dup (11q) in myeloid malignancies
2016-11-01 Soad Al Bahar , Adriana Zamecnikova Affiliation1./ Kuwait Cancer Control Center, Department of Hematology, Laboratory of Cancer Genetics, Kuwait [email protected]
Abstract
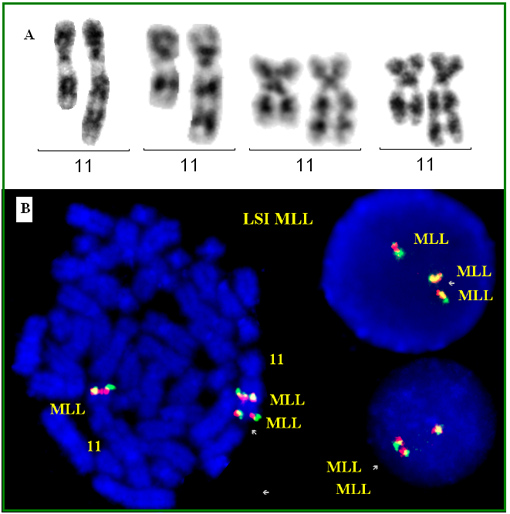
Partial gain of the long arm of chromosome 11, containing the unrearranged mixed lineage leukaemia (MLL) gene (KMT2A; lysine (K)-specific methyltransferase 2A) is a rare but recurrent anomaly in myeloid malignancies, and is often associated with an older age and a higly complex karyotype.
Clinics and Pathology
Disease
Phenotype stem cell origin
| Sex/ Age | Diagnosis | Karyotype | |
| 1 | F/0 | AML- M5 | 51,XX,+1,del(1)(p22)x2,+3,+6,+9,add(10)(p13),dup(11)(q11q21),+19/52,idem,+18 |
| 2 | F/60 | AML | 44,XX,der(1)t(1;4)(q11;p11),+der(1)t(1;7)(p11;p11),-4,del(5)(q14q34),der(7)t(7;17)(q11;q11),dup(11)(q12q13),-12,-17,-20,+mar |
| 3 | M/84 | RAEB | 46,XY,dup(11)(q13q25) |
| 4 | M/53 | AML | 44-45,XY,-3,-5,-10,dup(11)(q12q13),add(12)(p12),add(14)(q32),der(17)t(5;17)(p11;p11),i(18)(q10), +1-2mar |
| 5 | M/59 | AML-M1 | 49-52,XY,+1,+der(2)t(2;11)(q27;q11),der(3)t(3;3)(q23;p23),del(6)(q23),+10, del(11)(q21q23),+dup(11)(q23), +13,add(14)(p13),add(16)(p23),-17,-17,-18,der(19)t(17;19)(q21;p13),inc |
| 6 | F/83 | AML- M2 | 44-47,XX,-5,der(6)t(6;13)(p22;q21),+der(11)ins(11;11)(q24;q?12q14),+dup(11)(q23q25),+i(11)(q10),del(13)(q?),der(16)t(13;16)(q?;q?13),del(17)(p?),-18, der(22)t(13;22)(q?;p11) multiple myeloma, chemotherapy |
| 7 | M/45 | AML-M5a | 46,XX,dup(11)(q23q23) |
| 8 | F/15 | AML-M4 | 49,XX,+8,+8,+8,dup(11)(q14q23)/50,idem,+mar |
| 9 | M/77 | AML-M5b | 45,XY,-10,dup(11)(q23q24) |
| 10 | M/63 | RA | 43,X,-Y,del(5)(q13q33),der(7)t(7;13)(p22;q1?),-9,der(11)add(11)(p15)dup(11)(q14q23),-13 |
| 11 | M | AML-M1 | 43,XY,del(5)(q15q33),-7,-11,dup(11)(q13q23),-17,der(18)t(7;18)(q22;q23)/44, idem,+mar |
| 12 | F/76 | AML-M1 | 45,XX,del(5)(q13q33),del(7)(q?11q36),dup(11)(q23q23),-16,add(16),der(18)t(16;18)(?;p11) |
| 13 | F/66 | CML | 45,XX,-7,-9,t(9;22),add(10)(q26),dup(11)(q22q23),der(19),+del(22)(q?) |
| 14 | M/23 | APL/ AML-M5a | 46,XY,dup(11)(q?),t(15;17)(q22;q21) |
| 15 | F/66 | AML-M1 | 46,XX,del(5)(q13q33),dup(11)(q13q23),t(17;20)(q12;q11)/46,XX,del(5),del(7)(q21q31),+8,dup(11)(q23q23), -14,der(17)t(14;17)(q21;q12),der(20)t(17;20)/46,XX,del(5),del(7),+8,der(11)t(11;11)(q23;p?),-14,der(17) t(14;17),der(20)t(17;20) |
| 16 | M/1 | AML-M7 DS | 44,XY,t(9;15)(p11;q11),dup(11)(q11q13),-13,-15,-16,der(17)t(16;17)(q11;p11),+21c, der(21)t(13;21)(q21;p11) |
| 17 | F/61 | MDS AML-M2 | 46,XX,t(6;9)(p11;p11),t(6;13)(p25;q14),ins(17;1)(q25;q21q25),del(19)(q12)/47,idem,+21 46,XX,t(6;9),t(6;13),-13,+mar/46,XX,del(1)(p11p32),-4,-5,add(7)(q31-32),-9, +dup(11)(q13q23),-12,del(14) (q22),-16,-17,ins(17;1),+der(?)t(?;12)(?;q?), +2mar adenocarcinoma, chemotherapy, radiotherapy |
| 18 | M/80 | AML-M1 | 44,X,-Y,del(5)(q13q33),der(5)t(5;11)(q35;q13),-7,der(21)t(7;21)(?;q2?1)t(7;21)t(7;21)t(7;21) t(7;21)t(7;21)/ 44,X,-Y,del(5),-7,der(16)t(11;16)(q13;q24),der(21)/44,X,-Y,del(5),-7,dup(11)(q23q25),der(21) |
| 19 | F/68 | AML- M2 | 43,XX,-4,del(5)(q12q34),der(10)t(1;10)(p13;q26),dup(11)(q?24q?),-17,-18 |
| 20 | M/63 | AML-M0 | 45,XY,der(2)t(2;18)(p13;p11),del(5)(q13q33),der(7)t(7;11),der(11;12)(q10;q10),dup(11)(q?24q?),+13,der(13)t(7;13)(q?;q21)x2,-16,der(17)t(2;17) (p13;q21),der(17)t(7;17)(?;q24), der(18)t(18;20)(q?;p11),der(20)t(7;20) |
| 21 | M/76 | AML- M2 | 46,XY,der(1;19)t(1;19)(p13;p12)t(1;19)(p36;q13),der(5)t(5;17)(q13;q11-22), dup(11)(q?24q?),der(17)r(5;17) -19,+22,i(22)(q10)x2 |
| 22 | M | RAEB | 44-49,XY,+Y,del(1)(q32),add(2)(p25),del(5)(q15q31),der(6)t(2;6)(q21;q13),del(7)(q22),+9,add(9)(q34)x2, add(11(p13),dup(11)(q13q23),-17,add(17)(p11), add(19)(p13),-20,i(21)(q10),+2mar |
| 23 | F/86 | AML | 44,XX,dic(5;17)(q11;p11),-7,der(10)t(10;11)(q25;q22)/43,idem,del(3)(p21),dic(4;7)(q11;q11),+7,der(8)t(4;8) (q21;p21),-12,der(12)t(12;21)(p13;q22),der(21)t(12;21)(q?;q21)/43,idem,del(3),dic(4;7),+7,der(8)(4;8),dup(11)(q23q23),-12,der(12)t(12;21),der(21)t(12;21) adenocarcinoma, chemotherapy |
| 24 | F/60 | AML-M1 | 46,XX,der(3)t(3;11)(q29;q11)/46,XX,dup(11)(q14q23) |
| 25 | F/64 | AML-M2 | 44,XX,del(2)(q31q35),-4,del(5)(q13q33),-11,dup(11)(q21q23),-14,del(17)(q21),del(18)(q21),+mar |
| 26 | M/62 | AML | 45,XY,del(5)(q13q33),del(7)(q22q35),dup(11)(q?),der(17)t(17;21)(p11;?),der(20)t(20;21)(q12;?),-21/ 44,XY, del(4)(q32),del(5),der(7)t(3;7)(q?;q21),-15,der(16)t(15;16)(?;q22),der(17)t(17;21),der(20)t(20;21),-21 |
| 27 | F/66 | AML-M1 | 46,XX,del(5)(q13q33),del(7)(q21q33),dup(11)(q13q23),-17,der(20)t(17;20)(q12;q11)/46,XX,del(5), del(7),+8, dup(11),-14,der(17)t(14;17)(?;q12),der (20)t(17;20) |
| 28 | M/60 | AML | 46,XY,del(5)(q13q33),dup(11)(q23q23)/43,idem,-4,-17,-18/42,idem,del(2)(q21),-6,-dup(11), add(12)(p11), del(12)(p11),-16,-17,-18 |
| 29 | M/63 | AML | 42,X,-Y,del(1)(q32),add(2)(q13),-4,add(4)(q31),del(5)(q15q33), add(6)(p25), add(6)(q13),der(7;16)(p10;q10), -8,add(10)(p13),dup(11)(q23q25),add(13)(p11),-16,-17,add(18)(q12),add(20)(q13),+2mar diffuse large B-cell lymphoma, chemotherapy |
Abbreviations: F., female; M., male; AML., acute myeloid leukemia; AML-M5., acute monoblastic leukemia; RAEB., refractory anemia with excess of blasts; AML-M1., acute myeloblastic leukemia without maturation; AML-M2., acute myeloblastic leukemia with maturation; AML-M5a., acute monoblastic leukemia without differentiation; AML-M4., acute myelomonocytic leukemia; AML-M5b., acute monocytic leukemia; RA., refractory anemia; CML., chronic myeloid leukemia; APL., acute promyelocytic leukemia; DS., Downs syndrome; MDS., myelodysplastic syndrome; AML-M0., acute myeloblastic leukemia with minimal differentiation.
.
Prognosis
Epidemiology
Cytogenetics
Note
Cytogenetics morphological
Additional anomalies
Genes Involved and Proteins
Result of the Chromosomal Anomaly
Oncogenesis
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 15645489 | 2005 | Centromeric breakage and highly rearranged chromosome derivatives associated with mutations of TP53 are common in therapy-related MDS and AML after therapy with alkylating agents: an M-FISH study. | Andersen MK et al |
| 16102580 | 2005 | Screening by fluorescence in situ hybridization for MLL status at diagnosis in 239 unselected patients with acute myeloblastic leukemia. | Arnaud B et al |
| 16687173 | 2007 | Analysis of complex chromosomal rearrangements in adult patients with MDS and AML by multicolor FISH. | Babicka L et al |
| 12481904 | 2002 | Variations in MLL amplification in a patient with acute myeloid leukemia. | Brezinová J et al |
| 11069023 | 2000 | MLL amplification in acute leukaemia: a United Kingdom Cancer Cytogenetics Group (UKCCG) study. | Cuthbert G et al |
| 12091356 | 2002 | Cytogenetic profile of childhood and adult megakaryoblastic leukemia (M7): a study of the Groupe Français de Cytogénétique Hématologique (GFCH). | Dastugue N et al |
| 10602416 | 1999 | Lack of IRF-1 expression in acute promyelocytic leukemia and in a subset of acute myeloid leukemias with del(5)(q31). | Green WB et al |
| 9593286 | 1998 | Ten novel 11q23 chromosomal partner sites. European 11q23 Workshop participants. | Harrison CJ et al |
| 23483787 | 2013 | Therapy-related myeloid neoplasms in 39 Korean patients: a single institution experience. | Huh HJ et al |
| 6953984 | 1982 | Chromosome pattern in childhood acute nonlymphocytic leukemia (ANLL). | Kaneko Y et al |
| 7885035 | 1995 | Myelodysplastic syndromes and acute myeloid leukemia with 17p deletion. An entity characterized by specific dysgranulopoïesis and a high incidence of P53 mutations. | Lai JL et al |
| 17574959 | 2007 | Fluorescence in situ hybridization analysis of 110 hematopoietic disorders with chromosome 5 abnormalities: do de novo and therapy-related myelodysplastic syndrome-acute myeloid leukemia actually differ? | Lessard M et al |
| 15334543 | 2004 | Structural integrity and expression of the L3MBTL gene in normal and malignant hematopoietic cells. | MacGrogan D et al |
| 12454741 | 2002 | Pooled analysis of clinical and cytogenetic features in treatment-related and de novo adult acute myeloid leukemia and myelodysplastic syndromes based on a consecutive series of 761 patients analyzed 1976-1993 and on 5098 unselected cases reported in the literature 1974-2001. | Mauritzson N et al |
| 11979548 | 2002 | Spectral karyotyping in patients with acute myeloid leukemia and a complex karyotype shows hidden aberrations, including recurrent overrepresentation of 21q, 11q, and 22q. | Mrózek K et al |
| 10861810 | 2000 | Many unbalanced translocations show duplication of a translocation participant. Clinical and cytogenetic implications in myeloid hematologic malignancies. | Pedersen B et al |
| 12946992 | 2004 | Expression analyses identify MLL as a prominent target of 11q23 amplification and support an etiologic role for MLL gain of function in myeloid malignancies. | Poppe B et al |
| 16864856 | 2006 | Disclosure of candidate genes in acute myeloid leukemia with complex karyotypes using microarray-based molecular characterization. | Rücker FG et al |
| 2790747 | 1989 | Refractory anemia with excess of blasts in transformation. Clinical, hematologic, and cytogenetic findings in nine patients. | Smadja N et al |
| 8618437 | 1996 | Clinical and morphological features of cases of trisomy 13 in acute non-lymphocytic leukemia. | Soni M et al |
| 11746988 | 2002 | Identification of cytogenetic subclasses and recurring chromosomal aberrations in AML and MDS with complex karyotypes using M-FISH. | Van Limbergen H et al |
| 1636249 | 1992 | Chromosome analyses in patients with myelodysplastic syndromes: correlation with bone marrow histopathology and prognostic significance. | Werner M et al |
| 11516096 | 2001 | Molecular cytogenetic characterization and clinical relevance of additional, complex and/or variant chromosome abnormalities in acute promyelocytic leukemia. | Xu L et al |
Summary
Note
Citation
Soad Al Bahar ; Adriana Zamecnikova
dup (11q) in myeloid malignancies
Atlas Genet Cytogenet Oncol Haematol. 2016-11-01
Online version: http://atlasgeneticsoncology.org/haematological/1629/dup-(11q)-in-myeloid-malignancies
