AKT2 (v-akt murine thymoma viral oncogene homolog 2)
2007-07-01 Deborah A. Altomare , Joseph R. Testa AffiliationFox Chase Cancer Center, 333 Cottman Avenue, Philadelphia, PA 19111, USA
Identity
HGNC
LOCATION
19q13.2
IMAGE

LOCUSID
ALIAS
HIHGHH,PKBB,PKBBETA,PRKBB,RAC-BETA
FUSION GENES
DNA/RNA
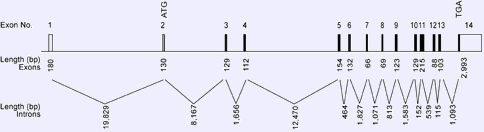
Genomic organization of human AKT2. Open boxes indicate untranslated regions and shaded boxes indicate coding regions of the gene. The ATG transcription start site is located in exon 2 and the TGA termination codon is located in exon 14.
Description
The entire gene is about 54.4 Kb and contains 14 exons. The open reading frame of the coding region is 1,445 bp.
Transcription
Transcript length: 4,623 bp
Pseudogene
No human pseudogene known. A mouse Akt2 pseudogene was cloned and mapped to proximal mouse chromosome 11 by fluorescence in situ hybridization.
Proteins

AKT proteins contain an amino terminal pleckstrin homology (PH) domain, followed by a short helical region and kinase domain that terminates in a regulatory hydrophobic motif. Activation of AKT kinases is a multi-step process that involves both membrane translocation and phosphorylation. AKT activation occurs by means of stimulation of the growth factor receptor-associated phosphatidylinositol 3-kinase (PI3K). PI3K generates 3-phosphorylated phosphoinositides, i.e., phosphatidylinositol-3,4,5-trisphosphate (PIP3) and phosphatidylinositol-3,4-bisphosphate (PIP2) at the plasma membrane. Both phospholipids bind with high affinity to the PH domain, mediating membrane translocation of AKT. At the membrane, AKT2 is phosphorylated at two sites, threonine 309 (T309) and serine 474 (S474).
Description
AKT2 protein consists of 481 amino acids, with a molecular weight of 55,769 Da.
Expression
Found in all human cell types so far analyzed; insulin responsive tissues such as normal brown fat, skeletal muscle and liver exhibit the highest expression levels of AKT2/Akt2.
Localisation
Predominantly cytoplasmic; also found at the plasma membrane and in the nucleus following its activation.
Function
AKT proteins mediate a variety of cellular functions, ranging from control of cell proliferation and survival to modulation of intermediary metabolism and angiogenesis. Such pleiotropic effects are the consequence of phosphorylation of numerous substrates, some of which are listed below. Most substrates share the consensus sequence for AKT phosphorylation, RXRXXS/T. For example, activated AKT exerts anti-apoptotic activity in part by preventing the release of cytochrome c from mitochondria, and phosphorylating and inactivating the pro-apoptotic factors BAD and pro-caspase-9. AKT also activates IkappaB kinase (IKK), a positive regulator of NF-kappaB, which results in the transcription of anti-apoptotic genes. AKT phosphorylates and inactivates FOXO transcription factors, which mediate the expression of genes critical for apoptosis, such as the Fas ligand gene.
AKT activation mediates cell cycle progression by phosphorylation and inhibition of glycogen synthase kinase 3 beta to inhibit cyclin D1 degradation. AKT phosphorylates the cell cycle inhibitors p21WAF1 and p27Kip1 near the nuclear localization signal to induce cytoplasmic retention of these cell cycle inhibitors. Moreover, phosphorylation of AKT kinases also results in increased translation of cyclin D1, D3 and E transcripts.
AKT activates the downstream mTOR kinase by inhibiting a complex formed by the tumor suppressor proteins TSC1 and TSC2, also known as hamartin and tuberin, respectively. mTOR broadly mediates cell growth and proliferation by regulating ribosomal biogenesis and protein translation and can regulate response to nutrients by restricting cell cycle progression in the presence of suboptimal growth conditions.
AKT signaling also contributes to other cellular processes considered to be cancer hallmarks. AKT promotes the phosphorylation and translocation of Mdm2 into the nucleus, where it downregulates p53 and thereby antagonizes p53-mediated cell cycle checkpoints. AKT signaling is linked to tumor cell migration, and it contributes to tumor invasion and metastasis by promoting the secretion of matrix metalloproteinases. Moreover, vascular endothelial growth factor (VEGF) effects on cell survival have been shown to be mediated by the Flk1/VEGFR2-PI3K-AKT pathway. In other cellular processes, AKT has been shown to phosphorylate human telomerase reverse transcriptase (hTERT), thereby stimulating telomerase activity and replication. Collectively, these findings implicate up-regulation of the AKT pathway in many aspects of tumorigenesis.
AKT activation mediates cell cycle progression by phosphorylation and inhibition of glycogen synthase kinase 3 beta to inhibit cyclin D1 degradation. AKT phosphorylates the cell cycle inhibitors p21WAF1 and p27Kip1 near the nuclear localization signal to induce cytoplasmic retention of these cell cycle inhibitors. Moreover, phosphorylation of AKT kinases also results in increased translation of cyclin D1, D3 and E transcripts.
AKT activates the downstream mTOR kinase by inhibiting a complex formed by the tumor suppressor proteins TSC1 and TSC2, also known as hamartin and tuberin, respectively. mTOR broadly mediates cell growth and proliferation by regulating ribosomal biogenesis and protein translation and can regulate response to nutrients by restricting cell cycle progression in the presence of suboptimal growth conditions.
AKT signaling also contributes to other cellular processes considered to be cancer hallmarks. AKT promotes the phosphorylation and translocation of Mdm2 into the nucleus, where it downregulates p53 and thereby antagonizes p53-mediated cell cycle checkpoints. AKT signaling is linked to tumor cell migration, and it contributes to tumor invasion and metastasis by promoting the secretion of matrix metalloproteinases. Moreover, vascular endothelial growth factor (VEGF) effects on cell survival have been shown to be mediated by the Flk1/VEGFR2-PI3K-AKT pathway. In other cellular processes, AKT has been shown to phosphorylate human telomerase reverse transcriptase (hTERT), thereby stimulating telomerase activity and replication. Collectively, these findings implicate up-regulation of the AKT pathway in many aspects of tumorigenesis.
Homology
All three AKT kinases belong to the more general class of AGC kinases (related to AMP/GMP kinase and protein kinase C). The kinase domain of AKT shares high similarity with other members of the AGC family of kinases such as PKA, PKC, p70 S6K, and p90 RSK. The sequence identities among the three AKTs in the kinase domain exceed 87%. The three AKT kinases are identical in the ATP binding region, except for one residue: Ala 230 of AKT1 is conserved in AKT2 (Ala 232), but switches to Val 228 in AKT3. In addition, each of the three AKT kinases has a carboxy terminal extension of about 40 amino acids.
Human AKT2 is 98.1% similar to M. musculus Akt2; 97.7% similar to the R. norvegicus homolog; 61.3% similar to D. melanogaster protein kinase RAC; 52.4% similar to C. elegans Akt/PKB serine/threonine kinase; 47.7% similar to S. cerevisiae protein kinase (see UniGene Hs.631535)
Human AKT2 is 98.1% similar to M. musculus Akt2; 97.7% similar to the R. norvegicus homolog; 61.3% similar to D. melanogaster protein kinase RAC; 52.4% similar to C. elegans Akt/PKB serine/threonine kinase; 47.7% similar to S. cerevisiae protein kinase (see UniGene Hs.631535)
Mutations
Germinal
Insulin resistance and a diabetes mellitus-like syndrome have been described in knockout mice lacking Akt2.
Somatic
Individuals carrying a G-to-A transition in the AKT2 gene resulting in an Arg-to-His substitution at codon 274 (R274H) were found to be markedly hyperinsulinemic. However, a large case-control study showed that variation in and around the AKT2 locus is unlikely to contribute significantly to increased risk of type 2 diabetes.
Mutations in AKT2 are uncommon in human tumors. For example, AKT2 mutations have been reported in 1 of 51 gastric carcinomas and 2 of 79 lung carcinomas. The mutations consisted of one missense mutation and 2 splice site mutations in an intron.
Mutations in AKT2 are uncommon in human tumors. For example, AKT2 mutations have been reported in 1 of 51 gastric carcinomas and 2 of 79 lung carcinomas. The mutations consisted of one missense mutation and 2 splice site mutations in an intron.
Implicated in
Entity name
Various cancers
Prognosis
Frequent activation of AKT has been reported in a broad range of human cancers including various carcinomas, glioblastoma multiforme, and hematological malignancies. In some of these tumor types, AKT activation has been shown to correlate with advanced disease and/or poor prognosis. AKT is a major mediator of survival signals that protect cells from undergoing apoptosis and, thus, is a potentially important therapeutic target. Ovarian cancer cell lines with either constitutive AKT1 activity or AKT2 gene amplification have been shown to be highly resistant to paclitaxel compared to cells with low AKT levels.
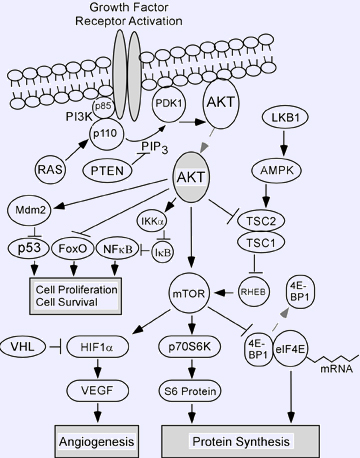
Hyperactivation of AKT kinases have been reported in a wide assortment of human solid tumors and hematological malignancies. Activation of growth factor receptors either by ligand stimulation or receptor overexpression/mutation is one of the mechanisms leading to the upregulation of AKT signaling. Other mechanisms include activation of oncoproteins and inactivation of tumor suppressors intersecting the AKT signal transduction pathway. AKT is now known to be a central player in a signaling pathway consisting of many components that have been implicated in tumorigenesis, including upstream phosphatidylinositol 3-kinase (PI3K) and PTEN (Phosphatase and Tensin homologue deleted on chromosome Ten). Several proteins, such as AKT, eIF4E, and the subunits of PI3K, can act as oncoproteins when activated or overexpressed. Germline mutations in PTEN, LKB1, TSC2/TSC1, and VHL are linked with different dominantly-inherited cancer syndromes. Each of these tumor suppressors is a negative regulator of the AKT pathway which, when deregulated, results in altered translation of cancer-related mRNAs that regulate cellular processes such as cell cycle progression, growth, cell survival, invasion, and communication with the extracellular environment.
Oncogenesis
In 1992, amplification and overexpression of AKT2 was reported in a subset of ovarian carcinomas. AKT2 was shown to be amplified and overexpressed in 2 of 8 ovarian carcinoma cell lines and 2 of 15 primary ovarian tumors. Recently, amplification of AKT2 was found in 18.2% of high-grade ovarian carcinomas.
Amplification and/or overexpression of AKT2 was reported in 10-20% of primary pancreatic carcinomas and pancreatic cancer cell lines. PANC1 and ASPC1 cell lines exhibited 30-fold and 50-fold amplification of AKT2, respectively, and highly elevated levels of AKT2 RNA and protein. As an early indication of the potential importance of molecularly targeting the AKT pathway, AKT2 expression and tumorigenicity of PANC1 cells in nude mice was markedly inhibited by transfection with an antisense AKT2 construct but not with a control AKT2 construct in the sense orientation. Through the use of in vitro kinase assays, activation of the AKT2 kinase has been observed in about 40% of ovarian and pancreatic cancers.
Amplification and/or overexpression of AKT2 was reported in 10-20% of primary pancreatic carcinomas and pancreatic cancer cell lines. PANC1 and ASPC1 cell lines exhibited 30-fold and 50-fold amplification of AKT2, respectively, and highly elevated levels of AKT2 RNA and protein. As an early indication of the potential importance of molecularly targeting the AKT pathway, AKT2 expression and tumorigenicity of PANC1 cells in nude mice was markedly inhibited by transfection with an antisense AKT2 construct but not with a control AKT2 construct in the sense orientation. Through the use of in vitro kinase assays, activation of the AKT2 kinase has been observed in about 40% of ovarian and pancreatic cancers.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 16288292 | 2005 | Perturbations of the AKT signaling pathway in human cancer. | Altomare DA et al |
| 16288294 | 2005 | Tuberous sclerosis complex: linking growth and energy signaling pathways with human disease. | Astrinidis A et al |
| 16095999 | 2005 | Activation of AKT kinases in cancer: implications for therapeutic targeting. | Bellacosa A et al |
| 1409633 | 1992 | AKT2, a putative oncogene encoding a member of a subfamily of protein-serine/threonine kinases, is amplified in human ovarian carcinomas. | Cheng JQ et al |
| 8622988 | 1996 | Amplification of AKT2 in human pancreatic cells and inhibition of AKT2 expression and tumorigenicity by antisense RNA. | Cheng JQ et al |
| 11387480 | 2001 | Insulin resistance and a diabetes mellitus-like syndrome in mice lacking the protein kinase Akt2 (PKB beta). | Cho H et al |
| 15209377 | 2004 | PI 3-kinase, Akt and cell survival. | Downward J et al |
| 15166380 | 2004 | A family with severe insulin resistance and diabetes due to a mutation in AKT2. | George S et al |
| 10647931 | 2000 | The hallmarks of cancer. | Hanahan D et al |
| 11410335 | 2001 | Expression, purification, characterization and homology modeling of active Akt/PKB, a key enzyme involved in cell survival signaling. | Kumar CC et al |
| 15800333 | 2005 | Possible future issues in the treatment of glioblastomas: special emphasis on cell migration and the resistance of migrating glioblastoma cells to apoptosis. | Lefranc F et al |
| 12851486 | 2003 | Multiple roles of the PI3K/PKB (Akt) pathway in cell cycle progression. | Liang J et al |
| 10593857 | 1999 | Studies of the molecular mechanisms in the regulation of telomerase activity. | Liu JP et al |
| 12217521 | 2002 | The PTEN, Mdm2, p53 tumor suppressor-oncoprotein network. | Mayo LD et al |
| 8780719 | 1996 | Isolation of DNA sequences amplified at chromosome 19q13.1-q13.2 including the AKT2 locus in human pancreatic cancer. | Miwa W et al |
| 9792703 | 1998 | Cyclin D expression is controlled post-transcriptionally via a phosphatidylinositol 3-kinase/Akt-dependent pathway. | Muise-Helmericks RC et al |
| 16721043 | 2006 | Sequence mutations and amplification of PIK3CA and AKT2 genes in purified ovarian serous neoplasms. | Nakayama K et al |
| 10769688 | 2000 | Overexpression of Akt/AKT can modulate chemotherapy-induced apoptosis. | Page C et al |
| 16288290 | 2005 | Akt-dependent transformation: there is more to growth than just surviving. | Plas DR et al |
| 15077155 | 2004 | Apoptosis defects and chemotherapy resistance: molecular interaction maps and networks. | Pommier Y et al |
| 9496907 | 1998 | Amplification and overexpression of the AKT2 oncogene in a subset of human pancreatic ductal adenocarcinomas. | Ruggeri BA et al |
| 16288289 | 2005 | The Akt of translational control. | Ruggero D et al |
| 12089061 | 2002 | Role of Akt signaling in vascular homeostasis and angiogenesis. | Shiojima I et al |
| 17047397 | 2006 | Mutational analysis of AKT1, AKT2 and AKT3 genes in common human carcinomas. | Soung YH et al |
| 17327441 | 2007 | Analysis of genetic variation in Akt2/PKB-beta in severe insulin resistance, lipodystrophy, type 2 diabetes, and related metabolic phenotypes. | Tan K et al |
| 11572954 | 2001 | AKT plays a central role in tumorigenesis. | Testa JR et al |
| 11467775 | 2000 | Fibronectin activates matrix metalloproteinase-9 secretion via the MEK1-MAPK and the PI3K-Akt pathways in ovarian cancer cells. | Thant AA et al |
| 15110188 | 2004 | Regulation of sensitivity to TRAIL by the PTEN tumor suppressor. | Whang YE et al |
| 10822383 | 2000 | Frequent activation of AKT2 and induction of apoptosis by inhibition of phosphoinositide-3-OH kinase/Akt pathway in human ovarian cancer. | Yuan ZQ et al |
Other Information
Locus ID:
NCBI: 208
MIM: 164731
HGNC: 392
Ensembl: ENSG00000105221
Variants:
dbSNP: 208
ClinVar: 208
TCGA: ENSG00000105221
COSMIC: AKT2
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA248 | NFKB1 | Gene | Pathway | associated | |||
| PA254 | NOS3 | Gene | Pathway | associated | 20124951 | ||
| PA27749 | ELK1 | Gene | Pathway | associated | |||
| PA28212 | FOS | Gene | Pathway | associated | |||
| PA296 | RELA | Gene | Pathway | associated | |||
| PA30006 | JUN | Gene | Pathway | associated | |||
| PA31353 | MYC | Gene | Pathway | associated | |||
| PA31600 | NFKB2 | Gene | Pathway | associated | |||
| PA33304 | PIK3C2A | Gene | Pathway | associated | 20124951 | ||
| PA33305 | PIK3C2B | Gene | Pathway | associated | 20124951 | ||
| PA337 | STAT3 | Gene | Pathway | associated | |||
| PA338 | STAT5A | Gene | Pathway | associated | |||
| PA36042 | SP1 | Gene | Pathway | associated | |||
| PA36183 | STAT1 | Gene | Pathway | associated | |||
| PA36184 | STAT2 | Gene | Pathway | associated | |||
| PA36185 | STAT4 | Gene | Pathway | associated | |||
| PA36186 | STAT5B | Gene | Pathway | associated |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38244769 | 2024 | Active AKT2 stimulation of SREBP1/SCD1-mediated lipid metabolism boosts hepatosteatosis and cancer. | 0 |
| 38396845 | 2024 | All Three AKT Isoforms Can Upregulate Oxygen Metabolism and Lactate Production in Human Hepatocellular Carcinoma Cell Lines. | 0 |
| 38478015 | 2024 | m 6 A reader IGF2BP1 reduces the sensitivity of nasopharyngeal carcinoma cells to Taxol by upregulation of AKT2. | 0 |
| 38528080 | 2024 | Differential prognostic values of the three AKT isoforms in acute myeloid leukemia. | 0 |
| 38244769 | 2024 | Active AKT2 stimulation of SREBP1/SCD1-mediated lipid metabolism boosts hepatosteatosis and cancer. | 0 |
| 38396845 | 2024 | All Three AKT Isoforms Can Upregulate Oxygen Metabolism and Lactate Production in Human Hepatocellular Carcinoma Cell Lines. | 0 |
| 38478015 | 2024 | m 6 A reader IGF2BP1 reduces the sensitivity of nasopharyngeal carcinoma cells to Taxol by upregulation of AKT2. | 0 |
| 38528080 | 2024 | Differential prognostic values of the three AKT isoforms in acute myeloid leukemia. | 0 |
| 36217693 | 2023 | Phosphatase and tensin homologue determine inflammatory status by differentially regulating the expression of Akt1 and Akt2 in macrophage alternative polarization of periodontitis. | 6 |
| 36409629 | 2023 | Finnish-specific AKT2 gene variant leads to impaired insulin signalling in myotubes. | 0 |
| 36719624 | 2023 | CircRNA Circ_0000118 Regulates Malignancy of Cervical Cancer Cells by Regulating miR-211-5p/miR-377-3p/AKT2 Axis. | 1 |
| 37567009 | 2023 | Hsa_circRNA_103124 upregulation in Crohn's disease promoted macrophage M1 polarization to maintain an inflammatory microenvironment via activation of the AKT2 and TLR4/NF-κB pathways. | 1 |
| 37879895 | 2023 | Differential activation of AKT isoforms by growth factors in human myotubes. | 1 |
| 36217693 | 2023 | Phosphatase and tensin homologue determine inflammatory status by differentially regulating the expression of Akt1 and Akt2 in macrophage alternative polarization of periodontitis. | 6 |
| 36409629 | 2023 | Finnish-specific AKT2 gene variant leads to impaired insulin signalling in myotubes. | 0 |
Citation
Deborah A. Altomare ; Joseph R. Testa
AKT2 (v-akt murine thymoma viral oncogene homolog 2)
Atlas Genet Cytogenet Oncol Haematol. 2007-07-01
Online version: http://atlasgeneticsoncology.org/gene/517/akt2-(v-akt-murine-thymoma-viral-oncogene-homolog-2)
