | 2022 | Single nucleotide polymorphisms in binding site of miRNA-135a and targeted gene IRS2 are correlated with multiple clinical features of PCOS: A study in Chinese women. | 0 |
Citation
Joao Agostinho Machado-Neto, PhD ; Paula de Melo Campos ; Fabiola Traina
IRS2 (insulin receptor substrate 2)
Atlas Genet Cytogenet Oncol Haematol. 2016-05-01
Online version: http://atlasgeneticsoncology.org/gene/40994/irs2-(insulin-receptor-substrate-2)
